Abstract
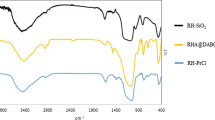
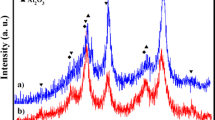
This work presents an efficient, easy and green method for the benzimidazoles and benzothiazoles synthesis by a condensation reaction of 1,2-diamino benzene and 2-amino thiophenol with various aromatic aldehydes using SnP2O7 as a new heterogeneous catalyst. This catalyst was prepared by adding MAP to the solution of SnCl2 and characterized by microscopic and spectroscopic methods namely X-ray diffraction, scanning electron microscopy and infrared which proved that the chemical structure of MAP has been modified when SnCl2 was added leading to the formation of SnP2O7. On the other hand, the reaction conditions were optimized namely the nature of the solvent and the mass of the catalyst. The high yields (85–97%) and very short reaction times (5–40 min) obtained for various benzimidazoles and benzothiazoles derivatives synthesis show that the new catalyst is very interesting because of its good catalytic activity and its reusability for at least five cycles without any degradation of its activity.









Similar content being viewed by others
References
Bahammou I, Esaady A, Boukhris S, Ghailane R, Habbadi N, Hassikou A, Souizi A (2016) Mediterr J Chem 5:615–623
Sibous S, Boukhris S, Ghailane R, Habbadi N, Hassikou A, Souizi A (2017) J Turk Chem Soc Sect Chem 4:477–488
Chen G, Liu Z, Zhang Y, Shan X, Jiang L, Zhao Y, He W, Feng Z, Yang S, Liang G (2013) ACS Med Chem Lett 4:69–74
Ibrahim E-SA, Omar A-MME, Khalil MA (1980) J Pharm Sci 69:1348–1350
Mathew B, Suresh J, Anbazhagan S (2016) J Saudi Chem Soc 20:S132–S139
Hameed PS, Raichurkar A, Madhavapeddi P, Menasinakai S, Sharma S, Kaur P, Nandishaiah R, Panduga V, Reddy J, Sambandamurthy VK, Sriram D (2014) ACS Med Chem Lett 5:820–825
Fang B, Zhou C-H, Rao X-C (2010) Eur J Med Chem 45:4388–4398
Shingalapur RV, Hosamani KM, Keri RS, Hugar MH (2010) Eur J Med Chem 45:1753–1759
MorningstarM L, Roth T, Farnsworth DW, Kroeger Smith M, Watson K, Buckheit RW, Das K, Zhang W, Arnold E, Julias JG, Hughes SH, Michejda CJ (2007) J Med Chem 50:4003–4015
Boiani M, Gonzalez M (2005) Mini-Rev Med Chem 5:409–424
Zarrinmayeh H, Zimmerman DM, Cantrell BE, Schober DA, Bruns RE, Gackenheimer SL, Ornstein PL, HipskindP A, Britton TC, Gehlert DR (1999) Bioorg Med Chem Lett 9:647–652
Camacho J, Barazarte A, Gamboa N, Rodrigues J, Rojas R, Vaisberg A, Gilman R, Charris J (2011) Bioorg Med Chem 19:2023–2029
Heynderickx A, Guglielmetti R, Dubest R, Aubard J, Samat A (2003) Synthesis 2003:1112–1116
Budow S, Kozlowska M, Gorska A, Kazimierczuk Z, Eickmeier H, LaColla P, Gosselin G, Seela F (2009) Arkivoc iii:225–250
Guruswamy B, Arul R (2011) Pharma Chem 3:483–486
Khan AT, Parvin T, Choudhury LH (2009) Synth Commun 39:2339–2346
Xiangming H, Huiqiang M, Yulu W (2007) Arkivocx iii:150–154
Gogoi P, Konwar D (2006) Tetrahedron Lett 47:79–82
Sammaiah B, Sumalatha D, Reddy G, Rajeswari M, Sharada L (2012) Int J Ind Chem 3:11–14
Varala R, Nasreen A, Enugala R, Adapa SR (2007) Tetrahedron Lett 48:69–72
Shelkar R, Sarode S, Nagarkar J (2013) Tetrahedron Lett 54:6986–6990
Alinezhad H (2014) Zare M Bulg. Chem Commun 46:347–352
Singh MP, Sasmal S, Lu W, Chatterjee MN (2000) Synthesis 2000:1380–1390
Trivedi R, De SK, Gibbs RA (2006) J Mol Catal Chem 245:8–11
Chari MA, Sadanandam P, Shobha D, Mukkanti K (2010) J Heterocycl Chem 47:153–155
Mukhopadhyay C, Datta A (2009) J Heterocycl Chem 46:91–95
Benzekri Z, Serrar H, Sibous S, Boukhris S, Ouasri A, Rhandour A, Souizi A (2016) Green Chem Lett Rev 9:223–228
Zakeri M, Moghadam M, Mirkhani V, Tangestaninejad S, Mohammadpoor-Baltork I, Pahlevanneshan Z (2018) Appl Organomet Chem 32:e3937
Sharghi H, Aberi M, Doroodmand MM, Shiri P (2017) J Iran Chem Soc 14:1557–1573
Kommula D, Madugula SRM (2017) J Iran Chem Soc 14:1665–1671
Kumar P, Bhatia R, Khanna R, Dalal A, Kumar D, Surain P, Kamboj RC (2017) J Sulfur Chem 38:585–596
Goswami M, Dutta MM, Phukan P (2018) Res Chem Intermed 44:1597–1615
Ye L, Chen J, Mao P, Mao Z, Zhang X, Yan M (2017) Tetrahedron Lett 58:874–876
Jiang Y, Jia S, Li X, Sun Y, Li W, Zhang W, Xu G (2018) Chem Pap 72:1265–1276
Jithendra Kumara KS, Krishnamurthy G, Sunil Kumar N, Naik N, Praveen T (2018) J Magn Magn Mater 451:808–821
Srivastava A, Shukla G, Yadav D, Singh MS (2018) Arkivoc 2:81–89
Feizpour F, Jafarpour M, Rezaeifard A (2018) Catal Lett 148:30–40
Shojaee S, Mahdavi Shahri M (2018) Appl Organomet Chem 32:e3934
Merroun Y, Chehab S, Ghailane T, Boukhris S, Ghailane R, Habbadi N, Hassikou A, Lakhrissi B, Souizi A (2018) J Turk Chem Soc Sect Chem 5:303–316
Sibous S, Ghailane T, Houda S, Ghailane R, Boukhris S, Souizi A (2017) Mediterr J Chem 6:53–59
Wang H, Chen J, Qiao R (2014) Ceram Int 40:8465–8468
Huang M, Huang X, Fei M, Deng Y, Huang X, Xu C, Cheng J, Luo P, Lu Y (2017) Int J Electrochem Sci 12:4585–4596
Attidekou PS, Connor P, Wormald P, Tunstall D, Francis S, Irvine JTS (2004) Solid State Ion 175:185–190
Huang C-H, Knop O, Othen DA, Woodhams FWD, Howie RA (1975) Can J Chem 53:79–91
Safaei-Ghomi J, Teymuri R, Shahbazi-Alavi H, Ziarati A (2013) Chin Chem Lett 24:921–925
Li L, Zong F, Cui X, Ma H, Wu X, Zhang Q, Wang Y, Yang F, Zhao J (2007) Mater Lett 61:4152–4155
Uzairu JOA, Gimba CE (2010) J Environ Chem Ecotoxicol 2:126–130
Nandeshwarappa BP, Manjappa S, Kishore B (2011) J Sulfur Chem 32:475–481
Gover RK, Withers ND, Allen S, Withers RL, Evans JSO (2002) J Solid State Chem 166:42–48
Fayon F, King IJ, Harris RK, Gover RKB, Evans JSO, Massiot D (2003) Chem Mater 15:2234–2239
Bezza I, Kaus M, Riekehr L, Pfaffmann L, Doyle S, Indris S, Ehrenberg H, Solhy A, Saadoune I (2016) Phys Chem Chem Phys 18:10375–10382
Patil MA, Ubale PA, Karhale SS, Helavi VB (2017) Medium Chem Sin 8:198–205
Teimouri A, Chermahini AN, Salavati H, Ghorbanian L (2013) J Mol Catal Chem 373:38–45
Sharghi H, Asemani O (2009) Synth Commun 39:860–867
Mao Z, Wang Z, Li J, Song X, Luo Y (2010) Synth Commun 40:1963–1977
Shingalapur RV, Hosamani KM (2010) Catal Lett 137:63–68
Mayo MS, Yu X, Zhou X, Feng X, Yamamoto Y, Bao M (2014) Org Lett 16:764–767
Martins GM, Puccinelli T, Gariani RA, Xavier FR, Silveira CC, Mendes SR (2017) Tetrahedron Lett 58:1969–1972
Ziarati A, Sobhani-Nasab A, Rahimi-Nasrabadi M, Ganjali MR, Badiei A (2017) J Rare Earths 35:374–381
Bardajee GR, Mohammadi M, Yari H, Ghaedi A (2016) Chin Chem Lett 27:265–270
Riadi Y, Mamouni R, Azzalou R, Haddad ME, Routier S, Guillaumet G, Lazar S (2011) Tetrahedron Lett 52:3492–3495
Chari MA, Shobha D, Kenawy E-R, Al-Deyab SS, Reddy BVS, Vinu A (2010) Tetrahedron Lett 51:5195–5199
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Merroun, Y., Chehab, S., Ghailane, T. et al. Preparation of tin-modified mono-ammonium phosphate fertilizer and its application as heterogeneous catalyst in the benzimidazoles and benzothiazoles synthesis. Reac Kinet Mech Cat 126, 249–264 (2019). https://doi.org/10.1007/s11144-018-1446-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-018-1446-5