Abstract
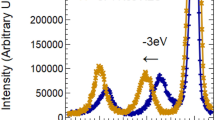
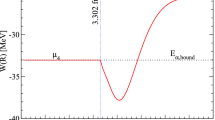
Fine structure of the X-ray photoelectron spectrum of UO2 at electron binding energies from 0 to ∼40 eV is primarily due to electrons of outer (0–15 eV) and inner (15–40 eV) valence molecular orbitals formed from unoccupied U5f, 6d, 7s and O2p and occupied low-energy U6p and O2s shells of the neighboring uranium and oxygen atoms, respectively. This is consistent with the results of the relativistic calculation of the electronic structure of the UO 12−8 cluster with O h symmetry, simulating the nearest surrounding of uranium in UO2, and is confirmed by the data of X-ray spectroscopy (conversion electron, nonresonance and resonance X-ray O 4,5(U) emission, O 4,5(U) XANES, photoelectron resonance, and Auger spectroscopy of oxygen). The fine structure of the X-ray photoelectron spectra, associated with electrons from outer valence and inner valence molecular orbitals, allows estimation of the degree of participation of U6p, 5f electrons in chemical bonding, as well as the structure of the nearest surrounding of the uranium atom and the bond length in its oxides. The total contribution from electrons of inner valence molecular orbitals to the absolute value of the chemical bonding energy can be compared with the corresponding contribution of the electrons from outer valence molecular orbitals to bonding of the atoms. Inner valence molecular orbitals can be formed in compounds of any elements, and this is an important new fact in chemistry and physics of condensed state.
Similar content being viewed by others
REFERENCES
Teterin, Yu. A. and Gagarin, S. G., Usp. Khim., 1996, vol. 65, no.10, pp. 825–847.
Teterin, Yu. A. and Teterin, A. Yu., Usp. Khim., 2002, vol. 71, no.5, pp. 403–441.
Teterin, Yu. A. and Teterin, A. Yu., Usp. Khim., 2004, vol. 73, no.6, pp. 588–631.
Teterin, Yu. A., Kulakov, V. M., Baev, A. S., et al., Phys. Chem. Miner., 1981, vol. 7, pp. 151–158.
Grechukhin, D. P., Zhudov, V. I., Zelenkov, A. G., et al., Pis'ma Zh. Eksp. Teor. Fiz., 1980, vol. 31, no.11, pp. 627–630.
Teterin, Yu. A., Terechov, V. A., Teterin, A. Yu., et al., J. Electron Spectrosc. Relat. Phenom., 1998, vol. 96, pp. 229–236.
Ivanov, K. E., Teterin, Yu. A., Shuh, D. K., et al., Proc. 4th Int. Yugoslav Nuclear Society Conf. (YUNSC-2002), Belgrade (Yugoslavia), September 30–October 4, 2002, Belgrade: VINCA Inst. of Nuclear Sciences, Yugoslav Nuclear Society, 2003, pp. 431–434.
Teterin, Yu. A., Utkin, I. O., Teterin, A. Yu., et al., Annual Report of BESSY GmbH, Berlin (Germany), 2004, no. 12 489.
Teterin, Yu. A, Maslakov, K. I., Ryzhkov, M. V., et al., Radiokhimiya, 2005, vol. 47, no.3, pp. 193–202.
Ryzhkov, M. V., Gubanov, V. A., Teterin, Yu. A., and Baev, A. S., Radiokhimiya, 1991, vol. 33, no.1, pp. 22–28.
Band, I. M., Kharitonov, Yu. I., and Trzhaskovskaya, M. B., At. Data Nucl. Data Tables, 1979, vol. 23, pp. 443–505.
Lyakhovskaya, I. I., Ipatov, V. M., and Zimkina, T. M., Zh. Strukt. Khim., 1977, vol. 18, no.4, pp. 668–672.
Zimkina, T. M., Lyakhovskaya, I. I., and Shulakov, A. S., Opt. Spektrosk., 1987, vol. 62, no.2, pp. 285–288.
Imoto, S., Miyake, C., Adachi, H., et al., Abstracts of Papers, Actinides-1981, California: LBL and LLNL Berkeley, September 10–15, 1981, pp. 99–101.
Cox, L., Ellis, W. P., Cowan, R., et al., Phys. Rev. B, 1987, vol. 35, no.11, pp. 5761–5765.
Teterin, Yu. A., Ivanov, K. E., Teterin, A. Yu., et al., J. Electron Spectrosc. Relat. Phenom., 1999, vols. 101–103, pp. 401–405.
Author information
Authors and Affiliations
Additional information
__________
Translated from Radiokhimiya, Vol. 47, No. 5, 2005, pp. 401–407.
Original Russian Text Copyright © 2005 by Yu. Teterin, A. Teterin.
Rights and permissions
About this article
Cite this article
Teterin, Y.A., Teterin, A.Y. X-Ray Spectral Methods in Investigation of the UO2 Electronic Structure. Radiochemistry 47, 440–446 (2005). https://doi.org/10.1007/s11137-005-0114-6
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11137-005-0114-6