Abstract
Purpose
To investigate the statistical relationship between the OM8-30 health-related quality of life measure for children with otitis media with effusion (OME) and measures of health utility (Health Utilities Index [HUI] Mark 3 and Mark 2) and to develop models to estimate HUI3 and HUI2 health utilities from OM8-30 scores.
Methods
A placebo-controlled, randomised trial (GNOME) evaluating intranasal mometasone in 217 children with OME provided concurrent responses to OM8-30 and HUI at three time points. Ordinary least squares (OLS), generalised linear models and two-step regression analyses were used to predict HUI3 and HUI2 utilities based on OM8-30 facet and domain scores.
Results
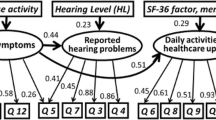
OLS models including all nine OM8-30 facets with or without predicted hearing level (HL) produced the best predictions of HUI3 utilities (mean absolute error: 0.134 with HL and 0.132 without; R 2: 0.63 with HL and 0.596 without). An OLS model predicting HUI3 utilities based on the two OM8-30 domain scores, reported hearing difficulties, predicted HL, age and sex also produced accurate predictions.
Conclusion
Regression equations predicting HUI3 and HUI2 utilities based on OM8-30 facet and domain scores have been developed. These provide an empirical basis for estimating quality-adjusted life years (QALYs) for interventions in children with OME.


Similar content being viewed by others
Abbreviations
- ACET:
-
Air conduction estimated from tympanometry
- AIC:
-
Akaike information criterion
- DEV:
-
Developmental domain of the OM8-30
- ENT:
-
Ear, nose and throat
- GLM:
-
Generalised linear model(s)
- GNOME:
-
GPRF [General Practice Research Framework] Nasal steroids for Otitis Media with Effusion
- HL:
-
Hearing level
- HRQoL:
-
Health-related quality of life
- HUI:
-
Health Utilities Index
- MAE:
-
Mean absolute error
- MAEVal :
-
Mean absolute error for the validation dataset
- OLS:
-
Ordinary least squares [regression]
- OM8-30:
-
Otitis media questionnaire
- OME:
-
Otitis media with effusion
- PHYS:
-
Physical health domain of the OM8-30
- PQoL:
-
Parent quality of life
- QALY:
-
Quality-adjusted life-year
- RHD:
-
Reported hearing difficulties
- RMSE:
-
Root mean squared error
- SD:
-
Standard deviation
References
Kind, P. (2001). Measuring quality of life in evaluating clinical interventions: An overview. Annals of Medicine, 33(5), 323–327.
von Neumann, J., & Morgenstern, O. (1953). Theory of games and economic behavior (3rd ed.). New York: Wiley.
Torrance, G. W. (1987). Utility approach to measuring health-related quality of life. Journal of Chronic Diseases, 40(6), 593–603.
Drummond, M. F., O’Brien, B., Stoddart, G. L., & Torrance, G. W. (1997). Chapter 6: Cost-utility analysis. Methods for the economic evaluation of health care programmes (2nd ed.). New York: Oxford Medical Publications.
Weinstein, M. C., & Stason, W. B. (1977). Foundations of cost-effectiveness analysis for health and medical practices. New England Journal of Medicine, 296(13), 716–721.
National Institute for Health and Clinical Excellence. (2008). Guide to the methods of technology appraisal. (http://www.nice.org.uk/media/B52/A7/TAMethodsGuideUpdatedJune2008.pdf).
Canadian Agency for Drugs and Technologies in Health. (2006). Guidelines for economic evaluation of pharmaceuticals: Canada. 3rd ed. Ottawa: Canadian Agency for Drugs and Technologies in Health (CADTH) (http://cadth.ca/media/pdf/186_EconomicGuidelines_e.pdf).
Scottish Medicines Consortium. (2007). Guidance to manufacturers for completion of new product assessment form (NPAF). (http://www.scottishmedicines.org.uk/smc/files/New%20Product%20Assessment%20Form%20(NPAF)Guidance%20NotesCombinedClinical%20and%20Economic(Master%20updated%20June%2007)%20FINAL.doc).
Brazier, J. E., Yang, Y., Tsuchiya, A., & Rowen, D. L. (2009). A review of studies mapping (or cross walking) non-preference based measures of health to generic preference-based measures. The European Journal of Health Economics. doi:10.1007/s10198-009-0168-z.
Nichol, M. B., Sengupta, N., & Globe, D. R. (2001). Evaluating quality-adjusted life years: Estimation of the health utility index (HUI2) from the SF-36. Medical Decision Making, 21(2), 105–112.
Franks, P., Lubetkin, E. I., Gold, M. R., Tancredi, D. J., & Jia, H. (2004). Mapping the SF-12 to the EuroQol EQ-5D Index in a national US sample. Medical Decision Making, 24(3), 247–254.
Gray, A. M., Rivero-Arias, O., & Clarke, P. M. (2006). Estimating the association between SF-12 responses and EQ-5D utility values by response mapping. Medical Decision Making, 26(1), 18–29.
Grootendorst, P., Marshall, D., Pericak, D., Bellamy, N., Feeny, D., & Torrance, G. W. (2007). A model to estimate health utilities index mark 3 utility scores from WOMAC index scores in patients with osteoarthritis of the knee. Journal of Rheumatology, 34(3), 534–542.
Lawrence, W. F., & Fleishman, J. A. (2004). Predicting EuroQoL EQ-5D preference scores from the SF-12 Health Survey in a nationally representative sample. Medical Decision Making, 24(2), 160–169.
Kulkarni, A. V. (2006). Distribution-based and anchor-based approaches provided different interpretability estimates for the Hydrocephalus Outcome Questionnaire. Journal of Clinical Epidemiology, 59(2), 176–184.
Zielhuis, G. A., Rach, G. H., & van den Broek, P. (1989). Screening for otitis media with effusion in preschool children. Lancet, 1(8633), 311–314.
Casselbrant, M. L., Brostoff, L. M., Cantekin, E. I., Flaherty, M. R., Doyle, W. J., Bluestone, C. D., et al. (1985). Otitis media with effusion in preschool children. Laryngoscope, 95(4), 428–436.
Higson, J., & Haggard, M. (2005). Parent versus professional views of the developmental impact of a multi-faceted condition at school age: otitis media with effusion (‘glue ear’). British Journal of Educational Psychology, 75(Pt 4), 623–643.
Mathers, C. D., Lopez, A. D., & Murray, C. J. L. (2006). Chapter 3: The Burden of disease and mortality by condition: Data, methods, and results for 2001. Global burden of disease and risk factors (pp. 45–93). New York: Oxford University Press.
Oh, P. I., Maerov, P., Pritchard, D., Knowles, S. R., Einarson, T. R., & Shear, N. H. (1996). A cost-utility analysis of second-line antibiotics in the treatment of acute otitis media in children. Clinical Therapeutics, 18(1), 160–182.
Kubba, H., Swan, I. R., & Gatehouse, S. (2004). How appropriate is the OM6 as a discriminative instrument in children with otitis media? Archives of Otolaryngology - Head and Neck Surgery, 130(6), 705–709.
Kubba, H., Swan, I. R., & Gatehouse, S. (2005). Measuring quality of life in preschool children with sore throats and otitis media using the TAPQOL questionnaire. Otolaryngology - Head and Neck Surgery, 132(4), 647–652.
Petrou, S. (2003). Methodological issues raised by preference-based approaches to measuring the health status of children. Health Economics, 12(8), 697–702.
Timmerman, A. A., Meesters, C. M., Speyer, R., & Anteunis, L. J. (2007). Psychometric qualities of questionnaires for the assessment of otitis media impact. Clinical Otolaryngology, 32(6), 429–439.
Timmerman, A. A., Meesters, C. M., Anteunis, L. J., Chenault, M. N., & Haggard, M. P. (2008). Psychometric evaluation of the OM8–30 questionnaire in Dutch children with otitis media. European Archives of Oto-Rhino-Laryngology, 265(9), 1047–1056.
Williamson, I., & Little, P. (2008). Otitis media with effusion: the long and winding road? Archives of Disease in Childhood, 93(4), 268–269.
Gray, R., Petrou, S., Hockley, C., & Gardner, F. (2007). Self-reported health status and health-related quality of life of teenagers who were born before 29 weeks’ gestational age. Pediatrics, 120(1), e86–e93.
Williamson, I., Benge, S., Barton, S., Petrou, S., Letley, L., & Fasey, N., et al. (2009). A double-blind randomised placebo controlled trial of topical intra-nasal corticosteroids in 4–11 year old children with persistent bilateral otitis media with effusion in primary care. Health Technology Assessment 13(37), 1–144.
Williamson, I., Benge, S., Barton, S., Petrou, S., Letley, L., & Fasey, N., et al. (2009). A double-blind randomised placebo controlled trial of topical intra-nasal corticosteroids in 4–11 year old children with persistent bilateral Otitis Media with Effusion in primary care. BMJ (in press).
Haggard, M., Spencer, H., & Gregori, M. (2007). Internationally Standardised Clinical Description: Can It Be Achieved ENT? ENT News, 16(1), 69–72.
Furlong, W., Feeny, D., Torrance, G. W., Goldsmith, C. H., DePauw, S., & Zhu, Z., et al. (1998). Multiplicative multi-attribute utility function for the health utilities index mark 3 (HUI3) system: A Technical Report. CHEPA Working Paper Series 98-11.
Feeny, D., Furlong, W., Torrance, G. W., Goldsmith, C. H., Zhu, Z., DePauw, S., et al. (2002). Multiattribute and single-attribute utility functions for the health utilities index mark 3 system. Medical Care, 40(2), 113–128.
Torrance, G. W., Feeny, D. H., Furlong, W. J., Barr, R. D., Zhang, Y., & Wang, Q. (1996). Multiattribute utility function for a comprehensive health status classification system. Health Utilities Index Mark 2. Medical Care, 34(7), 702–722.
Torrance, G. W., Feeny, D. H., Furlong, W. J., Barr, R. D., Zhang, Y., & Wang, Q. (2000). Influence of age, type of audiometry and child’s concentration on hearing thresholds. MRC Multi-centre Otitis Media Study Group. British Journal of Audiology, 34(4), 231–240.
Torrance, G. W., Feeny, D. H., Furlong, W. J., Barr, R. D., Zhang, Y., & Wang, Q. (2001). Surgery for persistent otitis media with effusion: generalizability of results from the UK trial (TARGET). Trial of Alternative Regimens in Glue Ear Treatment. Clin Otolaryngol. Allied Sciences, 26(5), 417–424.
Torrance, G. W., Feeny, D. H., Furlong, W. J., Barr, R. D., Zhang, Y., & Wang, Q. (1999). Sensitivity, specificity and predictive value of tympanometry in predicting a hearing impairment in otitis media with effusion. MRC Multi-Centre Otitis Media Study Group. Clin Otolaryngol. Allied Sciences, 24(4), 294–300.
Haggard, M., Spencer, H., & Gregori, M. (2006). OM8-30 for assessment and outcome in OME: origins, applications & ultra-short OM2-13 (http://www.bacda.org.uk/Revised%20Jan06%20BACDA%20with%20OM2-131.ppt). Presented at the BACDA 20th Anniversary Meeting, London, 27 January.
Haggard, M., Spencer, H., & Gregori, M. (2007). Overcoming international differences for clinical comparisons and research. ENT News, 16(3), 68–71.
MRC Multi-centre Otitis Media Study Group. (2009). Air-conduction estimated from tympanometry (ACET) 1: relationship to measured hearing in OME. International Journal of Pediatric Otorhinolaryngology, 73(1), 21–42.
Furlong, W. J., Feeny, D. H., Torrance, G. W., & Barr, R. D. (2001). The Health Utilities Index (HUI) system for assessing health-related quality of life in clinical studies. Annals of Medicine, 33(5), 375–384.
Cox, C. L., Lensing, S., Rai, S. N., Hinds, P., Burghen, E., & Pui, C. H. (2005). Proxy assessment of quality of life in pediatric clinical trials: Application of the health utilities index 3. Quality of Life Research, 14(4), 1045–1056.
Eiser, C., & Morse, R. (2001). The measurement of quality of life in children: past and future perspectives. Journal of Developmental and Behavioral Pediatrics, 22(4), 248–256.
Eiser, C., & Morse, R. (2001). A review of measures of quality of life for children with chronic illness. Archives of Disease in Childhood, 84(3), 205–211.
Sung, L., Greenberg, M. L., Doyle, J. J., Young, N. L., Ingber, S., Rubenstein, J., et al. (2003). Construct validation of the health utilities index and the child health questionnaire in children undergoing cancer chemotherapy. British Journal of Cancer, 88(8), 1185–1190.
Barr, R. D., Petrie, C., Furlong, W., Rothney, M., & Feeny, D. (1997). Health-related quality of life during post-induction chemotherapy in children with acute lymphoblastic leukemia in remission: an influence of corticosteroid therapy. International Journal of Oncology, 11, 333–339.
Trudel, J. G., Rivard, M., Dobkin, P. L., Leclerc, J. M., & Robaey, P. (1998). Psychometric properties of the Health Utilities Index Mark 2 system in paediatric oncology patients. Quality of Life Research, 7(5), 421–432.
Drummond, M. F., Sculpher, M. J., Torrance, G. W., O’Brien, B. J., & Stoddart, G. L. (2005). Methods for the economic evaluation of health care programmes (3rd ed.). Oxford: Oxford University Press.
Levy, A. R., Kowdley, K. V., Iloeje, U., Tafesse, E., Mukherjee, J., Gish, R., et al. (2008). The impact of chronic hepatitis B on quality of life: a multinational study of utilities from infected and uninfected persons. Value Health, 11(3), 527–538.
Le Gales, C., Buron, C., Costet, N., Rosman, S., & Slama, P. R. (2002). Development of a preference-weighted health status classification system in France: the Health Utilities Index 3. Health Care Management Science, 5(1), 41–51.
StataCorp. (2008). Stata version 10.0 (http://www.stata.com). College Station, Texas.
StataCorp. (2007). Regress—Linear regression. Stata base reference manual: Release 10 (pp. 79–101). College Station, TX: StataCorp LP.
StataCorp. (2007). 20.15: Obtaining robust variance estimates. Stata base reference manual: release 10 (pp. 268–273). College Station, TX: StataCorp LP.
StataCorp. (2007). GLM—Generalised linear models. Stata base reference manual: Release 10 (pp. 493–522). College Station, TX: StataCorp LP.
Bozdogan, H. (1987). Model selection and Akaike’s information criterion (AIC): The general theory and its analytical extensions. Psychometrika, 52(3), 345–370.
Feeny, D., Furlong, W., Saigal, S., & Sun, J. (2004). Comparing directly measured standard gamble scores to HUI2 and HUI3 utility scores: group- and individual-level comparisons. Social Science and Medicine, 58(4), 799–809.
Stade, B. C., Stevens, B., Ungar, W. J., Beyene, J., & Koren, G. (2006). Health-related quality of life of Canadian children and youth prenatally exposed to alcohol. Health Qual Life Outcomes, 4, 81.
Haggard, M., Spencer, H., Williamson, I., & Benge, S. E. (2008). Caseloads in otitis media: effects of selective referral to secondary care on severity of cases in UK & elsewhere. Podium presentation at the European Society of Paediatric ORL, Budapest June 10, 2008.
Tsuchiya, A., Brazier, J., McColl, E., & Parkin, D. (2002). Deriving preference-based single indices from non- preference based condition specific instruments: converting AQLQ into EQ-5D indices. HEDS discussion paper.
Knies, S., Evers, S. M., Candel, M. J., Severens, J. L., & Ament, A. J. (2009). Utilities of the EQ-5D: Transferable or Not? Pharmacoeconomics, 27(9), 767–779.
Grutters, J. P., Joore, M. A., van der Horst, F., Verschuure, H., Dreschler, W. A., & Anteunis, L. J. (2007). Choosing between measures: comparison of EQ-5D, HUI2 and HUI3 in persons with hearing complaints. Quality of Life Research, 16(8), 1439–1449.
Acknowledgments
The authors would like to thank all researchers who were involved in the GNOME trial, in addition to the children and parents who participated. In particular, we would like to thank Giselle Abangma for preparing the dataset of HUI utilities, Helen Spencer for preparing datasets of OM8-30 question, facet and domain scores and predicted hearing levels and the two anonymous reviewers for their helpful comments on the manuscript. This project was funded by the UK National Institute for Health Research Health Technology Assessment Programme (project number 01/72/02). The views and opinions expressed are those of the authors and do not necessarily reflect those of the Department of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dakin, H., Petrou, S., Haggard, M. et al. Mapping analyses to estimate health utilities based on responses to the OM8-30 otitis media questionnaire. Qual Life Res 19, 65–80 (2010). https://doi.org/10.1007/s11136-009-9558-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-009-9558-z