Abstract
Aims
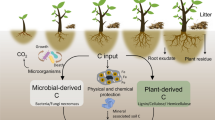
Lignin has long been considered a recalcitrant component of plant materials. Its degradation is critical for litter decomposition and the formation of soil organic matter. However, little is known about lignin degradation dynamics and its controlling factors during leaf litter decomposition, especially in forest ecosystems. The objective of this study was to evaluate lignin degradation dynamics during litter decomposition and its controlling factors.
Methods
We conducted a lab incubation experiment for 180 days with leaves of two tree species (Fagus lucida and Schima parviflora) from a mid-subtropical forest in China. The CuO oxidation method was applied for lignin determination. The mass loss of bulk litter and lignin (including V-, S- and C-type phenols) and the dynamics of litter microbial community composition, carbon, nitrogen (N) and phosphorus (P) were determined during the incubation.
Results
Lignin degradation appeared to be a two-phase process over the incubation period, including a fast decomposition phase (early stage) followed by a relatively slow decomposition phase (late stage). Additionally, 21.9% and 18.5% of the initial lignin contents were lost from F. lucida and S. parviflora litter, respectively. The acid to aldehyde ratios of V-type phenols [(Ac/Al)V] and S-type phenols [(Ac/Al)S] of both litters increased with incubation. Lignin degradation was significantly affected by the litter types but not by the soil microbial communities, given that the soil microbial communities were distinctly different for the two plant communities. In the early stage, the mass loss of lignin from the F. lucida litter was faster than the S. parviflora litter: the mass loss of S-type phenols from the F. lucida litter was faster than the S. parviflora litter, whereas the dynamics of the V-type phenols were comparable for the two species. For F. lucida litter, lignin was lost faster than the bulk litter in the early stage but no difference was observed after day 133. For S. parviflora litter, the degradation of lignin and bulk litter were generally comparable over the incubation period. The mass loss of lignin in the early stage was significantly correlated with lignin, N and P contents, and the carbon/N, carbon/P and N/P ratios. Among these factors, P played a prominent role in controlling lignin degradation.
Conclusions
The important role of litter chemistry emphasized the importance of taking this into account in plant-soil models assessing carbon dynamics in forest ecosystems. These results improve our conceptual understanding of lignin degradation and its controlling factors.






Similar content being viewed by others
References
Ainley LB, Bishop MJ (2015) Relationships between estuarine modification and leaf litter decomposition vary with latitude. Estuar Coast Shelf S 164:244–252
Arcand MM, Helgason BL, Lemke RL (2016) Microbial crop residue decomposition dynamics in organic and conventionally managed soils. Appl Soil Ecol 107:347–359
Asplund J, Kauserud H, Bokhorst S, Lie MH, Ohlson M, Nybakken L (2018) Fungal communities influence decomposition rates of plant litter from two dominant tree species. Funct Ecol 32:1–8
Ayres E, Steltzer H, Simmons BL, Simpson RT, Steinweg JM, Wallenstein MD, Mellor N, Parton WJ, Moore JC, Wall DH (2009) Home-field advantage accelerates leaf litter decomposition in forests. Soil Biol Biochem 41:606–610
Bahri H, Dignac M-F, Rumpel C, Rasse DP, Chenu C, Mariotti A (2006) Lignin turnover kinetics in an agricultural soil is monomer specific. Soil Biol Biochem 38:1977–1988
Bahri H, Rasse DP, Rumpel C, Dignac MF, Bardoux G, Mariotti A (2008) Lignin degradation during a laboratory incubation followed by 13C isotope analysis. Soil Biol Biochem 40:916–1922
Bai Z, Liang C, Bodé S, Huygens D, Boeckx P (2016) Phospholipid 13C stable isotopic probing during decomposition of wheat residues. Appl Soil Ecol 98:65–74
Berg B, McClaugherty C (2003) Plant litter, decomposition, humus formation, carbon sequestration. Springer, Berlin
Berg B, McClaugherty C (2014) Decomposition as a process: some main features. Plant Litter. Springer, Berlin, Heidelberg, pp 11–34
Berg B, McClaugherty C, Johansson M-B (1993) Litter mass-loss rates in late stages of decomposition at some climatically and nutritionally different pine sites. Long-term decomposition in a scots pine forest. VIII. Can J Bot 71:680–692
Berg B, Staaf H (1980) Decomposition rate and chemical changes of scots pine needle litter. II Influence of chemical composition Ecol Bull:373–390
Boerjan W, Ralph J, Baucher M (2003) Lignin biosynthesis. Annu Rev Plant Biol 54:519–546
Bonner MTL, Shoo LP, Brackin R, Schmidt S (2018) Relationship between microbial composition and substrate use efficiency in a tropical soil. Geoderma 315:96–103
Bossio DA, Fleck JA, Scow KM, Fujii R (2006) Alteration of soil microbial communities and water quality in restored wetlands. Soil Biol Biochem 38:1223–1233
Bossio DA, Scow KM (1998) Impacts of carbon and flooding on soil microbial communities: phospholipid fatty acid profiles and substrate utilization patterns. Microb Ecol 35:265–278
Bugg TDH, Ahmad M, Hardiman EM, Singh R (2011) The emerging role for bacteria in lignin degradation and bio-product formation. Curr Opin Biotech 22:394–400
Cadisch G, Giller K E (1997) Driven by nature: plant litter quality and decomposition
Carreiro M, Sinsabaugh R, Repert D, Parkhurst D (2000) Microbial enzyme shifts explain litter decay responses to simulated nitrogen deposition. Ecology 81:2359–2365
Ceballos SJ, Yu C, Claypool JT, Singer SW, Simmons BA, Thelen MP, Simmons CW, VanderGheynst JS (2017) Development and characterization of a thermophilic, lignin degrading microbiota. Process Biochem 63:193–203
Cleveland CC, Reed SC, Keller AB, Nemergut DR, O'Neill SP, Ostertag R, Vitousek PM (2014) Litter quality versus soil microbial community controls over decomposition: a quantitative analysis. Oecologia 174:283–294
Coulis M, Hättenschwiler S, Rapior S, Coq S (2009) The fate of condensed tannins during litter consumption by soil animals. Soil Biol Biochem 41:2573–2578
Craine JM, Morrow C, Fierer N (2007) Microbial nitrogen limitation increases decomposition. Ecology 88:2105–2113
Dao TT, Gentsch N, Mikutta R, Sauheitl L, Shibistova O, Wild B, Schnecker J, Bárta J, Čapek P, Gittel A, Lashchinskiy N, Urich T, Šantrůčková H, Richter A, Guggenberger G (2018) Fate of carbohydrates and lignin in north-east Siberian permafrost soils. Soil Biol Biochem 116:311–322
DeAngelis KM, Allgaier M, Chavarria Y, Fortney JL, Hugenholtz P, Simmons B, Sublette K, Silver WL, Hazen TC (2011) Characterization of trapped lignin-degrading microbes in tropical forest soil. PLoS One 6:9
Duboc O, Dignac M-F, Djukic I, Zehetner F, Gerzabek MH, Rumpel C (2014) Lignin decomposition along an alpine elevation gradient in relation to physicochemical and soil microbial parameters. Glob Chang Biol 20:2272–2285
Ertel JR, Hedges JI (1984) The lignin component of humic substances: distribution among soil and sedimentary humic, fulvic, and base-insoluble fractions. Geochim Cosmochim Ac 48:2065–2074
Ferry L, Dorez G, Taguet A, Otazaghine B, Lopez-Cuesta J (2015) Chemical modification of lignin by phosphorus molecules to improve the fire behavior of polybutylene succinate. Polym Degrad Stabil 113:135–143
Fierer N, Schimel JP, Holden PA (2003) Variations in microbial community composition through two soil depth profiles. Soil Biol Biochem 35:167–176
Hättenschwiler S, Tiunov AV, Scheu S (2005) Biodiversity and litter decomposition in terrestrial ecosystems. Annu Rev Ecol Evol Syst 36:191–218
Hall SJ, Silver WL, Timokhin VI, Hammel KE (2015) Lignin decomposition is sustained under fluctuating redox conditions in humid tropical forest soils. Glob Chang Biol 21(7):2818–2828
He W, Wu F, Yang W, Tan B, Zhao Y, Wu Q, He M (2015) Lignin degradation in foliar litter of two shrub species from the gap center to the closed canopy in an alpine fir forest. Ecosystems 19:115–128
Hedges JI, Blanchette RA, Weliky K, Devol AH (1988) Effects of fungal degradation on the CuO oxidation products of lignin: a controlled laboratory study. Geochim Cosmochim Acta 52:2717–2726
Hedges JI, Ertel JR (1982) Characterization of lignin by gas capillary chromatography of cupric oxide oxidation products. Anal Chem 54:174–178
Heim A, Schmidt MWI (2007) Lignin turnover in arable soil and grassland analysed with two different labelling approaches. Eur J Soil Sci 58:599–608
Hilli S, Stark S, Willför S, Smeds A, Reunanen M, Hautajärvi R (2012) What is the composition of AIR? Pyrolysis-GC–MS characterization of acid-insoluble residue from fresh litter and organic horizons under boreal forests in southern Finland. Geoderma 179-180:63–72
Hu Z, Xu C, McDowell NG, Johnson DJ, Wang M, Luo Y, Zhou X, Huang Z (2017) Linking microbial community composition to C loss rates during wood decomposition. Soil Biol Biochem 104:108–116
Kögel-Knabner I (2002) The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter. Soil Biol Biochem 34:139–162
Kögel I (1986) Estimation and decomposition pattern of the lignin component in forest humus layers. Soil Biol Biochem 18:589–594
Kaspari M, Garcia MN, Harms KE, Santana M, Wright SJ, Yavitt JB (2008) Multiple nutrients limit litterfall and decomposition in a tropical forest. Ecol Lett 11:35–43
Kiem R, Kögel-Knabner I (2003) Contribution of lignin and polysaccharides to the refractory carbon pool in C-depleted arable soils. Soil Biol Biochem 35:101–118
Kirk TK, Chang H-m, Lorenz L (1975) Topochemistry of the fungal degradation of lignin in birch wood as related to the distribution of guaiacyl and syringyl lignins. Wood Sci Technol 9:81–86
Kirk TK, Farrell RL (1987) Enzymatic "combustion": the microbial degradation of lignin. Ann Rev Microbiol 41:465–501
Klotzbücher T, Kaiser K, Guggenberger G, Gatzek C, Kalbitz K (2011) A new conceptual model for the fate of lignin in decomposing plant litter. Ecology 92:1052–1062
Kourtev PS, Ehrenfeld JG, Huang WZ (2002) Enzyme activities during litter decomposition of two exotic and two native plant species in hardwood forests of New Jersey. Soil Biol Biochem 34:1207–1218
Li H, Wu FZ, Yang WQ, Xu LY, Ni XY, He J, Tan B, Hu Y (2016) Effects of forest gaps on litter lignin and cellulose dynamics vary seasonally in an alpine forest. Forests 7:17
Liu G, Cornwell WK, Pan X, Ye D, Liu F, Huang Z, Dong M, Cornelissen JH (2015) Decomposition of 51 semidesert species from wide-ranging phylogeny is faster in standing and sand-buried than in surface leaf litters: implications for carbon and nutrient dynamics. Plant Soil 396:175–187
Lu W, Liu N, Zhang Y, Zhou J, Guo Y, Yang X (2017) Impact of vegetation community on litter decomposition: evidence from a reciprocal transplant study with 13 C labeled plant litter. Soil Biol Biochem 112:248–257
Makkonen M, Berg MP, Handa IT, Hättenschwiler S, van Ruijven J, van Bodegom PM, Aerts R (2012) Highly consistent effects of plant litter identity and functional traits on decomposition across a latitudinal gradient. Ecol Lett 15:1033–1041
Marklein AR, Winbourne JB, Enders SK, Gonzalez DJX, van Huysen TL, Izquierdo JE, Light DR, Liptzin D, Miller KE, Morford SL, Norton RA, Houlton BZ (2016) Mineralization ratios of nitrogen and phosphorus from decomposing litter in temperate versus tropical forests. Glob Ecol Biogeogr 25:335–346
McClaugherty C (1987) Cellulose, lignin and nitrogen concentrations as rate regulating factors in late stages of forest litter decomposition. Pedobiologia 30:101–112
Moorhead DL, Lashermes G, Sinsabaugh RL, Weintraub MN (2013) Calculating co-metabolic costs of lignin decay and their impacts on carbon use efficiency. Soil Biol Biochem 66:17–19
Norgren M, Edlund H (2014) Lignin: recent advances and emerging applications. Curr Opin Colloid In 19:409–416
Oksanen J, Blanchet F, Kindt R, Legendre P, O’Hara R (2016) Vegan: community ecology package. R Package 2:3–3
Otto A, Simpson MJ (2006) Evaluation of CuO oxidation parameters for determining the source and stage of lignin degradation in soil. Biogeochem 80:121–142
Pei Z, Leppert KN, Eichenberg D, Bruelheide H, Niklaus PA, Buscot F, Gutknecht JLM (2017) Leaf litter diversity alters microbial activity, microbial abundances, and nutrient cycling in a subtropical forest ecosystem. Biogeochem 134:163–181
Pourhassan N, Bruno S, Jewell MD, Shipley B, Roy S, Bellenger J-P (2016) Phosphorus and micronutrient dynamics during gymnosperm and angiosperm litters decomposition in temperate cold forest from eastern Canada. Geoderma 273:25–31
Preston CM, Nault JR, Trofymow JA (2009) Chemical changes during 6 years of decomposition of 11 litters in some Canadian Forest sites. Part 2. 13C abundance, solid-state 13C NMR spectroscopy and the meaning of “lignin”. Ecosystems 12:1078–1102
Rahman MM, Tsukamoto J, Rahman MM, Yoneyama A, Mostafa KM (2013) Lignin and its effects on litter decomposition in forest ecosystems. Chem Ecol 29:540–553
Ralph J, Helm RF, Quideau S, Hatfield RD (1992) Lignin–feruloyl ester cross-links in grasses. Part 1. Incorporation of feruloyl esters into coniferyl alcohol dehydrogenation polymers. J Chem Soc Perkin Trans 1:2961–2969
Rashid MI, Lantinga EA, Brussaard L, de Goede RGM (2017) The chemical convergence and decomposer control hypotheses explain solid cattle manure decomposition in production grasslands. Appl Soil Ecol 113:107–116
Rinkes ZL, Bertrand I, Amin BAZ, Grandy AS, Wickings K, Weintraub MN (2016) Nitrogen alters microbial enzyme dynamics but not lignin chemistry during maize decomposition. Biogeochem 128:171–186
Sanaullah M, Chabbi A, Lemaire G, Charrier X, Rumpel C (2009) How does plant leaf senescence of grassland species influence decomposition kinetics and litter compounds dynamics? Nutr Cycl Agroecosyst 88:159–171
Scalbert A, Monties B, Lallemand J-Y, Guittet E, Rolando C (1985) Ether linkage between phenolic acids and lignin fractions from wheat straw. Phytochem 24:1359–1362
Sjoberg G, Nilsson SI, Persson T, Karlsson P (2004) Degradation of hemicellulose, cellulose and lignin in decomposing spruce needle litter in relation to N. Soil Biol Biochem 36:1761–1768
Smith AP, Marin-Spiotta E, Balser T (2015) Successional and seasonal variations in soil and litter microbial community structure and function during tropical postagricultural forest regeneration: a multiyear study. Glob Chang Biol 21:3532–3547
St. John MG, Orwin KH, Dickie IA (2011) No ‘home’ versus ‘away’ effects of decomposition found in a grassland–forest reciprocal litter transplant study. Soil Biol Biochem 43:1482–1489
Sterner RW, Elser JJ (2002) Ecological stoichiometry: the biology of elements from molecules to the biosphere. Princeton University Press
Talbot JM, Treseder KK (2012) Interactions among lignin, cellulose, and nitrogen drive litter chemistry–decay relationships. Ecology 93:345–354
Talbot JM, Yelle DJ, Nowick J, Treseder KK (2011) Litter decay rates are determined by lignin chemistry. Biogeochem 108:279–295
Team RC (2016) R: a language and environment for statistical computing [online]. R Foundation for Statistical Computing, Vienna, Austria
Thevenot M, Dignac M-F, Rumpel C (2010) Fate of lignins in soils: a review. Soil Biol Biochem 42:1200–1211
Torres IF, Bastida F, Hernández T, Bombach P, Richnow HH, García C (2014) The role of lignin and cellulose in the carbon-cycling of degraded soils under semiarid climate and their relation to microbial biomass. Soil Biol Biochem 75:152–160
Veen GFC, Keiser AD, van der Putten WH, Wardle DA, Hart M (2018) Variation in home-field advantage and ability in leaf litter decomposition across successional gradients. Funct Ecol 00:1–12
Veen GFC, Sundqvist MK, Wardle DA, Briones MJ (2015) Environmental factors and traits that drive plant litter decomposition do not determine home-field advantage effects. Funct Ecol 29:981–991
Virzo De Santo A, De Marco A, Fierro A, Berg B, Rutigliano FA (2008) Factors regulating litter mass loss and lignin degradation in late decomposition stages. Plant Soil 318:217–228
Xu G, Chen J, Berninger F, Pumpanen J, Bai J, Yu L, Duan B (2015) Labile, recalcitrant, microbial carbon and nitrogen and the microbial community composition at two Abies faxoniana forest elevations under elevated temperatures. Soil Biol Biochem 91:1–13
Yang H, Effland M, Kirk TK (1980) Factors influencing fungal degradation of lignin in a representative lignocellulosic, thermomechanical pulp. Biotechnol Bioeng 22:65–77
Yue K, Peng C, Yang W, Peng Y, Zhang C, Huang C, Wu F (2016) Degradation of lignin and cellulose during foliar litter decomposition in an alpine forest river. Ecosphere 7:1–11
Zheng Z, Mamuti M, Liu H, Shu Y, Hu S, Wang X, Li B, Lin L, Li X (2017) Effects of nutrient additions on litter decomposition regulated by phosphorus-induced changes in litter chemistry in a subtropical forest, China. For Ecol Manag 400:123–128
Acknowledgements
This research was supported by the Natural Science Foundation of China (31470526, 31700462), and the Chinese National Key Development Program for Basic Research (2014CB954004).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Eric Paterson.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1578 kb)
Rights and permissions
About this article
Cite this article
He, M., Zhao, R., Tian, Q. et al. Predominant effects of litter chemistry on lignin degradation in the early stage of leaf litter decomposition. Plant Soil 442, 453–469 (2019). https://doi.org/10.1007/s11104-019-04207-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-04207-6