Abstract
Background and aims
The trend of soil degradation in intensive open coffee systems is well-documented. This study highlights the impact of young shade trees on soil quality only 4 years after their intercropping with coffee.
Methods
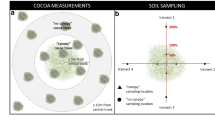
18 young shade trees belonging to three tree species (Cinnamomum camphora, Bishofia javanica and Jacaranda mimosifolia) were selected in an intensive coffee system in Southern Yunnan. Soil samples (0–20 cm) were tested for chemical composition, soil communities and soil enzyme activities under their canopies and in open areas, both in coffee rows and inter-rows, once during the rainy and once during the dry season. Additionally, root systems were characterized using trenches. Soil water profiles and litterfall were monitored along the production cycle. Coffee yield was recorded for two consecutive years.
Results
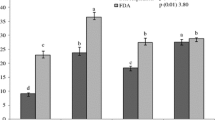
We detected a positive impact of all shade tree species on soil chemical, biological and biochemical components, especially during the dry season. This positive impact included higher soil organic matter (+10%) and more abundant soil microbial communities (+64%) under shaded coffee than under open coffee. Furthermore, shaded coffee trees yielded as much as open coffee trees, except under C. camphora, probably due to high below-ground competition.
Conclusions
These results demonstrate that carefully selected shade trees can rapidly contribute to preserving and/or restoring soil quality in intensive coffee systems, while maintaining high coffee yield.





Similar content being viewed by others
References
Adetunji AT, Lewu FB, Mulidzi R, Ncube B (2017) The biological activities of β-glucosidase, phosphatase and urease as soil quality indicators: a review. J Soil Sci Plant Nutr 17:794–807
Andrade SAL, Mazzafera P, Schiavinato MA, Silveira APD (2009) Arbuscular mycorrhizal association in coffee. Anglais 147:105–115
Bagyaraj DJ, Thilagar G, Ravisha C, Kushalappa CG, Krishnamurthy KN, Vaast P (2015) Below ground microbial diversity as influenced by coffee agroforestry systems in the Western Ghats, India. Agric Ecosyst Environ 202:198–202. https://doi.org/10.1016/j.agee.2015.01.015
Bainard LD, Klironomos JN, Gordon AM (2011) Arbuscular mycorrhizal fungi in tree-based intercropping systems: a review of their abundance and diversity. Pedobiologia 54:57–61. https://doi.org/10.1016/j.pedobi.2010.11.001
Bainard LD, Koch AM, Gordon AM, Klironomos JN (2013) Growth response of crops to soil microbial communities from conventional monocropping and tree-based intercropping systems. Plant Soil 363:345–356. https://doi.org/10.1007/s11104-012-1321-5
Barrios E (2007) Soil biota, ecosystem services and land productivity. Ecol Econ 64:269–285. https://doi.org/10.1016/j.ecolecon.2007.03.004
Barrios E, Sileshi G, Shepherd K, Sinclair F (2012) Agroforestry and soil health: linking trees, soil biota and ecosystem services. In: Wall DH (ed) Soil ecology and ecosystem services. Oxford University Press, Oxford, pp 315–330
Beer J, Muschler R, Kass D, Somarriba E (1998) Shade management in coffee and cacao plantations. Agrofor Syst 38:139–164. https://doi.org/10.1023/A:1005956528316
Borken W, Matzner E (2009) Reappraisal of drying and wetting effects on C and N mineralization and fluxes in soils. Glob Chang Biol 15:808–824. https://doi.org/10.1111/j.1365-2486.2008.01681.x
Bossio DA, Scow KM (1998) Impacts of carbon and flooding on soil microbial communities: phospholipid fatty acid profiles and substrate utilization patterns. Microb Ecol 35:265–278. https://doi.org/10.1007/s002489900082
Bowles TM, Acosta-Martínez V, Calderón F, Jackson LE (2014) Soil enzyme activities, microbial communities, and carbon and nitrogen availability in organic agroecosystems across an intensively-managed agricultural landscape. Soil Biol Biochem 68:252–262. https://doi.org/10.1016/j.soilbio.2013.10.004
Bremner JM, Mulvaney C (1982) Nitrogen-total. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis part 2 chemical and microbiological properties. American Society of Agronomy, Madison, pp 595–624
Cannavo P, Sansoulet J, Harmand JM, Siles P, Dreyer E, Vaast P (2011) Agroforestry associating coffee and Inga densiflora results in complementarity for water uptake and decreases deep drainage in Costa Rica. Agric Ecosyst Environ 140:1–13. https://doi.org/10.1016/j.agee.2010.11.005
Cannavo P, Harmand JM, Zeller B, Vaast P, Ramírez JE, Dambrine E (2013) Low nitrogen use efficiency and high nitrate leaching in a highly fertilized Coffea arabica–Inga densiflora agroforestry system: a 15N labeled fertilizer study. Nutr Cycl Agroecosyst 95:377–394. https://doi.org/10.1007/s10705-013-9571-z
Ciesielski H, Sterckeman T, Santerne M, Willery J (1997) A comparison between three methods for the determination of cation exchange capacity and exchangeable cations in soils. Agronomie 17:9–16
DaMatta FM (2004) Ecophysiological constraints on the production of shaded and unshaded coffee: a review. Field Crop Res 86:99–114. https://doi.org/10.1016/j.fcr.2003.09.001
de Souza HN, de Goede RGM, Brussaard L, Cardoso IM, Duarte EMG, Fernandes RBA, Gomes LC, Pulleman MM (2012) Protective shade, tree diversity and soil properties in coffee agroforestry systems in the Atlantic rainforest biome. Agric Ecosyst Environ 146:179–196. https://doi.org/10.1016/j.agee.2011.11.007
Defrenet E, Roupsard O, Van den Meersche K, Charbonnier F, Perez-Molina JP, Khac E, Prieto I, Stokes A, Roumet C, Rapidel B, Virginio EDV, Vargas VJ, Robelo D, Barquero A, Jourdan C (2016) Root biomass, turnover and net primary productivity of a coffee agroforestry system in Costa Rica: effects of soil depth, shade trees, distance to row and coffee age. Ann Bot 118:833–851. https://doi.org/10.1093/aob/mcw153
Descroix F, Snoeck J (2008) Environemental factors suitable for coffee cultivation. In: Coffee: growing, processing, sustainable production. Wiley-VCH, Weinheim, pp 164–177
Djigal D, Chabrier C, Duyck P-F, Achard R, Quénéhervé P, Tixier P (2012) Cover crops alter the soil nematode food web in banana agroecosystems. Soil Biol Biochem 48:142–150. https://doi.org/10.1016/j.soilbio.2012.01.026
Doran JW, Parkin TB (1994) Defining and assessing soil quality. In: Doran JW, Coleman DC, Bezdicek DF, Stewart BA (eds) Defining soil quality for a sustainable environment. Soil Science Society of America Special Publication, Madison, pp 3–21
Feng Y, Motta AC, Reeves DW, Burmester CH, van Santen E, Osborne JA (2003) Soil microbial communities under conventional-till and no-till continuous cotton systems. Soil Biol Biochem 35:1693–1703. https://doi.org/10.1016/j.soilbio.2003.08.016
Finney DM, Buyer JS, Kaye JP (2017) Living cover crops have immediate impacts on soil microbial community structure and function. J Soil Water Conserv 72:361–373. https://doi.org/10.2489/jswc.72.4.361
Fonte SJ, Barrios E, Six J (2010) Earthworms, soil fertility and aggregate-associated soil organic matter dynamics in the Quesungual agroforestry system. Geoderma 155:320–328. https://doi.org/10.1016/j.geoderma.2009.12.016
Frostegård A, Bååth E (1996) The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol Fertil Soils 22:59–65. https://doi.org/10.1007/bf00384433
Geisseler D, Scow KM (2014) Long-term effects of mineral fertilizers on soil microorganisms – a review. Soil Biol Biochem 75:54–63. https://doi.org/10.1016/j.soilbio.2014.03.023
Haggar J, Barrios M, Bolaños M, Merlo M, Moraga P, Munguia R, Ponce A, Romero S, Soto G, Staver C, de M. F. Virginio E (2011) Coffee agroecosystem performance under full sun, shade, conventional and organic management regimes in Central America. Agrofor Syst 82:285–301. https://doi.org/10.1007/s10457-011-9392-5
Hergoualc’h K, Blanchart E, Skiba U, Hénault C, Harmand J-M (2012) Changes in carbon stock and greenhouse gas balance in a coffee (Coffea arabica) monoculture versus an agroforestry system with Inga densiflora, in Costa Rica. Agric Ecosyst Environ 148:102–110. https://doi.org/10.1016/j.agee.2011.11.018
Lagerlöf J, Adolfsson L, Börjesson G, Ehlers K, Vinyoles GP, Sundh I (2014) Land-use intensification and agroforestry in the Kenyan highland: impacts on soil microbial community composition and functional capacity. Appl Soil Ecol 82:93–99. https://doi.org/10.1016/j.apsoil.2014.05.015
Mathew RP, Feng Y, Githinji L, Ankumah R, Balkcom KS (2012) Impact of no-tillage and conventional tillage systems on soil microbial communities. Appl Environ Soil Sci 2012:10. https://doi.org/10.1155/2012/548620
Mbuthia LW, Acosta-Martínez V, DeBruyn J, Schaeffer S, Tyler D, Odoi E, Mpheshea M, Walker F, Eash N (2015) Long term tillage, cover crop, and fertilization effects on microbial community structure, activity: implications for soil quality. Soil Biol Biochem 89:24–34. https://doi.org/10.1016/j.soilbio.2015.06.016
Mikha MM, Rice CW, Milliken GA (2005) Carbon and nitrogen mineralization as affected by drying and wetting cycles. Soil Biol Biochem 37:339–347. https://doi.org/10.1016/j.soilbio.2004.08.003
Mortimer PE, Gui H, Xu J, Zhang C, Barrios E, Hyde KD (2015) Alder trees enhance crop productivity and soil microbial biomass in tea plantations. Appl Soil Ecol 96:25–32. https://doi.org/10.1016/j.apsoil.2015.05.012
Muleta D, Assefa F, Nemomissa S, Granhall U (2008) Distribution of arbuscular mycorrhizal fungi spores in soils of smallholder agroforestry and monocultural coffee systems in southwestern Ethiopia. Biol Fertil Soils 44:653–659. https://doi.org/10.1007/s00374-007-0261-3
Nelson D, Sommers L (1982) Total carbon, organic carbon, and organic matter. In: Page AL (ed) Methods of soil analysis part 2 chemical and microbiological properties. American Society of Agronomy, Soil Science Society of America, Madison, pp 539–579
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O'Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2018) Vegan: community ecology package. R package version 2. 5–2. https://CRAN.R-project.org/package=vegan. Accessed 1 June 2018
Olsen S, Sommers L (1982) Phosphorous. In: Page AL (ed) Methods of soil analysis part 2 chemical and microbiological properties. American Society of Agronomy, Soil Science Society of America, Madison, pp 403–430
Padovan MP, Cortez VJ, Navarrete LF, Navarrete ED, Deffner AC, Centeno LG, Munguía R, Barrios M, Vílchez-Mendoza JS, Vega-Jarquín C, Costa AN, Brook RM, Rapidel B (2015) Root distribution and water use in coffee shaded with Tabebuia rosea Bertol. and Simarouba glauca DC. Compared to full sun coffee in sub-optimal environmental conditions. Agrofor Syst 89:857–868. https://doi.org/10.1007/s10457-015-9820-z
Payán F, Jones D, Beer J, Harmand J-M (2009) Soil characteristics below Erythrina poeppigiana in organic and conventional Costa Rican coffee plantations. Agrofor Syst 76:81–93. https://doi.org/10.1007/s10457-008-9201-y
Rigal C, Vaast P, Xu J (2018) Using farmers’ local knowledge of tree provision of ecosystem services to strengthen the emergence of coffee-agroforestry landscapes in Southwest China. PLoS One 13:e0204046. https://doi.org/10.1371/journal.pone.0204046
Romero-Alvarado Y, Soto-Pinto L, García-Barrios L, Barrera-Gaytán JF (2002) Coffee yields and soil nutrients under the shades of Inga sp. vs. multiple species in Chiapas, Mexico. Agrofor Syst 54:215–224. https://doi.org/10.1023/A:1016013730154
Sandhu HS, Wratten SD, Cullen R (2010) Organic agriculture and ecosystem services. Environ Sci Pol 13:1–7. https://doi.org/10.1016/j.envsci.2009.11.002
Scharroba A, Kramer S, Kandeler E, Ruess L (2016) Spatial and temporal variation of resource allocation in an arable soil drives community structure and biomass of nematodes and their role in the micro-food web. Pedobiologia 59:111–120. https://doi.org/10.1016/j.pedobi.2016.03.005
Siles P, Harmand J-M, Vaast P (2010) Effects of Inga densiflora on the microclimate of coffee (Coffea arabica L.) and overall biomass under optimal growing conditions in Costa Rica. Agrofor Syst 78:269–286. https://doi.org/10.1007/s10457-009-9241-y
Six J, Frey SD, Thiet RK, Batten KM (2006) Bacterial and fungal contributions to carbon sequestration in agroecosystems. Soil Sci Soc Am J 70:555–569. https://doi.org/10.2136/sssaj2004.0347
Snoeck J, Lambot C (2008) Fertilization. In: Coffee: growing, processing, sustainable production. Wiley-VCH, Weinheim, pp 246–269
Tabatabai M (1994) Soil enzymes. In: methods of soil analysis: part 2-microbiological and biochemical properties. Soil science Society of America. In: 775–833
Treseder KK (2008) Nitrogen additions and microbial biomass: a meta-analysis of ecosystem studies. Ecol Lett 11:1111–1120. https://doi.org/10.1111/j.1461-0248.2008.01230.x
Tully KL, Lawrence D (2011) Closing the loop: nutrient balances in organic and conventional coffee agroforests. J Sustain Agric 35:671–695. https://doi.org/10.1080/10440046.2011.586599
Tully KL, Lawrence D (2012) Canopy and leaf composition drive patterns of nutrient release from pruning residues in a coffee agroforest. Ecol Appl 22:1330–1344. https://doi.org/10.1890/10-2342.1
Tully KL, Lawrence D, Scanlon TM (2012) More trees less loss: nitrogen leaching losses decrease with increasing biomass in coffee agroforests. Agric Ecosyst Environ 161:137–144. https://doi.org/10.1016/j.agee.2012.08.002
Ugarte CM, Zaborski ER, Wander MM (2013) Nematode indicators as integrative measures of soil condition in organic cropping systems. Soil Biol Biochem 64:103–113. https://doi.org/10.1016/j.soilbio.2013.03.035
UNEP (2007) Global environment outlook : GEO 4 environment for development. United Nations Environment Programme, Valetta
Vallejo VE, Roldan F, Dick RP (2010) Soil enzymatic activities and microbial biomass in an integrated agroforestry chronosequence compared to monoculture and a native forest of Colombia. Biol Fertil Soils 46:577–587. https://doi.org/10.1007/s00374-010-0466-8
Van Bezooijen J (2006) Methods and techniques for nematology. Wageningen University, Wageningen
van Kanten R, Vaast P (2006) Transpiration of Arabica coffee and associated shade tree species in sub-optimal, low-altitude conditions of Costa Rica. Agrofor Syst 67:187–202. https://doi.org/10.1007/s10457-005-3744-y
van Noordwijk M, Brouwer G, Meijboom F, do Rosário G, Oliveira M, Bengough AG (2000) Trench profile techniques and core break methods. In: Root methods: a handbook. Springer Berlin Heidelberg, pp 211–233
Van Soest PJ, Robertson JB (1980) Systems of analysis for evaluating fibrous feeds. In: Pigden WJ, Balch CC, Graham M (eds) Standardization of analytical methodology for feeds. International Development Research Centre, Ottawa, pp 49–60
Willers C, van Rensburg PJJ, Claassens S (2015) Phospholipid fatty acid profiling of microbial communities-a review of interpretations and recent applications. J Appl Microbiol 119:1207–1218. https://doi.org/10.1111/jam.12902
Youkhana AH, Idol TW (2015) Leucaena-KX2 mulch additions increase growth, yield and soil C and N in a managed full-sun coffee system in Hawaii. Agrofor Syst 90:325–337. https://doi.org/10.1007/s10457-015-9857-z
Zelles L (1999) Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: a review. Biol Fertil Soils 29:111–129. https://doi.org/10.1007/s003740050533
Zhang Q, Liang GQ, Zhou W, Sun JW, Wang XB, He P (2016) Fatty-acid profiles and enzyme activities in soil particle-size fractions under long-term fertilization. Soil Sci Soc Am J 80:97–111. https://doi.org/10.2136/sssaj2015.07.0255
Zhao QY, Xiong W, Xing YZ, Sun Y, Lin XJ, Dong YP (2018) Long-term coffee monoculture alters soil chemical properties and microbial communities. Sci Rep 8:11. https://doi.org/10.1038/s41598-018-24537-2
Acknowledgements
The authors thank 岩庄香 for his invaluable help in the field, Nestlé for its support on soil chemical analysis and all the farmers who welcomed us in their farms for this experiment.
Funding
This research was funded through a PhD program by the Chinese Academy of Sciences and the Kunming Institute of Botany, with additional financial support from the CGIAR program on Forests, Trees and Agroforestry (FTA), and supported by Key Research Program of Frontier Sciences, CAS, Grant No. QYZDY-SSW-SMC014.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Remi Cardinael.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 100 kb)
Rights and permissions
About this article
Cite this article
Rigal, C., Xu, J. & Vaast, P. Young shade trees improve soil quality in intensively managed coffee systems recently converted to agroforestry in Yunnan Province, China. Plant Soil 453, 119–137 (2020). https://doi.org/10.1007/s11104-019-04004-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-04004-1