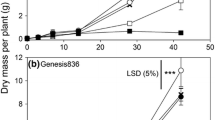
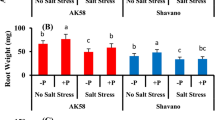
Abstract
Supplemental calcium (Ca2+) is used in hydroponic studies on salinity to lessen the potential for Ca2+ deficiency. However, the Ca2+ concentration and the sodium (Na+): Ca2+ ratio used vary considerably. The implications of using a wide range of Na+: Ca2+ ratios for studies of salinity tolerance in wheat are not known. Also, despite the risk of development of Ca2+ deficiency under salinity stress, there are few reliable reports on the critical level of Ca2+ which can be used to diagnose Ca2+ deficiency in wheat. Two experiments were conducted to examine Ca2+ requirements of wheat under saline and non-saline conditions and to derive a critical level for Ca2+. Four bread wheat genotypes (Triticum aestivum L.) and a durum wheat genotype [Triticum turgidum subsp. durum) (Desf.) Husn.] with known differences in salinity tolerance were grown at 100 mM NaCl for four weeks with varying levels of external Ca2+ which resulted in Na+:Ca2+ ratios of 30, 20, 15, 5 and 2. The critical Ca2+ concentration was defined in a second experiment by growing the same wheat genotypes at seven levels of Ca2+ (0.05, 0.1, 0.2, 0.5, 1, 2 and 10 mM) under non-saline conditions. When grown at 100 mM NaCl salinity tolerance was greatest when the Na+:Ca2+ ratio ranged from 5 to 15. Growing plants at lower or higher Na+:Ca2+ ratios induced nutrient imbalances and additional osmotic stress which reduced the growth of plants. Transient Ca2+ deficiency occurred at high Na+:Ca2+ ratios and low Mg2+ occurred at the lowest Na+:Ca2+ ratio. Adding NaCl raised the tissue Na+ concentration and reduced the Ca2+ concentration and the most appropriate Na+:Ca2+ ratio in the solution was that which resulted in tissue Ca2+ concentrations similar to those of non-salinised plants. The critical level of Ca2+ in the youngest fully emerged leaf blades was 15–23 mmol kg-1 DW (600–900 mg kg-1 DW).







Similar content being viewed by others
References
Adcock KG, Gartrell JW, Brennan RF (2001) Calcium deficiency of wheat grown in acidic sandy soil from Southwestern Australia. Journal of Plant Nutrition 24:1217–1227
Bergmann W (1992) Nutritional disorders of plants: Developments, visual and analytical diagnosis. Gustav Fisher, Jena, Stuttgart, New York
Borg H (1989) The relationships between the concentration of total soluble salts and osmotic potential in soil, ground and surface waters for several regions of Western Australia. Resource Management Technical Report No. 83. Department of Agriculture, Western Australia.
Brennan RF, Bolland MDA, Walton GH (2007) Comparing the calcium requirements of wheat and canola. Journal of Plant Nutrition 30:1167–1184
Colmer TD, Flowers TJ, Munns R (2006) Use of wild relatives to improve salt tolerance in wheat. Journal of Experimental Botany 57:1059–1078
Cramer GR (2002) Sodium-calcium interactions under salinity stress. In: Lauchli A, Luttge U (eds) Salinity: Environment-plants-molecules. Kluwer, Dordrecht, pp 205–227
Cramer GR, Läuchli A (1986) Ion activities in solution in relation to Na+–Ca2+ interactions at the plasmalemma. Journal of Experimental Botany 37:321–330
Cramer GR, Läuchli A, Polito VS (1985) Displacement of Ca2+ by Na+ from plasmalemma of root cells. A primary response to salt stress? Plant Physiology 79:207–211
Cramer GR, Epstein E, Läuchli A (1989) Na-Ca relations in barley seedlings: relationship to ion transport and growth. Plant Cell and Environment 12:551–558
Ehret DI, Redmann RE, Harvey BL, Cipywnyk A (1990) Salinity-induced calcium deficiencies in wheat and barley. Plant and Soil 128:143–151
Flowers TJ (2004) Improving crop salt tolerance. Journal of Experimental Botany 55:307–319
Forster BP, Phillips MS, Miller TE, Baird E, Powell W (1990) Chromosome location of genes controlling tolerance to salt (NaCl) and vigour in Hordeum vulgare and H. chilense. Heredity 65:99–107
Fowler DB, Hamm JW (1980) Crop response to saline soil conditions in the parkland area of Saskatchewan. Canadian Journal of Soil Science 60:439–449
Genc Y, McDonald GK, Tester M (2007) Re-assessment of tissue Na+ concentration as a criterion for salinity tolerance in bread wheat. Plant Cell and Environment 30:1486–1498
GENSTAT 8.2. (2005) Lawes Agricultural Trust (Rothamsted Experimental Station), United Kingdom.
Gorham J (1990) Salt tolerance in the Triticeae: K/Na discrimination in synthetic hexaploid wheats. Journal of Experimental Botany 41:623–627
Greenway H, Munns R (1980) Mechanisms of salt tolerance in nonhalophytes. Annual Review of Plant Physiology 31:149–190
Grieve CM, Francois LE (1992) The importance of seed size in wheat plant response to salinity. Plant and Soil 147:197–205
Grieve CM, Fujiyama H (1987) The response of two rice cultivars to external Na/Ca ratio. Plant and Soil 103:245–250
Houshmand S, Arzani A, Maibody SAM, Feizi M (2005) Evaluation of salt-tolerant genotypes of durum wheat derived from in vitro and field experiments. Field Crops Research 91:345–354
Husain S, von Caemmerer S, Munns R (2004) Control of salt transport from roots to shoots of wheat in saline soil. Functional Plant Biology 31:1115–1126
Jaradat AA, Shahid M, Al-Maskri AZ (2004) Genetic diversity in the batani barley landrace from Oman II. Response to salinity stress. Crop Science 44:997–1007
Jurinak JJ (1990) The chemistry of salt-affected soils and waters. In: Tanji KK (ed) Agriculture Salinity Assessment and Management. The American Society of Civil Engineers, New York, pp 42–63
Kinraide TB (1999) Interactions among Ca2+, Na+ and K+ in salinity tolerance: quantitative resolution of multiple toxic and ameliorative effects. Journal of Experimental Botany 50:1495–1505
LaHaye PA, Epstein E (1969) Salt tolerance by plants: enhancement with calcium. Science 166:395–396
Loneragan JF, Snowball K (1969) Calcium requirements of plants. Australian Journal of Agricultural Research 20:465–478
Munns R, Husain S, Rivelli AR, James RA, Condon AG, Lindsay MP, Lagudah ES, Schachtman DP, Hare RA (2002) Avenues for increasing salt tolerance of crops, and the role of physiologically based selection traits. Plant and Soil 247:93–105
Munns R, James RA (2003) Screening methods for salinity tolerance: a case study with tetraploid wheat. Plant and Soil 253:201–218
Munns R, Tester M (2008) Mechanisms of salt tolerance. Annual Review of Plant Biology 59:651–681
Naidu R, Rengasamy P, de Lacy NJ, Zarcinas BA (1995) Soil solution composition of some sodic soils. In: Naidu R, Sumner ME, Rengasamy P (eds) Australian sodic soils: distribution, properties and management. CSIRO Publishing, Melbourne, pp 155–161
Reid RJ, Smith FA (2000) The limits of sodium/calcium interaction in plant growth. Australian Journal of Plant Physiology 27:709–715
Reuter DJ, Robinson AD (1997) Plant Analysis. An Interpretation Manual. 2nd edn. CSIRO, Australia.
Rivelli AR, James RA, Munns R, Condon AG (2002) Effect of salinity on water relations and growth of wheat genotypes with contrasting sodium uptake. Functional Plant Biology 29:1065–1074
Salam A, Hollington PA, Gorham J, Wyn Jones RG, Gliddon C (1999) Physiological genetics of salt tolerance in wheat (Triticum aestivum L): Performance of wheat varieties, inbred lines and reciprocal F1 hybrids under saline conditions. Journal of Agronomy and Crop Science 183:145–156
Schachtman DP, Lagudah ES, Munns R (1992) The expression of salt tolerance from Triticum tauschii in hexaploid wheat. Theoretical and Applied Genetics 84:714–719
Tester M, Davenport R (2003) Na+ tolerance and Na+ transport in higher plants. Annals of Botany 91:503–527
Walia H, Wilson C, Wahid A, Condamine P, Cui X, Close TJ (2006) Expression analysis of barley (Hordeum vulgare L.) during salinity stress. Functional and Integrative Genomics 6:143–146
Weir RG (1983) Tissue analysis for pastures and field crops. NSW Department of Agriculture Advisory Note No.11/83.
White PJ, Broadley MR (2001) Chloride in soils and its uptake and movement within the plant: A review. Annals of Botany 88:967–988
White PJ, Broadley MR (2003) Calcium in plants. Annals of Botany 92:1–25
Zadoks JC, Chang TT, Konzak CF (1974) A decimal code for the growth stages of cereals. Weed Research 14:415–421
Zarcinas BA, Cartwright B, Spouncer LR (1987) Nitric acid digestion and multi-element analysis of plant material by inductively coupled plasma spectrometry. Communications in Soil Science and Plant Analysis 18:131–146
Acknowledgements
We thank Mr. Robin Hosking (the Australian Centre for Plant Functional Genomics) for his construction and creative modification of the supported hydroponic system and technical support throughout this project, Mrs. Teresa Fowles and Mr. Lyndon Palmer (The University of Adelaide) for their help with ICP-OES analysis, Mr. Stewart Coventry (The University of Adelaide) for his assistance with osmotic potential measurements, and Dr. Hugh Wallwork (South Australian Research and Development Corporation) for kindly supplying the seed for this study. We also thank Dr. Robert M Norton (University of Melbourne, Australia) and Dr. John Angus (the Commonwealth Scientific and Industrial Research Organisation, Canberra, Australia) for providing data on soil solution Ca2+ concentrations, and Dr. Rana Munns (Commonwealth Scientific and Industrial Research Organisation, Canberra, Australia), Dr. Christina Morris (the Australian Centre for Plant Functional Genomics), the editor and anonymous reviewers for their constructive comments on this manuscript. This work was supported by the Molecular Plant Breeding Cooperative Research Centre, the Australian Centre for Plant Functional Genomics, the Grains Research and Development Corporation, the Australian Research Council and The University of Adelaide.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Timothy J. Flowers.
Rights and permissions
About this article
Cite this article
Genc, Y., Tester, M. & McDonald, G.K. Calcium requirement of wheat in saline and non-saline conditions. Plant Soil 327, 331–345 (2010). https://doi.org/10.1007/s11104-009-0057-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-009-0057-3