Abstract
Key message
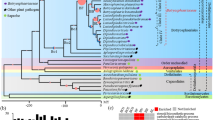
An exhaustive analysis of a group of closely related parasitic plants shows a predominantly gradual reduction in plastid genome composition and provides the most reduced plastomes in the genus Cuscuta.
Abstract
Parasitic plants have a diminished to completely absent reliance on photosynthesis, and are characterized by sweeping morphological, physiological, and genomic changes. The plastid genome (plastome) is highly conserved in autotrophic plants but is often reduced in parasites, and provides a useful system for documenting the genomic effects of a loss of photosynthesis. Previous studies have shown a substantial degree of heterogeneity in plastome length and composition across the species of the genus Cuscuta. Specifically, species in Cuscuta sect. Ceratophorae were suspected to exhibit even more dynamic plastome evolution than the rest of the genus. This complex of eight closely related species was exhaustively sampled here, and one accession per species was sequenced via a high-throughput approach. Complete plastid genomes were assembled and annotated for each of these species and were found to be 61–87 kbp in length, representing a 45–60% reduction relative to autotrophic Convolvulaceae. The most reduced plastomes on this spectrum have lost the bulk of their photosynthetic genes and are the first fully holoparasitic plastomes described for Cuscuta. The fine-scale nature of the system introduced here allowed us to phylogenetically triangulate the locations of gene loss and pseudogenization events precisely, and to construct a step-by-step model of plastome evolution in these plants. This model reveals an intense burst of gene loss along the branch leading to the most reduced plastomes, and a few idiosyncratic changes elsewhere, allowing us to conclude that the tempo of plastid evolution in sect. Ceratophorae is a blend of gradual and punctuated mode.





Similar content being viewed by others
Change history
24 September 2019
Regrettably, an in-text citation wasn’t listed in the reference section of the above mentioned publication. The citation reads Schneider et al. (2018) and the correct reference is published here.
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Barkman TJ, McNeal JR, Lim SH, Coat G, Croom HB, Young ND, Depamphilis CW (2007) Mitochondrial DNA suggests at least 11 origins of parasitism in angiosperms and reveals genomic chimerism in parasitic plants. BMC Evol Biol 7:248. https://doi.org/10.1186/1471-2148-7-248
Barrett CF, Davis JI (2012) The plastid genome of the mycoheterotrophic Corallorhiza striata (Orchidaceae) is in the relatively early stages of degradation. Am J Bot 99:1513–1523. https://doi.org/10.3732/ajb.1200256
Barrett CF, Wicke S, Sass C (2018) Dense infraspecific sampling reveals rapid and independent trajectories of plastome degradation in a heterotrophic orchid complex. New Phytol. https://doi.org/10.1111/nph.15072
Bellot S, Renner SS (2015) The plastomes of two species in the endoparasite genus pilostyles (Apodanthaceae) each retain just five or six possibly functional genes. Genome Biol Evol 8:189–201. https://doi.org/10.1093/gbe/evv251
Braukmann T, Kuzmina M, Stefanovic S (2013) Plastid genome evolution across the genus Cuscuta (Convolvulaceae): two clades within subgenus Grammica exhibit extensive gene loss. J Exp Bot 64:977–989. https://doi.org/10.1093/jxb/ers391
Braukmann TWA, Broe MB, Stefanovic S, Freudenstein JV (2017) On the brink: the highly reduced plastomes of nonphotosynthetic Ericaceae. New Phytol 216:254–266. https://doi.org/10.1111/nph.14681
Chang CC et al (2006) The chloroplast genome of Phalaenopsis aphrodite (Orchidaceae): comparative analysis of evolutionary rate with that of grasses and its phylogenetic implications. Mol Biol Evol 23:279–291. https://doi.org/10.1093/molbev/msj029
Colwell AE (1994) Genome evolution in a non-photosynthetic plant, Conopholis americana. Washington University, Louis
Costea M, Garcia Ruiz I, Stefanovic S (2011) Systematics of “horned” dodders: phylogenetic relationships, taxonomy, and two new species within the Cuscuta chapalana complex (Convolvulaceae). Botany 89:715–730
Costea M, Garcia MA, Stefanovic S (2015) A phylogenetically based infrageneric classification of the parasitic plant genus Cuscuta (dodders, Convolvulaceae). Syst Bot 40:269–285
Darling AE, Mau B, Perna NT (2010) progressiveMauve: multiple genome alignment with gene gain, loss and rearrangement. PLoS ONE 5:e11147. https://doi.org/10.1371/journal.pone.0011147
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772. https://doi.org/10.1038/nmeth.2109
Dawson JH, Musselman LJ, Wolswinkel P, Dorr I (1994) Biology and control of Cuscuta. Rev Weed Sci 6:265–317
Downie SR, Palmer JD (1992) Restriction site mapping of the chloroplast DNA inverted repeat—a molecular phylogeny of the Asteridae. Ann Mo Bot Gard 79:266–283. https://doi.org/10.2307/2399769
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Funk HT, Berg S, Krupinska K, Maier UG, Krause K (2007) Complete DNA sequences of the plastid genomes of two parasitic flowering plant species, Cuscuta reflexa and Cuscuta gronovii. BMC Plant Biol 7:45. https://doi.org/10.1186/1471-2229-7-45
Garcia MA, Costea M, Kuzmina M, Stefanovic S (2014) Phylogeny, character evolution, and biogeography of Cuscuta (dodders; Convolvulaceae) inferred from coding plastid and nuclear sequences. Am J Bot 101:670–690. https://doi.org/10.3732/ajb.1300449
Graham SW, Lam VK, Merckx VS (2017) Plastomes on the edge: the evolutionary breakdown of mycoheterotroph plastid genomes. New Phytol 214:48–55. https://doi.org/10.1111/nph.14398
Greiner S, Lehwark P, Bock R (2019) OrganellarGenomeDRAW (OGDRAW) version 1.3.1: expanded toolkit for the graphical visualization of organellar genomes. Nucleic Acids Res. https://doi.org/10.1093/nar/gkz238
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704
Heide-Jorgensen H (2008) Parasitic flowering plants. Brill Academic Publishers, Leiden
Hibberd JM, Bungard RA, Press MC, Jeschke WD, Scholes JD, Quick WP (1998) Localization of photosynthetic metabolism in the parasitic angiosperm Cuscuta reflexa. Planta 205:506–513. https://doi.org/10.1007/s004250050349
Joshi NA, Fass JN (2011) Sickle: a sliding-window, adaptive, quality-based trimming tool for FastQ files [Software]. https://github.com/najoshi/sickle
Kohzuma K, Dal Bosco C, Kanazawa A, Dhingra A, Nitschke W, Meurer J, Kramer DM (2012) Thioredoxin-insensitive plastid ATP synthase that performs moonlighting functions. Proc Natl Acad Sci USA 109:3293–3298. https://doi.org/10.1073/pnas.1115728109
Krause K (2011) Piecing together the puzzle of parasitic plant plastome evolution. Planta 234:647–656. https://doi.org/10.1007/s00425-011-1494-9
Krause K, Berg S, Krupinska K (2003) Plastid transcription in the holoparasitic plant genus Cuscuta: parallel loss of the rrn16 PEP-promoter and of the rpoA and rpoB genes coding for the plastid-encoded RNA polymerase. Planta 216:815–823. https://doi.org/10.1007/s00425-002-0933-z
Kuijt J (1969) The biology of parasitic flowering plants. University of California Press, Berkeley
Lowe TM, Chan PP (2016) tRNAscan-SE On-line: integrating search and context for analysis of transfer RNA genes. Nucleic Acids Res 44:W54–W57. https://doi.org/10.1093/nar/gkw413
Mayr E (1982) Speciation and macroevolution. Evolution 36:1119–1132. https://doi.org/10.2307/2408147
McNeal JR, Arumugunathan K, Kuehl JV, Boore JL, Depamphilis CW (2007a) Systematics and plastid genome evolution of the cryptically photosynthetic parasitic plant genus Cuscuta (Convolvulaceae). BMC Biol 5:55. https://doi.org/10.1186/1741-7007-5-55
McNeal JR, Kuehl JV, Boore JL, de Pamphilis CW (2007b) Complete plastid genome sequences suggest strong selection for retention of photosynthetic genes in the parasitic plant genus Cuscuta. BMC Plant Biol 7:57. https://doi.org/10.1186/1471-2229-7-57
McNeal JR, Kuehl JV, Boore JL, Leebens-Mack J, dePamphilis CW (2009) Parallel loss of plastid introns and their maturase in the genus Cuscuta. PLoS ONE 4:e5982. https://doi.org/10.1371/journal.pone.0005982
Molina J et al (2014) Possible loss of the chloroplast genome in the parasitic flowering plant Rafflesia lagascae (Rafflesiaceae). Mol Biol Evol 31:793–803. https://doi.org/10.1093/molbev/msu051
Murrell B, Moola S, Mabona A, Weighill T, Sheward D, Kosakovsky Pond SL, Scheffler K (2013) FUBAR: a fast, unconstrained bayesian approximation for inferring selection. Mol Biol Evol 30:1196–1205. https://doi.org/10.1093/molbev/mst030
Naumann J et al (2016) Detecting and characterizing the highly divergent plastid genome of the nonphotosynthetic parasitic plant Hydnora visseri (Hydnoraceae). Genome Biol Evol 8:345–363. https://doi.org/10.1093/gbe/evv256
Park I, Yang S, Kim WJ, Noh P, Lee HO, Moon BC (2018) The complete chloroplast genomes of six ipomoea species and indel marker development for the discrimination of authentic pharbitidis semen (seeds of I. nil or I. purpurea). Front Plant Sci 9:965. https://doi.org/10.3389/fpls.2018.00965
Peltier G, Aro EM, Shikanai T (2016) NDH-1 and NDH-2 plastoquinone reductases in oxygenic photosynthesis. Annu Rev Plant Biol 67:55–80. https://doi.org/10.1146/annurev-arplant-043014-114752
Pond SL, Frost SD, Muse SV (2005) HyPhy: hypothesis testing using phylogenies. Bioinformatics 21:676–679. https://doi.org/10.1093/bioinformatics/bti079
Schwender J, Goffman F, Ohlrogge JB, Shachar-Hill Y (2004) Rubisco without the Calvin cycle improves the carbon efficiency of developing green seeds. Nature 432:779–782. https://doi.org/10.1038/nature03145
Stefanovic S, Kuzmina M, Costea M (2007) Delimitation of major lineages within Cuscuta subgenus Grammica (Convolvulaceae) using plastid and nuclear DNA sequences. Am J Bot 94:568–589. https://doi.org/10.3732/ajb.94.4.568
van der Kooij TA, Krause K, Dorr I, Krupinska K (2000) Molecular, functional and ultrastructural characterisation of plastids from six species of the parasitic flowering plant genus Cuscuta. Planta 210:701–707. https://doi.org/10.1007/s004250050670
Westwood JH, Yoder JI, Timko MP, dePamphilis CW (2010) The evolution of parasitism in plants. Trends Plant Sci 15:227–235. https://doi.org/10.1016/j.tplants.2010.01.004
Wicke S, Schneeweiss GM, dePamphilis CW, Muller KF, Quandt D (2011) The evolution of the plastid chromosome in land plants: gene content, gene order, gene function. Plant Mol Biol 76:273–297. https://doi.org/10.1007/s11103-011-9762-4
Wicke S et al (2013) Mechanisms of functional and physical genome reduction in photosynthetic and nonphotosynthetic parasitic plants of the broomrape family. Plant Cell 25:3711–3725. https://doi.org/10.1105/tpc.113.113373
Wilgenbusch JC, Swofford D (2003) Inferring evolutionary trees with PAUP*, Chapter 6. Curr Protoc Bioinform. https://doi.org/10.1002/0471250953.bi0604s00
Wolfe KH, Morden CW, Ems SC, Palmer JD (1992) Rapid evolution of the plastid translational apparatus in a nonphotosynthetic plant—loss or accelerated sequence evolution of transfer-rna and ribosomal-protein genes. J Mol Evol 35:304–317. https://doi.org/10.1007/Bf00161168
Wu FH et al (2010) Complete chloroplast genome of Oncidium Gower Ramsey and evaluation of molecular markers for identification and breeding in Oncidiinae. BMC Plant Biol 10:68. https://doi.org/10.1186/1471-2229-10-68
Young ND, Steiner KE, dePamphilis CW (1999) The evolution of parasitism in Scrophulariaceae/Orobanchaceae: plastid gene sequences refute an evolutionary transition series. Ann Mo Bot Gard 86:876–893. https://doi.org/10.2307/2666173
Yuncker TG (1932) The genus Cuscuta. Mem Torrey Bot Club 18:113–331
Zoschke R, Nakamura M, Liere K, Sugiura M, Borner T, Schmitz-Linneweber C (2010) An organellar maturase associates with multiple group II introns. Proc Natl Acad Sci USA 107:3245–3250. https://doi.org/10.1073/pnas.0909400107
Acknowledgements
Special thanks to Eleazer Carranza, Mihai Costea, Ignacio Garcia Ruiz, Marc Johnson, and Tom Van Devender for tissue collections crucial to this project. We are also grateful to two anonymous reviewers for their valuable comments that have improved the quality of the manuscript. This work was supported by the Natural Sciences and Engineering Research Council of Canada (Grant No. 326439); the Canada Foundation for Innovation (Grant No. 12810); and Ontario Research Funds, all to S.S.
Author information
Authors and Affiliations
Contributions
AB and SS conceived the research conducted here. SS obtained plant tissue and extracted DNA. AB prepared samples for high-throughput sequencing and performed plastome assemblies and annotations. Analyses were conducted by both AB and SS. AB produced the first draft of the manuscript. Both authors have read and approved the final version of the manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11103_2019_884_MOESM2_ESM.eps
Supplementary material 2 (EPS 56 kb) Fig. S1 Whole-plastome alignment for the species assembled from Cuscuta section Ceratophorae
11103_2019_884_MOESM3_ESM.eps
Supplementary material 3 (EPS 287 kb) Fig. S2 dN vs. dS branch lengths for four additional genes from Cuscuta section Ceratophorae plastomes
Rights and permissions
About this article
Cite this article
Banerjee, A., Stefanović, S. Caught in action: fine-scale plastome evolution in the parasitic plants of Cuscuta section Ceratophorae (Convolvulaceae). Plant Mol Biol 100, 621–634 (2019). https://doi.org/10.1007/s11103-019-00884-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-019-00884-0