ABSTRACT
Purpose
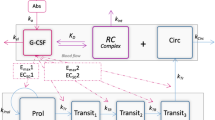
Granulocyte colony-stimulating factor (G-CSF) is often used in cancer patients receiving cytotoxic drugs to prevent or reduce high grade neutropenia. We propose a pharmacokinetic/pharmacodynamic model to describe myelotoxicity in both G-CSF treated and non-treated patients that shall increase our understanding of G-CSF effects.
Methods
The model was built from absolute neutrophil counts (ANC) obtained in 375 carboplatin-treated patients, 47 of whom received G-CSF. It includes some prior information on G-CSF taken from the literature. Simulations were performed to understand differences in G-CSF effects and explore the impact of G-CSF formulation.
Results
Our model well described the data in all patients. Model simulations showed that G-CSF was not as beneficial as expected in some patients. Furthermore, a longer and stronger effect was observed for the pegylated formulation in comparison with the daily standard formulation even if the latter was given for 11 consecutive days.
Conclusions
The proposed model allows a mechanistic interpretation of G-CSF effects on ANC and raises the question of a systematic beneficial effect of G-CSF treatment. Other studies are needed to confirm these findings and help identifying patients for whom G-CSF is beneficial.






Similar content being viewed by others
Abbreviations
- Abs 1 :
-
Absorption compartment for filgrastim/lenograstim
- Abs 2 :
-
Absorption compartment for pegfilgrastim
- ANC :
-
Absolute neutrophil count
- Base :
-
Baseline level of absolute neutrophil count
- C carbo :
-
Ultrafiltrable circulating (plasma) concentration of carboplatin
- C u :
-
Free circulating concentration controlling G-CSF effects on bone marrow, calculated as the sum of non-pegylated and pegylated G-CSF free circulating (serum) concentrations
- Circ :
-
Circulating mature neutrophil count (=ANC)
- E max 1 :
-
Maximal effect of non-pegylated or pegylated G-CSF on proliferation
- E max 2 :
-
Maximal effect of non-pegylated or pegylated G-CSF on maturation
- EC 50 1 :
-
Value of Cu eliciting 50% of the maximal effect on proliferation
- EC 50 2 :
-
Value of Cu eliciting 50% of the maximal effect on maturation
- F1(F2) :
-
Absolute bioavailability of filgrastim/lenograstim(pegfilgrastim) after subcutaneous administration (which, in the model, is taking into account via the apparent volume of distribution)
- G-CSF :
-
Granulocyte colony-stimulating factor
- k :
-
Transit rate constant between compartments of granulopoiesis (function of Cu) \( \left(k={k}_{tr}\times \left(1+\frac{E_{\max }2\times {C}_u}{E{C}_{50}2+{C}_u}\right)\right) \)
- k a 1(2) :
-
Absorption rate constant for filgrastim/lenograstim(pegfilgrastim)
- k circ :
-
Rate constant of elimination of neutrophils from the systemic blood circulation
- K D :
-
Dissociation constant of RC complex ( = k off /k on )
- k el 1 :
-
Rate constant for the linear, non-specific elimination of endogenous G-CSF and filgrastim/lenograstim
- kel2:
-
Rate constant for the linear, non-specific elimination of pegfilgrastim
- k GCSF :
-
Rate constant of endogenous G-CSF production
- k int :
-
Rate constant for non-pegylated or pegylated G-CSF elimination after binding to receptors and internalization
- k prol :
-
Proliferation rate constant
- k tr :
-
“Virtual” transit rate constant when Cu = 0 (cf. k)
- MTT :
-
Mean transit time for maturing precursors in bone marrow \( \left( MTT=4/{k}_{tr}\times \left(1+\frac{E_{\max }2\times {C}_u}{E{C}_{50}2+{C}_u}\right)\right) \)
- PK :
-
Pharmacokinetic(s)
- PK/PD :
-
Pharmacokinetic(s)/pharmacodynamic(s)
- Prol :
-
Stem cell and progenitor cell count (i.e. proliferative cells) in bone marrow
- R :
-
Concentration in G-CSF receptors present on circulating neutrophils
- RC :
-
Concentration in bound G-CSF complex (pegylated and non-pegylated G-CSF)
- R max :
-
Maximal amount of receptors involved in nonlinear, specific clearance of pegylated and on-pegylated G-CSF (=R + RC)
- RSE :
-
Relative standard error
- Slope :
-
Sensitivity to carboplatin myelotoxicity
- Transit 1,2,3 :
-
Maturating granulocyte precursor count in transit compartments 1, 2 and 3, respectively
- VD a 1(2) :
-
Apparent volume of distribution of G-CSF (pegylated G-CSF) after subcutaneous administration of filgrastim/lenograstim (pegfilgrastim) (VD1(2)/F1(2))
- ξ :
-
Proportionality constant for the amount of G-CSF receptors per cell
REFERENCES
Cameron D. Management of chemotherapy-associated febrile neutropenia. Br J Cancer. 2009;101 suppl 1:S18–22.
Segal BH, Freifeld AG, Baden LR, Brown AE, Casper C, Dubberke E, et al. Prevention and treatment of cancer-related infections. J Natl Compr Cancer Netw. 2008;6:122–74.
Kelly S, Wheatley D. Prevention of febrile neutropenia: use of granulocyte colony-stimulating factors. Br J Cancer. 2009;101 suppl 1:S6–S10.
Aapro M, Crawford J, Kamioner D. Prophylaxis of chemotherapy-induced febrile neutropenia with granulocyte colony-stimulating factors: where are we now? Support Care Cancer. 2010;18:529–41.
Cooper KL, Madan J, Whyte S, Stevenson MD, Akehurst RL. Granulocyte colony-stimulating factors for febrile neutropenia prophylaxis following chemotherapy: systematic review and meta-analysis. BMC Cancer. 2011;11:404.
Lord BI, Bronchud MH, Owens S, Chang J, Howell A, Souza L, et al. The kinetics of human granulopoiesis following treatment with granulocyte colony-stimulating factor in vivo. Proc Natl Acad Sci U S A. 1989;86:9499–503.
Price TH, Chatta GS, Dale DC. Effect of recombinant granulocyte colony-stimulating factor on neutrophil kinetics in normal young and elderly humans. Blood. 1996;88:335–40.
Aapro MS, Bohlius J, Cameron DA, Dal Lago L, Donnelly JP, Kearney N, et al. 2010 update of EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphoproliferative disorders and solid tumours. Eur J Cancer. 2011;47:8–32.
Crawford J, Caserta C, Roila F. Hematopoietic growth factors: ESMO recommendations for the applications. Ann Oncol. 2009;20 Suppl 4:162–5.
Tan H, Tomic K, Hurley D, Daniel G, Barron R, Malin J. Comparative effectiveness of colony-stimulating factors for febrile neutropenia: a retrospective study. Curr Med Res Opin. 2011;27:79–86.
Weycker D, Malin J, Barron R, Edelsberg J, Kartashov A, Oster G. Comparative effectiveness of filgrastim, pegfilgrastim, and sargramostim as prophylaxis against hospitalization for neutropenic complications in patients with cancer receiving chemotherapy. Am J Clin Oncol. 2012;35:267–74.
Yang BB, Lum PK, Hayashi MM, Roskos LK. Polyethylene glycol modification of filgrastim results in decreased renal clearance of the protein in rats. J Pharm Sci. 2004;93:1367–73.
Yang BB, Kido A. Pharmacokinetics and pharmacodynamics of pegfilgrastim. Clin Pharmacokinet. 2011;50:295–306.
Friberg LE, Karlsson MO. Mechanistic models for myelosuppression. Investig New Drugs. 2003;21:183–94.
Shochat E, Rom-Kedar V, Segel LA. G-CSF control of neutrophils dynamics in the blood. Bull Math Biol. 2007;69:2299–338.
Testart-Paillet D, Girard P, You B, Freyer G, Pobel C, Tranchand B. Contribution of modelling chemotherapy-induced hematological toxicity for clinical practice. Crit Rev Oncol Hematol. 2007;63:1–11.
Friberg LE, Henningsson A, Maas H, Nguyen L, Karlsson MO. Model of chemotherapy-induced myelosuppression with parameter consistency across drugs. J Clin Oncol. 2002;20:4713–21.
Quartino AL, Friberg LE, Karlsson MO. A simultaneous analysis of the time-course of leukocytes and neutrophils following docetaxel administration using a semi-mechanistic myelosuppression model. Investig New Drugs. 2012;30:833–45.
Sandstrom M, Lindman H, Nygren P, Johansson M, Bergh J, Karlsson MO. Population analysis of the pharmacokinetics and the haematological toxicity of the fluorouracil-epirubicin-cyclophosphamide regimen in breast cancer patients. Cancer Chemother Pharmacol. 2006;58:143–56.
Ramon-Lopez A, Nalda-Molina R, Valenzuela B, Perez-Ruixo JJ. Semi-mechanistic model for neutropenia after high dose of chemotherapy in breast cancer patients. Pharm Res. 2009;26:1952–62.
Zandvliet AS, Schellens JH, Copalu W, Beijnen JH, Huitema AD. Covariate-based dose individualization of the cytotoxic drug indisulam to reduce the risk of severe myelosuppression. J Pharmacokinet Pharmacodyn. 2009;36:39–62.
Sugiura M, Yamamoto K, Sawada Y, Iga T. Pharmacokinetic/pharmacodynamic analysis of neutrophil proliferation induced by recombinant granulocyte colony-stimulating factor (rhG-CSF): comparison between intravenous and subcutaneous administration. Biol Pharm Bull. 1997;20:684–9.
Hayashi N, Kinoshita H, Yukawa E, Higuchi S. Pharmacokinetic and pharmacodynamic analysis of subcutaneous recombinant human granulocyte colony stimulating factor (lenograstim) administration. J Clin Pharmacol. 1999;39:583–92.
Wang B, Ludden TM, Cheung EN, Schwab GG, Roskos LK. Population pharmacokinetic-pharmacodynamic modeling of filgrastim (r-metHuG-CSF) in healthy volunteers. J Pharmacokinet Pharmacodyn. 2001;28:321–42.
Engel C, Scholz M, Loeffler M. A computational model of human granulopoiesis to simulate the hematotoxic effects of multicycle polychemotherapy. Blood. 2004;104:2323–31.
Sugiura M, Ohno Y, Yamada Y, Suzuki H, Iga T. Pharmacokinetic/pharmacodynamic analysis of neutrophil proliferation induced by rhG-CSF in patients receiving antineoplastic drugs. Yakugaku Zasshi. 2004;124:599–604.
Scholz M, Engel C, Loeffler M. Modelling human granulopoiesis under poly-chemotherapy with G-CSF support. J Math Biol. 2005;50:397–439.
Vainstein V, Ginosar Y, Shoham M, Ranmar DO, Ianovski A, Agur Z. The complex effect of granulocyte colony-stimulating factor on human granulopoiesis analyzed by a new physiologically-based mathematical model. J Theor Biol. 2005;234:311–27.
Roskos LK, Lum P, Lockbaum P, Schwab G, Yang BB. Pharmacokinetic/pharmacodynamic modeling of pegfilgrastim in healthy subjects. J Clin Pharmacol. 2006;46:747–57.
Shochat E, Rom-Kedar V. Novel strategies for granulocyte colony-stimulating factor treatment of severe prolonged neutropenia suggested by mathematical modeling. Clin Cancer Res. 2008;14:6354–63.
Foley C, Mackey MC. Mathematical model for G-CSF administration after chemotherapy. J Theor Biol. 2009;257:27–44.
Krzyzanski W, Wiczling P, Lowe P, Pigeolet E, Fink M, Berghout A, et al. Population modeling of filgrastim PK-PD in healthy adults following intravenous and subcutaneous administrations. J Clin Pharmacol. 2010;50:101S–12S.
Scholz M, Schirm S, Wetzler M, Engel C, Loeffler M. Pharmacokinetic and -dynamic modelling of G-CSF derivatives in humans. Theor Biol Med Model. 2012;9:32.
Schmitt A, Gladieff L, Laffont CM, Evrard A, Boyer JC, Lansiaux A, et al. Factors for hematopoietic toxicity of carboplatin: refining the targeting of carboplatin systemic exposure. J Clin Oncol. 2010;28:4568–74.
Schmitt A, Gladieff L, Lansiaux A, Bobin-Dubigeon C, Etienne-Grimaldi MC, Boisdron-Celle M, et al. A universal formula based on cystatin C to perform individual dosing of carboplatin in normal weight, underweight, and obese patients. Clin Cancer Res. 2009;15:3633–9.
Molineux G. The design and development of pegfilgrastim (PEG-rmetHuG-CSF, Neulasta). Curr Pharm Des. 2004;10:1235–44.
Aapro MS, Cameron DA, Pettengell R, Bohlius J, Crawford J, Ellis M, et al. EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphomas and solid tumours. Eur J Cancer. 2006;42:2433–53.
Brendel K, Comets E, Laffont C, Laveille C, Mentre F. Metrics for external model evaluation with an application to the population pharmacokinetics of gliclazide. Pharm Res. 2006;23:2036–49.
Layton JE, Hockman H, Sheridan WP, Morstyn G. Evidence for a novel in vivo control mechanism of granulopoiesis: mature cell-related control of a regulatory growth factor. Blood. 1989;74:1303–7.
Takatani H, Soda H, Fukuda M, Watanabe M, Kinoshita A, Nakamura T, et al. Levels of recombinant human granulocyte colony-stimulating factor in serum are inversely correlated with circulating neutrophil counts. Antimicrob Agents Chemother. 1996;40:988–91.
Quartino LQ, Karlsson MO, Lindman H, and Friberg LE. An integrated G-CSF-myelosuppression model characterizing the target mediated disposition of endogenous G-CSF in breast cancer patients following chemotherapy. PAGE 20 (Athens) abstr 2255, 2011.
Avalos BR, Gasson JC, Hedvat C, Quan SG, Baldwin GC, Weisbart RH, et al. Human granulocyte colony-stimulating factor: biologic activities and receptor characterization on hematopoietic cells and small cell lung cancer cell lines. Blood. 1990;75:851–7.
Hanazono Y, Hosoi T, Kuwaki T, Matsuki S, Miyazono K, Miyagawa K, et al. Structural analysis of the receptors for granulocyte colony-stimulating factor on neutrophils. Exp Hematol. 1990;18:1097–103.
Avalos BR. Molecular analysis of the granulocyte colony-stimulating factor receptor. Blood. 1996;88:761–77.
Yang BB, Kido A, Salfi M, Swan S, Sullivan JT. Pharmacokinetics and pharmacodynamics of pegfilgrastim in subjects with various degrees of renal function. J Clin Pharmacol. 2008;48:1025–31.
Todd RC, Lippard SJ. Inhibition of transcription by platinum antitumor compounds. Metallomics. 2009;1:280–91.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pastor, M.L., Laffont, C.M., Gladieff, L. et al. Model-Based Approach to Describe G-CSF Effects in Carboplatin-Treated Cancer Patients. Pharm Res 30, 2795–2807 (2013). https://doi.org/10.1007/s11095-013-1099-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-013-1099-z