Purpose
The purpose of the present study was to explore the involvement of indoxyl sulfate (IS) in nephrotoxicity and central nervous system (CNS) toxicity in cisplatin (CDDP)-treated rats.
Materials and Methods
Renal function was evaluated by serum creatinine and BUN levels. The IS levels in the serum, brain and kidney was monitored by high-performance liquid chromatography method. Body weight and rectal temperature were monitored. Real-time PCR analysis was performed to examine rPer2 mRNA expression.
Results
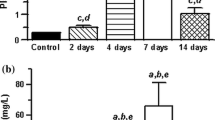
Renal function deteriorated in a time-dependent manner after administration of CDDP. The concentration of IS in the serum, brain and kidney markedly increased 24–84 h after commencement of CDDP treatment. The observed increase in the levels of serum creatinine, BUN and IS was suppressed by concomitant administration of AST-120. Rectal temperature was significantly lowered 72–92 h after CDDP-treatment, which was partially restored by coadministration of AST-120. Moreover, the amplitude of rectal temperature rhythms was disrupted by treatment with CDDP. Circadian rhythm of rPer2 mRNA expression, a clock gene, in suprachiasmatic nucleus (SCN) and kidney was disturbed in CDDP-treated rats.
Conclusions
An increase in the IS level and the associated disturbance to the circadian rhythm are involved in the renal and CNS toxicities in CDDP-treatment.





Similar content being viewed by others
Abbreviations
- IS:
-
indoxyl sulfate
- ARF:
-
acute renal failure
- CRF:
-
chronic renal failure
- SCN:
-
suprachiasmatic nucleus
- CNS:
-
central nervous system
- ZT:
-
zeitgeber time
- Per2 :
-
period 2
- SCr:
-
serum creatinine
References
F. Ries and J. Klastersky. Nephrotoxicity induced by cancer chemotherapy with special emphasis on cisplatin toxicity. Am. J. Kidney Dis. 8:368–379 (1986).
J. Deng, Y. Kohda, H. Chiao, Y. Wang, X. Hu, S. M. Hewitt, T. Miyaji, P. McLeroy, B. Nibhanupudy, S. Li, and R. A. Star. Interleukin-10 inhibits ischemic and cisplatin-induced acute renal injury. Kidney Int. 60:2118–2128 (2001).
G. Ramesh, and W. B. Reeves. TNF-alpha mediates chemokine and cytokine expression and renal injury in cisplatin nephrotoxicity. J. Clin. Invest. 110:835–842 (2002).
W. Lieberthal, V. Triaca, and J. Levine. Mechanisms of death induced by cisplatin in proximal tubular epithelial cells: apoptosis vs. necrosis. Adv. Physiol. Educ. 270:700–708 (1996).
T. Niwa, and M. Ise. Indoxyl sulfate, a circulating uremic toxin, stimulates the progression of glomerular sclerosis. J. Lab. Clin. Med. 124:96–104 (1994).
D. H. Bueschkens, and M. E. Stiles. Escherichia coli variants for gas and indole production at elevated incubation temperatures. Appl. Environ. Microbiol. 48:601–605 (1984).
E. Banoglu, G. G. Jha, and R. S. King. Hepatic microsomal metabolism of indole to indoxyl, a precursor of indoxyl sulfate. Eur. J. Drug Metab. Pharmacokinet. 26:235–240 (2001).
T. Miyazaki, M. Ise, H. Seo, and T., Niwa. Indoxyl sulfate increases the gene expression of TGF-beta 1, TIMP-1 and pro-α1(É) collagen in uremic rat kidneys. Kidney Inter., Suppl. 62:S15–S22 (1997).
T. Niwa, T. Nomura, S. Sugiyama, T. Miyazaki, S. Tsukushi, and S. Tsutsui. The protein metabolite hypothesis, a model for the progression of renal failure: an oral adsorbent lowers indoxyl sulfate levels in undialyzed uremic patients. Kidney Inter., Suppl. 62:S23–S28 (1997).
T. Miyazaki, I. Aoyama, M. Ise, H. Seo, and T. Niwa. An oral sorbent reduces overload of indoxyl sulphate and gene expression of TGF-β1 in uremic rat kidneys. Nephrol. Dial. Transplant. 15:1773–1781 (2000).
M. Motojima, A. Hosokawa, H. Yamato, T. Muraki, and T. Yoshioka. Uremic toxins of organic anion up-regulate PAI-1 expression by induction of NF-κB and free radical in proximal tubular cells. Kidney Int. 63:1671–1680 (2003).
A. van Coevorden, J. Mockel, E. Laurent, M. Kerkhofs, M. L’Hermite-Baleriaux, C. Decoster, P. Neve, and E. van Cauter. Neuroendocrine rhythms and sleep in aging men. Am. J. Physiol. 260:E651–E661 (1991).
D. Gupta, A. N. Aggarwal, S. Chaganti, and S. K. Jindal. Reducing the number of daily measurements results in poor estimation of diurnal variation of peak expiratory flow in healthy individuals. J. Postgrad. Med. 46:262–264 (2000).
A. M. Richards, M. G. Nicholls, E. A. Espiner, H. Ikram, M. Cullens, and D. Hinton. Diurnal patterns of blood pressure, heart rate and vasoactive hormones in normal man. Clin. Exp. Hypertens., Part A Theory Pract. 8:153–166 (1986).
W. E. Scales, A. J. Vander, M. B. Brown, and M. J. Kluger. Human circadian rhythms in temperature, trace metal, and blood variables. J. Appl. Physiol. 65:1840–1846 (1988).
M. H. Hastings. Central clocking. Trends Neurosci. 20:459–464 (1997).
H. Tei, H. Okamura, Y. Shigeyoshi, C. Fukuhara, R. Ozawa, M. Hirose, and Y. Sakaki. Circadian oscillation of a mammalian homologue of the Drosophila period gene. Nature 389:512–516 (1997).
K. Kume, M. J. Zylka, S. Sriram, L. P. Shearman, D. R. Weaver, X. Jin, E. S. Maywood, M. H. Hastings, and S. M. Reppert. mCRY1 and mCRY2 are essential component of negative limb of the circadian clock feedback loop. Cell 98:193–205 (1999).
S. Yamazaki, R. Numano, M. Abe, A. Hida, R. Takahashi, M. Ueda, G. D. Block, Y. Sakaki, M. Menaker, and H. Tei. Resetting central and peripheral circadian oscillators in transgenic rats. Science 288:682–685 (2000).
A. B. Reddy, M. D. Field, E. S. Maywood, and M. H. Hastings. Differential resynchronization of circadian clock gene expression within the suprachiasmatic nuclei of mice subjected to experimental jet lag. J. Neurosci. 22:7326–7330 (2002).
A. C. Schoots, P. M. De Vries, R. Thiemann, W. A. Hazejager, S. L. Visser, and P. L. Oe. Biochemical and neurophysiological parameters in hemodialyzed patients with chronic renal failure. Clin. Chim. Acta 185:91–107 (1989).
C. L. Robert, P. S. Richard, and C. Harold. The antitumor agent cis-Pt (NH3)2Cl2: distribution studies and dose calculation for 192mPt and 195mPt. J. Nucl. Med. 14:191–195 (1972).
I. J. Berman, and M. P. Mann. Seizures and transient cortical blindness associated with cis-platinum (II) diamminedichloride (PPD) therapy in a 30-year-old man. Cancer 45:764–766 (1980).
C. Verschraegen, C. A. Conrad, and W. K. Hong. Subacute encephalopathic toxicity of cisplatin. Lung Cancer 13:305–309 (1995).
T. Deguchi, M. Nakamura, Y. Tsutsumi, A. Suenaga, and M. Otagiri. Pharmacokinetics and tissue distribution of uraemic indoxyl sulphate in rats. Biopharm. Drug Dispos. 24:345–355 (2003).
T. Deguchi, S. Ohtsuki, M. Otagiri, H. Takanaga, H. Asaba, S. Mori, and T. Terasaki. Major role of organic anion transporter 3 in the transport of indoxyl sulphate in the kidney. Kidney Int. 61:1760–1768 (2002).
I. Aoyama, A. Enomoto, and T. Niwa. Effects of oral adsorbent on gene expression profile in uremic rat kidney: cDNA array analysis. Am. J. Kidney Dis. 41:S8–S14 (2003).
M. Motojima, A. Hosokawa, H. Yamato, T. Muraki, and T. Yoshioka. Uraemic toxins induce proximal tubular injury via organic anion transporter 1-mediated uptake. Br. J. Pharmacol. 135:555–563 (2002).
J. A. Boulant. Hypothalamic mechanisms in thermoregulation. Fed. Proc. 40:2843–2850 (1981).
T. Sato, S. Miyazaki, and S. Mohri. Effects of an oral adsorbent on cisplatin-induced nephropathy in rats. Nippon Jinzo Gakkai Shi 38:290–295 (1996).
S. Ohtsuki, H. Asaba, H. Takanaga, T. Deguchi, K. Hosoya, M. Otagiri, and T. Terasaki. Role of blood-brain barrier organic anion teansporter 3 (OAT3) in the efflux of indoxyl sulfate, a uremic toxin: its involvement in neurotransmitter metabolite clearance from the brain. J. Neurochem. 83:57–66 (2002).
M. Hohenegger, H. Echsel, M. Vermes, and H. Raneburger. Influence of some uremic toxins on oxygen consumption of rats in vivo and in vitro. Adv. Exp. Med. Biol. 212:99–104 (1987).
D. I. Sessler. Mild perioperative hypothermia. N. Engl. J. Med. 336:1730–1737 (1997).
R. W. Powell, D. L. Dyess, J. N. Collins, W. S. Roberts, E. J. Tacchi, A. N. Swafford Jr, J. J. Ferrara, and J. L. Ardell. Regional blood flow response to hypothermia in premature, newborn, and neonatal piglets. J. Pediatr. Surg. 34:193–198 (1999).
N. F. Ruby, J. Dark, D. E. Burns, H. C. Heller, and I. Zucker. The suprachiasmatic nucleus is essential for circadian body temperature rhythms in hibernating ground squirrels. J. Neurosci. 22:357–364 (2002).
C. K. Song, and T. J. Bartness. CNS sympathetic outflow neurons to white fat that express MEL receptors may mediate seasonal adiposity. Am. J. Physiol., Regul. Integr. Comp. Physiol. 281:R666–R672 (2001).
R. M. Buijs, S. J. Chun, A. Niijima, H. J. Romijn, and K. Nagai. Parasympathetic and sympathetic control of the pancreas: a role for the suprachiasmatic nucleus and other hypothalamic centers that are involved in the regulation of food intake. J. Comp. Neurol. 431:405–423 (2001).
C. Kopp, U. Albrecht, B. Zheng, and I. Tobler. Homeostatic sleep regulation is preserved in mPer1 and mPer2 mutant mice. Eur. J. Neurosci. 16:1099–1106 (2002).
K. L. Toh, C. R. Jones, Y. He, E. J. Eide, W. A. Hinz, D. M. Virshup, L. J. Ptacek, and Y. H. Fu. An hPer2 phosphorylation site mutation in familial advanced sleep phase syndrome. Science 291:1040–1043 (2001).
K. Ohishi, K. Sakamoto, T. Okada, T. Nagase, and N. Ishida. Humoral signals mediate the circadian expression of rat period homologue (rPer2) mRNA in peripheral tissues. Neurosci. Lett. 256:117–119 (1998).
F. Damiola, N. Le Minh, N. Preitner, B. Kornmann, F. Fleury-Olela, and U. Schibler. Restricted feeding uncouples circadian oscillators in peripheral tissues from the central pacemaker in the suprachiasmatic nucleus. Genes Dev. 14:2950–2961 (2000).
Acknowledgments
This work was supported, in part, by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, and Culture of Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
K.I. and H.W. contributed equally to this work.
Rights and permissions
About this article
Cite this article
Iwata, K., Watanabe, H., Morisaki, T. et al. Involvement of Indoxyl Sulfate in Renal and Central Nervous System Toxicities During Cisplatin-induced Acute Renal Failure. Pharm Res 24, 662–671 (2007). https://doi.org/10.1007/s11095-006-9183-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-006-9183-2