Abstract
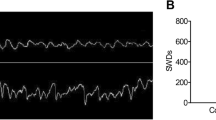
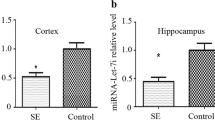
Epilepsy is characterized by recurrent unprovoked seizures and some seizures can cause neuronal apoptosis, which is possible to make contributions to the epilepsy phenotype, impairments in cognitive function or even epileptogenesis. Moreover, many studies have indicated that microRNA-34a (miRNA-34a) is involved in apoptosis through regulating Notch signaling. However, whether miRNA-34a participates in neuronal apoptosis after seizures remain unclear. Therefore, we aimed to explore the expression of miRNA-34a and its effects on the epileptiform discharge in spontaneous recurrent epileptiform discharges (SREDs) rat hippocampal neuronal pattern. Mg2+-free medium was used to induce SREDs, quantitative reverse-transcription polymerase chain reaction was used to detect the expression of miRNA-34a, western blot was used to determine the expression of Notch pathway and apoptosis-related proteins, and whole cell current clamp recordings was used to observe the alteration of epileptiform discharge. We found obvious apoptosis, increased expression of miRNA-34a and decreased expression of Notch signaling in Mg2+-free-treated neurons. Treatment with miRNA-34a inhibitor decreased the frequency of action potentials, activated Notch signaling and prevented neuronal apoptosis in Mg2+-free-treated neurons. However, treatment with miRNA-34a mimics increased the frequency of action potentials, down-regulated Notch signaling and promoted neuronal apoptosis in Mg2+-free-treated neurons. Furthermore, γ-secretase inhibitor N-[N-(3,5-di-uorophenacetyl)-1-alanyl]-S-phenylglycine t-butylester (DAPT), an inhibitor of Notch signaling, could weaken anti-apoptosis effect of miRNA-34a inhibitor. These results suggest that inhibition of miRNA-34a could suppress epileptiform discharges through regulating Notch signaling and apoptosis in the rat hippocampal neuronal model of SREDs.





Similar content being viewed by others
Abbreviations
- SREDs:
-
Spontaneous recurrent epileptiform discharges
- qRT-PCR:
-
Quantitative reverse-transcription polymerase chain reaction
- MAP2:
-
Protein-2 associated with anti-microtubule
- pBRS:
-
Physiological basal recording solution
- AP:
-
Action potentials
- Bax:
-
Bcl-2-associated X protein
- Bcl-2:
-
B-cell lymphoma-2
- HRP:
-
Horseradish peroxidase
- NIC:
-
Notch intracellular domain
References
Egbenya DL, Hussain S, Lai YC, Xia J, Anderson AE, Davanger S (2018) Changes in synaptic AMPA receptor concentration and composition in chronic temporal lobe epilepsy. Mol Cell Neurosci 92:93–103
Kwan P, Arzimanoglou A, Berg AT, Brodie MJ, Allen HW, Mathern G, Moshé SL, Perucca E, Wiebe S, French J (2010) Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE Commission on therapeutic strategies. Epilepsia 51:1069–1077
Ma Y (2018) The challenge of microRNA as a biomarker of epilepsy. Curr Neuropharmacol 16:37–42
Reschke CR, Henshall DC (2015) microRNA and epilepsy. Adv Exp Med Biol 888:41–70
Henshall DC (2014) MicroRNA and epilepsy: profiling, functions and potential clinical applications. Curr Opin Neurol 27:199–205
Chen F, Hu SJ (2012) Effect of microRNA-34a in cell cycle, differentiation, and apoptosis: a review. J Biochem Mol Toxicol 26:79–86
Lacombe J, Zenhausern F (2017) Emergence of miR-34a in radiation therapy. Crit Rev Oncol Hematol 109:69–78
Hu K, Xie YY, Zhang C, Ouyang DS, Long HY, Sun DN, Long LL, Feng L, Li Y, Xiao B (2012) MicroRNA expression profile of the hippocampus in a rat model of temporal lobe epilepsy and miR-34a-targeted neuroprotection against hippocampal neurone cell apoptosis post-status epilepticus. BMC Neurosci 13:115
Henshall DC, Simon RP (2005) Epilepsy and apoptosis pathways. J Cereb Blood Flow Metab 25:1557–1572
Deshpande LS, Nagarkatti N, Ziobro JM, Sombati S, DeLorenzo RJ (2008) Carisbamate prevents the development and expression of spontaneous recurrent epileptiform discharges and is neuroprotective in cultured hippocampal neurons. Epilepsia 49:1795–1802
Sombati S, Delorenzo RJ (1995) Recurrent spontaneous seizure activity in hippocampal neuronal networks in culture. J Neurophysiol 73:1706–1711
Li XY, Wen JY, Jia CC, Wang TT, Li X, Dong M, Lin QU, Chen ZH, Ma XK, Wei LI, Lin ZX, Ruan DY, Chen J, Wu DH, Liu W, Tai Y, Xiong ZY, Wu XY, Zhang QI (2015) MicroRNA-34a-5p enhances sensitivity to chemotherapy by targeting AXL in hepatocellular carcinoma MHCC-97L cells. Oncol Lett 10:2691–2698
Guan J, Wei X, Qu S, Lv T, Fu Q, Yuan Y (2017) Osthole prevents cerebral ischemia-reperfusion injury via the Notch signaling pathway. Biochem Cell Biol 95:459–467
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108
Henshall DC (2007) Apoptosis signalling pathways in seizure-induced neuronal death and epilepsy. Biochem Soc Trans 35:421–423
Pollard H, Charriaut-Marlangue C, Cantagrel S, Represa A, Robain O, Moreau J, Ben-Ari Y (1994) Kainate-induced apoptotic cell death in hippocampal neurons. Neuroscience 63:7–18
Guo XQ, Cao YL, Hao F, Yan ZR, Wang ML, Liu XW (2017) Tangeretin alters neuronal apoptosis and ameliorates the severity of seizures in experimental epilepsy-induced rats by modulating apoptotic protein expressions, regulating matrix metalloproteinases, and activating the PI3K/Akt cell survival pathway. Adv Med Sci 62:246–253
Li Q, Li QQ, Jia JN, Cao S, Wang ZB, Wang X, Luo C, Zhou HH, Liu ZQ, Mao XY (2018) Sodium valproate ameliorates neuronal apoptosis in a kainic acid model of epilepsy via enhancing PKC-dependent GABAAR γ2 serine 327 phosphorylation. Neurochem Res 43:2343–2352
Zhang B, Zhang JW, Wang WP, Dong RF, Tian S, Zhang C (2017) Effect of lamotrigine on epilepsy-induced cognitive impairment and hippocampal neuronal apoptosis in pentylenetetrazole-kindled animal model. Synapse 71(2):e21945
Zeng C, Xing R, Liu J, Xing F (2016) Role of CSL-dependent and independent Notch signaling pathways in cell apoptosis. Apoptosis 21:1–12
Miele L, Osborne B (1999) Arbiter of differentiation and death: notch signaling meets apoptosis. J Cell Physiol 181:393–409
Chen G, Zhang Z, Cheng Y, Xiao W, Qiu Y, Yu M, Sun L, Wang W, Du G, Gu Y, Peng K, Xu C, Yang H (2014) The canonical Notch signaling was involved in the regulation of intestinal epithelial cells apoptosis after intestinal ischemia/reperfusion injury. Int J Mol Sci 15:7883–7896
Ohtsuka T, Ishibashi M, Gradwohl G, Nakanishi S, Guillemot F, Kageyama R (1999) Hes1 and Hes5 as notch effectors in mammalian neuronal differentiation. EMBO J 18:2196–2207
Kageyama R, Ohtsuka T (1999) The Notch-Hes pathway in mammalian neural development. Cell Res 9:179–188
Li Z, Wang J, Zhao C, Ren K, Xia Z, Yu H, Jiang K (2016) Acute blockage of notch signaling by DAPT induces neuroprotection and neurogenesis in the neonatal rat brain after stroke. Transl Stroke Res 7:132–140
Sha L, Wu X, Yao Y, Wen B, Feng J, Sha Z, Wang X, Xing X, Dou W, Jin L, Li W, Wang N, Shen Y, Wang J, Wu L, Xu Q (2014) Notch signaling activation promotes seizure activity in temporal lobe epilepsy. Mol Neurobiol 49:633–644
Sibbe M, Häussler U, Dieni S, Althof D, Haas CA, Frotscher M (2012) Experimental epilepsy affects Notch1 signalling and the stem cell pool in the dentate gyrus. Eur J Neurosci 36:3643–3652
Karimzadeh F, Modarres MSM, Alipour F, Hosseini RH, Kovac S, Gorji A (2017) Developmental changes in Notch1 and NLE1 expression in a genetic model of absence epilepsy. Brain Struct Funct 222:2773–2785
Yan D, Hao C, Xiao-Feng L, Yu-Chen L, Yu-Bin F, Lei Z (2018) Molecular mechanism of Notch signaling with special emphasis on microRNAs: implications for glioma. J Cell Physiol 234:158–170
Li Y, Guessous F, Zhang Y, Dipierro C, Kefas B, Johnson E, Marcinkiewicz L, Jiang J, Yang Y, Schmittgen TD, Lopes B, Schiff D, Purow B, Abounader R (2009) MicroRNA-34a inhibits glioblastoma growth by targeting multiple oncogenes. Cancer Res 69:7569–7576
Li J, Gong J, Li X, Shen L, Xie Y, Zhang R (2019) MicroRNA-34a promotes CMECs apoptosis and upregulate inflammatory cytokines, thus worsening CMECs damage and inhibiting angiogenesis by negatively targeting the Notch signaling pathway. J Cell Biochem 120(2):1598–1609
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical Approval
Every animal use process was supported by the Animal Care and Use Committee of Shandong University and was in severe accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, J., Zheng, Y., Cheng, X. et al. Inhibition of microRNA-34a Suppresses Epileptiform Discharges Through Regulating Notch Signaling and Apoptosis in Cultured Hippocampal Neurons. Neurochem Res 44, 1252–1261 (2019). https://doi.org/10.1007/s11064-019-02772-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-019-02772-x