Abstract
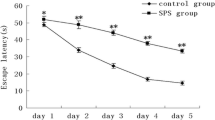
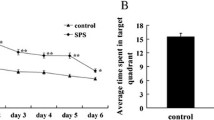
The dorsal raphe nucleus (DRN) has been suggested playing an important role in the pathophysiology of post-traumatic stress disorder (PTSD), however the underlying cellular mechanisms are not fully understood. The endoplasmic reticulum (ER) is a critical organelle for synthesis of membrane and secretory proteins, and perturbations in ER lead to the unfolded protein response (UPR). In the present experiment, we hypothesized UPR may be associated with the PTSD, and there is an induction of UPR in the DRN neurons of the PTSD-like rats. We first observed the morphological changes of ER in the DRN neurons of the rats exposed to single-prolonged stress (SPS), a model of PTSD, and then we also detected the expression of ER chaperones glucose regulated protein 78 (GRP78) and glucose regulated protein (GRP94) which are two key sensors and mediators of the UPR and are considered an ER stress-specific inducible proteins using methods of western blot and immunohistochemical analysis. Our results demonstrated there were abnormal expansion of ER and up-regulation expression of GRP78 and GRP94 after SPS, which indicated that the UPR was triggered in the DRN neurons of the PTSD-like rats. These results are consistent with our speculation that UPR may be associated with the PTSD, and suggest us the UPR may be a new critical cellular mechanisms of PTSD.



Similar content being viewed by others
References
American Psychiatric Association (1994) Diagnostic and Statistical Manual of Mental Disorders. 4th ed.(DSM-IV). American Psychiatric Association, Washington
Kessler RC, Sonnega A, Bromet E et al (1995) Posttraumatic stress disorder in the National Comorbidity Survey. Arch Gen Psychiatry 52:1048–1060
Vieweg WV, Julius DA, Fernandez A et al (2006) Posttraumatic stress disorder: clinical features, pathophysiology, and treatment. Am J Med 119:383–390
Shin LM, Rauch SL, Pitman RK (2006) Amygdala, medial prefrontal cortex, and hippocampal function in PTSD. Ann N Y Acad Sci 1071:67–79
Bremner JD, Elzinga B, Schmahl C et al (2008) Structural and functional plasticity of the human brain in posttraumatic stress disorder. Prog Brain Res 167:171–186
Shin LM, Rauch SL, Pitman RK (2006) Amygdala, medial prefrontal cortex, and hippocampal function in PTSD. Ann N Y Acad Sci 1071:67–79
Lucki I (1998) The spectrum of behaviors influenced by serotonin. Biol Psychiatry 44:151–162
McAllister-Williams RH, Ferrier IN, Young AH (1998) Mood and neuropsychological function in depression: the role of corticosteroids and serotonin. Psychol Med 28:573–584
Lowry C, Hale MV, Evans A et al (2008) Serotonergic systems, anxiety, and affective disorder: focus on the dorsomedial part of the dorsal raphe nucleus. Ann N Y Acad Sci 1148:86–94
Spannuth BM, Hale MW, Evans AK et al (2011) Investigation of a central nucleus of the amygdala/dorsal raphe nucleus serotonergic circuit implicated in fear-potentiated startle. Neuroscience 179:104–119
Schröder M (2008) Endoplasmic reticulum stress responses. Cell Mol Life Sci 65:862–894
Ni M, Lee AS (2007) ER chaperones in mammalian development and human diseases. FEBS Lett 581:3641–3651
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Science 334:1081–1086
Zhang K, Kaufman RJ (2004) Signaling the unfolded protein response from the endoplasmic reticulum. J Biol Chem 279:25935–25938
Rasheva VI, Domingos PM (2009) Cellular responses to endoplasmic reticulum stress and apoptosis. Apoptosis 14:996–1007
Rao RV, Peel A, Logvinova A et al (2002) Coupling endoplasmic reticulum stress to the cell death program: role of the ER chaperone GRP78. FEBS Lett 514:122–128
Kamauchi S, Nakatani H, Nakano C et al (2005) Gene expression in response to endoplasmic reticulum stress in Arabidopsis thaliana. FEBS J 272:3461–3476
Nakanishi T, Shimazawa M, Sugitani S et al (2013) Role of endoplasmic reticulum stress in light-induced photoreceptor degeneration in mice. J Neurochem 125:111–124
Kunte MM, Choudhury S, Manheim JF et al (2012) ER stress is involved in T17Mrhodopsin-induced retinal degeneration. Invest Ophthalmol Vis Sci 53:3792–3800
Shinde VM, Sizova OS, Lin JH et al (2012) ER stress in retinal degeneration in S334ter Rho rats. PLoS One 7:e33266
Xie H, Han F, Shi X (2012) Single-prolonged stress induce changes of CaM/CaMKIIα in the rats of dorsal raphe nucleus. Neurochem Res 37:1043–1049
Liberzon I, López JF, Flagel SB et al (1999) Differential regulation of hippocampal glucocorticoid receptors mRNA and fast feedback: relevance to post-traumatic stress disorder. J Neuroendocrinol 11:11–17
Iwamoto Y, Morinobu S, Takahashi T et al (2007) Single prolonged stress increases contextual freezing and the expression of glycine transporter 1 and vesicle-associated membrane protein 2 mRNA in the hippocampus of rats. Prog Neuropsychopharmacol Biol Psychiatry 31:642–651
Liberzon I, Krstov M, Young EA (1997) Stress-restress: effects on ACTH and fast feedback. Psychoneuroendocrinology 22:443–453
Khan S, Liberzon I (2004) Topiramate attenuates exaggerated acoustic startle in an animal model of PTSD. Psychopharmacology 172:225–229
Takahashi T, Morinobu S, Iwamoto Y et al (2006) Effect of paroxetine on enhanced contextual fear induced by single prolonged stress in rats. Psychopharmacology 189:165–173
Palkovits M, Brownstein M, Kizer JS et al (1976) Effect of stress on serotonin concentration and tryptophan hydroxylase activity of brain nuclei. Neuroendocrinology 22:298–304
Arora RC, Fichtner CG, O’Connor F, Crayton JW (1993) Paroxetine binding in the blood platelets of post-traumatic stress disorder patients. Life Sci 53:919–928
Grah M, Mihanović M, Svrdlin P et al (2010) Serotonin and cortisol as suicidogenic factors in patients with PTSD. Coll Antropol 34:1433–1439
Baldwin D, Woods R et al (2011) Efficacy of drug treatments for generalised anxiety disorder: systematic review and meta-analysis. BMJ 342:d1199
Koen N, Stein DJ (2011) Pharmacotherapy of anxiety disorders: a critical review. Dialog Clin Neurosci 13(4):423–437
Abrams JK, Johnson PL, Hollis JH et al (2004) Anatomic and functional topography of the dorsal raphe nucleus. Ann N Y Acad Sci 1018:46–57
Little E et al (1994) The glucose-regulated proteins (GRP78 and GRP94): functions, gene regulation, and applications. Crit Rev Eukaryot Gene Expr 4:1–18
Michalak M, Groenendyk J, Szabo E et al (2009) Calreticulin, a multiprocess calcium-buffering chaperone of the endoplasmic reticulum. Biochem J 417:651–666
Schonthal AH (2013) Pharmacological targeting of endoplasmic reticulum stress signaling in cancer. Biochem Pharmacol 85:653–666
Logue SE, Cleary P, Saveljeva S et al (2013) New directions in ER stress-induced cell death. Apoptosis 18:537–546
Liu D, Xiao B, Han F et al (2012) Single-prolonged stress induces apoptosis in dorsal raphe nucleus in the rat model of posttraumatic stress disorder. BMC Phychiatry 12:211
Tate CG, Whiteley E, Betenbaugh MJ (1999) Molecular chaperones stimulate the functional expression of the cocaine-sensitive serotonin transporter. J Biol Chem 274:17551–17558
Acknowledgments
The authors are grateful to all of the staff members of the China Medical University Experiment Center for their technical support. In addition, this research was supported by a grant from the National Natural Science Foundation of China (No. 81171282 and No. 31200772) and China National Doctoral Fund (No. 20132104110021).
Conflict of interest
The authors indicate no potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xie, J., Han, F. & Shi, Y. The Unfolded Protein Response is Triggered in Rat Neurons of the Dorsal Raphe Nucleus After Single-Prolonged Stress. Neurochem Res 39, 741–747 (2014). https://doi.org/10.1007/s11064-014-1262-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-014-1262-x