Abstract
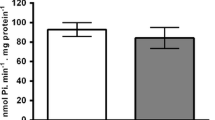
Nucleotides and nucleosides play an important role in neurodevelopment acting through specific receptors. Ectonucleotidases are the major enzymes involved in controlling the availability of purinergic receptors ligands. ATP is co-released with several neurotransmitters and is the most important source of extracellular adenosine by catabolism exerted by ectonucleotidases. The main ectonucleotidases are named NTPDases (1–8) and 5′-nucleotidase. Adenosine is a powerful modulator of neurotransmitter release. Caffeine blocks adenosine receptor activity as well as adenosine-mediated neuromodulation. Considering the susceptibility of the immature brain to caffeine and the need for correct purinergic signaling during fetal development, we have analyzed the effects of caffeine exposure during gestational and lactational periods on nucleotide degradation and ectonucleotidase expression from the hippocampi of 7-, 14- and 21-days-old rats. Nucleotides hydrolysis was assessed by colorimetric determination of inorganic phosphate released. Ectonucleotidases expression was performed by RT-PCR. ATP and ADP hydrolysis displayed parallel age-dependent decreases in both control and caffeine-treated groups. AMP hydrolysis increased with caffeine treatment in 7-days-old rats (75%); although there was no significant difference in AMP hydrolysis between control (non caffeine-treated) rats and 14- or 21-days caffeine-treated rats. ADP hydrolysis was not affected by caffeine treatment. Caffeine treatment in 7- and 14-days-old rats decreased ATP hydrolysis when compared to the control group (19% and 60% decrease, respectively), but 21-days-treated rats showed an increase in ATP hydrolysis (39%). Expression levels of NTPDase 1 and 5 decreased in hippocampi of caffeine-treated rats. The expression of 5′-nucleotidase was not affected after caffeine exposure. The changes observed in nucleotide hydrolysis and ectonucleotidases expression could promote subtle effects on normal neural development considering the neuromodulatory role of adenosine.


Similar content being viewed by others
References
Herlenius E, Lagercrantz H (2004) Development of neurotransmitter systems during critical periods. Exp Neurol 190(1):S8–S21
Fredholm BB, Bättig K, Holmén J, Nehlig A, Zvartau EE (1999) Actions of caffeine in the brain with special reference to factors that contribute to its widespread use. Pharmacol Rev 51(1):83–133
Herrick-Davis K, Chippari S, Luttinger D, Ward SJ (1989) Evaluation of adenosine agonists as potential analgesics. Eur J Pharmacol 162(2):365–369
Costa MS, Botton PH, Mioranzza S, Souza DO, Porciúncula LO (2008) Caffeine prevents age-associated recognition memory decline and changes brain-derived neurotrophic factor and tirosine kinase receptor (TrkB) content in mice. Neuroscience 153(4):1071–1078
Takahashi RN, Pamplona FA, Prediger RD (2008) Adenosine receptor antagonists for cognitive dysfunction: a review of animal studies. Front Biosci 13:2614–2632
Black AM, Pandya S, Clark D, Armstrong EA, Yager JY (2008) Effect of caffeine and morphine on the developing pre-mature brain. Brain Res 1219:136–142
Zimmerberg B, Carr KL, Scott A, Lee HH, Weider JM (1991) The effects of postnatal caffeine exposure on growth, activity and learning in rats. Pharmacol Biochem Behav 39(4):883–888
Da Silva RS, Hoffman A, Souza DO, Lara DR, Bonan CD (2005) Maternal caffeine intake impairs MK-801-induced hyperlocomotion in young rats. Eur J Pharmacol 509(2–3):155–159
Schmidt B, Roberts RS, Davis P, Doyle LW, Barrington KJ, Ohlsson A, Solimano A, Tin W (2007) Long-term effects of caffeine therapy for apnea of prematurity. New Eng J Med 357(19):1893–1902
Aliyu MH, Wilson RE, Zoorob R, Brown K, Alio AP, Clayton H, Salihu HM (2009) Prenatal alcohol consumption and fetal growth restriction: potentiation effect by concomitant smoking. Nicotine Tobacco Res 11(1):36–43
Millar D, Schmidt B (2004) Controversies surrounding xanthine therapy. Semin Neonatol 9:239–244
Adén U, Herlenius E, Tang LQ, Fredholm BB (2000) Maternal caffeine intake has minor effects on adenosine receptor ontogeny in the rat brain. Pediatric Res 48(2):177–183
Léon D, Albasanz JL, Ruíz MA, Fernandéz M, Martín M (2002) Adenosine A1 receptor down-regulation in mothers and fetal brain after caffeine and theophylline treatments to pregnant rats. J Neurochem 82:625–634
Johansson B, Ahlberg S, van der Ploeg I, Brené S, Lindefors N, Persson H, Fredholm BB (1993) Effect of long term caffeine treatment on A1 and A2 adenosine receptor binding and on mRNA levels in rat brain. Naunyn-Schmiedeberg’s Arch Pharmacol 347:407–414
León D, Albasanz JL, Ruíz MA, Martín M (2005) Chronic caffeine or theophylline intake during pregnancy inhibits A1 receptor function in the rat brain. Neuroscience 131:481–489
Rivkees SA, Zhao Z, Porter G, Turner CP (2001) Influences of adenosine on the fetus and newborn. Mol Genet Metab 74:160–171
Turner CP, Yan H, Schwartz M, Othman T, Rivkees SA (2002) A1 adenosine receptor activation induces ventriculomegaly and white matter loss. Neuroreport 13:1199–1204
Zimmermann H (2001) Ecto-nucleotidases: some recent developments and a note on nomenclature. Drug Dev Res 52:44–56
Abbracchio MP, Burnstock G, Verkhratsky A, Zimmermann H (2009) Purinergic signalling in the nervous system: an overview. Trends Neurosci 32(1):19–29
Zimmermann H (2006) Ectonucleotidases in the nervous system. Novartis Found Symp 276:113–128
Braun N, Sévigny J, Mishra SK, Robson SC, Barth SW, Gerstberger R, Hammer K, Zimmermann H (2003) Expression of the ecto-ATPase NTPDase2 in the germinal zones of the developing and adult rat brain. Eur J Neurosci 17(7):1355–1364
Langer D, Ikehara Y, Takebayashi H, Hawkes R, Zimmermann H (2007) The ectonucleotidases alkaline phosphatase and nucleoside triphosphate diphosphohydrolase 2 are associated with subsets of progenitor cell populations in the mouse embryonic, postnatal and adult neurogenic zones. Neuroscience 150(4):863–879
Stanojevic I, Bjelobabab I, Nedeljkovicc N, Drakulica D, Petrovica S, Stojiljkovicb M, Horvat A (2011) Ontogenetic profile of ecto-5′-nucleotidase in rat brain synaptic plasma membranes. Int J Dev Neurosci 29:397–403
Bruno AN, Bonan CD, Wofchuk ST, Sarkis JJ, Battastini AMO (2002) ATP diphosphohydrolase (NTPDase 1) in rat hippocampal slices and effect of glutamate on the enzyme activity in different phases of development. Life Sci 71(2):215–225
Acquas E, Tanda G, Di Chiara G (2002) Differencial effects of caffeine on dopamine and acetylcholine transmission in brain areas of drug-naïve and caffeine-preteated rats. Neuropsychopharmacology 27:182–193
Solinas M, Ferré S, You ZB, Karcz-Kubicha M, Popoli P, Goldberg SR (2002) Caffeine induces dopamine and glutamate release in the shell of the nucleus accumbens. J Neurosci 22(15):6321–6324
Da Silva RS, Bruno AN, Battastini AMO, Sarkis JJF, Lara DR, Bonan CD (2003) Acute caffeine treatment increases extracellular nucleotide hydrolysis from rat striatal and hippocampal synaptosomes. Neurochem Res 28(8):1273–1278
Chan K, Delfert D, Junguer KD (1986) A direct colorimetric assay for Ca 2-ATPase activity. Anal Biochem 157:375–380
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:218–254
Oses JP, Viola GG, de Paula Cognato G, Júnior VH, Hansel G, Böhmer AE, Leke R, Bruno AN, Bonan CD, Bogo MR, Portela LV, Souza DO, Sarkis JJ (2007) Pentylenetetrazol kindling alters adenine and guanine nucleotide catabolism in rat hippocampal slices and cerebrospinal fluid. Epilep Res 75(2–3):104–111
Bona E, Adén U, Fredholm BB, Hagberg H (1995) The effect of long term caffeine treatment on hypoxic-ischemic brain damage in the neonate. Pediatr Res 38(3):312–318
Benedetti MD, Bower JH, Maraganore DM, McDonnell SK, Peterson BJ, Ahlskog JE, Schaid DJ, Rocca WA (2000) Smoking, alcohol, and coffee consumption preceding Parkinson’s disease: a case-control study. Neurology 55(9):1350–1358
Ross GW, Abbott RD, Petrovitch H, Morens DM, Grandinetti A, Tung KH, Tanner CM, Masaki KH, Blanchette PL, Curb JD, Popper JS, White LR (2000) Association of coffee and caffeine intake with the risk of Parkinson disease. J Am Med Assoc 283(20):2674–2679
Lindsay J, Laurin D, Verreault R, Hébert R, Helliwell B, Hill GB, McDowell I (2002) Risk factors for Alzheimer’s disease: a prospective analysis from the Canadian study of health and aging. Am J Epidem 156(5):445–453
Dall’Igna OP, Porciúncula LO, Souza DO, Cunha RA, Lara DR (2003) Neuroprotection by caffeine and adenosine A2A receptor blockade of beta-amyloid neurotoxicity. Br J Pharmacol 138(7):1207–1209
Arendash GW, Schleif W, Rezai-Zadeh K, Jackson EK, Zacharia LC, Cracchiolo JR, Shippy D, Tan J (2006) Caffeine protects Alzheimer’s mice against cognitive impairment and reduces brain beta-amyloid production. Neuroscience 142(4):941–952
Higdon JV, Frei B (2006) Coffee and health: a review of recent human research. Crit Rev Food Sci Nutri 46(2):101–123
Ritchie K, Carrière I, de Mendonca A, Portet F, Dartigues JF, Rouaud O, Barberger-Gateau P, Ancelin ML (2007) The neuroprotective effects of caffeine: a prospective population study (the three city study). Neurology 69(6):536–545
Karcz-Kubicha M, Antoniou K, Terasmaa A, Quarta D, Solinas M, Justinova A, Pezzola A, Regio R, Muller CE, Fuxe K, Goldberg SR, Popoli P, Ferre S (2003) Involvement of adenosine A1 and A2a receptors in the motor effects of caffeine after its acute and chronic administration. Neuropsichopharmacology 28(7):1281–1291
Noschang CG, Krolow R, Pettenuzzo LF, Avila MC, Fachin A, Arcego D, von Pozzer Toigo E, Crema LM, Diehl LA, Vendite D, Dalmaz C (2009) Interactions between chronic stress and chronic consumption of caffeine on the enzymatic antioxidant system. Neurochem Res 34(9):1568–1574
Da Silva RS, Richetti SK, Da Silveira VG, Battastini AM, Bogo MR, Lara DR, Bonan CD (2008) Maternal caffeine intake affects acetylcholinesterase in hippocampus of neonate rats. Int J Dev Neurosci 26(3–4):339–343
Gasior M, Jaszyna M, Peters J, Goldberg SR (2000) Changes in the ambulatory activity and discriminative stimulus effects of psychostimulant drugs in rats chronically exposed to caffeine: effect of caffeine dose. J Pharmacol Exp Therap 295(3):1101–1111
Zhang LI, Poo MM (2001) Electrical activity and development of neural circuits. Nat Neurosci 4:1207–1214
Neary JT (1996) Trophic actions of extracellular ATP on astrocytes, synergistic interactions with fibroblast growth factors and underlying signal transduction mechanisms. Ciba Found Symp 198:130–139
Rathbone MP, Middlemiss PJ, Gysbers JW, Andrew C, Herman MA, Reed JK, Ciccarelli R, Di Iorio P, Caciagli F (1999) Trophic effects of purine in neurons and glial cells. Prog Neurobiol 59(6):663–690
Nedeljkovic N, Banjac A, Horvat A, Stojiljkovic M, Nikezic G (2005) Developmental profile of NTPDase activity in synaptic plasma membranes isolated from rat cerebral cortex. Int J Dev Neurosci 23:45–51
Li W, Dai S, An J, Li P, Chen X, Xiong R, Liu P, Wang H, Zhao Y, Zhu M, Liu X, Zhu P, Chen JF, Zhou Y (2008) Chronic but not acute treatment with caffeine attenuates traumatic brain injury in the mouse cortical impact model. Neuroscience 151(4):1198–1207
Queiroz G, Meyer DK, Meyer A, Starke K, von Kugelgen I (1999) A study of the mechanism of the release of ATP from rat cortical astroglial cells evoked by activation of glutamate receptors. Neuroscience 91:1171–1181
Sneddon P, Burnstock G (1984) ATP as a co-transmitter in rat tail artery. Eur J Pharmacol 106(1):149–152
Weaver DR (1996) A1—adenosine receptor gene expression in fetal rat brain. Dev Brain Res 94:205–223
Desfrere L, Olivier P, Schwendimann L, Catherine V, Gressens P (2007) Transient inhibition of astrocytogenesis in developing mouse brain following postanatal caffeine exposure. Pediatric Res 62(5):604–609
Heyliger CE, Panagia V, Dhalla NS (1981) Effect of cyclic AMP phosphodiesterase inhibitors on cardiac sarcolemmal 5′-nucleotidase. J Pharmacol Exp Therap 217(2):489–493
Acknowledgments
This work was supported by CNPq (Conselho Nacional de Pesquisa e Desenvolvimento – Brasil), Fundação de Amparo a Pesquisa do Estado do Rio Grande do Sul (FAPERGS) and by the FINEP research grant “Rede Instituto Brasileiro de Neurociência (IBN-Net)” 01.06.0842-00.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Da Silva, R.S., Richetti, S.K., Tonial, E.M. et al. Profile of Nucleotide Catabolism and Ectonucleotidase Expression from the Hippocampi of Neonatal Rats After Caffeine Exposure. Neurochem Res 37, 23–30 (2012). https://doi.org/10.1007/s11064-011-0577-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-011-0577-0