Abstract
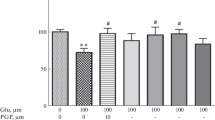
The mechanisms of protective effect of N-methyl-D-aspartate (NMDA) receptor stimulation on apoptosis of neurons at their early stage of development are poorly understood. In the present study, we investigated the effects of NMDA on staurosporine (St)- and low-potassium (LP)-evoked apoptotic cell death in primary cerebellar granule cell (CGC) cultures at 7 days in vitro (DIV). We found that NMDA (200 μM) attenuated the St (0.5 μM)- and LP (5 mM KCl)-induced neuronal cell death in 7 but not 12 DIV CGC as confirmed by LDH release and MTT reduction assays. Moreover, NMDA attenuated St-and LP-evoked DNA fragmentation and cytosolic apoptosis inducing factor (AIF) protein level but not caspase-3 activation induced by both pro-apoptotic factors. Neuroprotective effects of NMDA on St-induced apoptosis in CGC were attenuated by inhibitors of ERK/MAPK-signaling, PD 98059 and U0126 but not by NMDA receptor antagonists, AP-5 (100 μM) and MK-801 (1 μM) or by inhibitors of PI3-K/Akt pathway (LY 294002 and wortmannin). In contrast to staurosporine model of apoptosis, AP-5 and MK-801 but not inhibitors of PI3-K/Akt and MAPK/ERK1/2 prevented the NMDA-mediated neuroprotection in LP-induced apoptosis of CGC. In separate experiments, we observed also the anti-apoptotic action of NMDA on St (0.5 μM)- and salsolinol (250 μM)-evoked cell death in human neuroblastoma SH-SY5Y cells without its influence on caspase-3 activity, induced by these pro-apoptotic factors. These data indicate that neuroprotection evoked by NMDA in CGC strongly depends on used pro-apoptotic agent and could engage NMDA channel function or be connected with the activation of pro-survival MAPK/ERK1/2 pathway. It is also suggested that anti-apoptotic effects of NMDA is connected with inhibition of fragmentation of DNA via caspase-3-independent mechanism.









Similar content being viewed by others
Abbreviations
- AIF:
-
Apoptosis inducing factor
- Akt, PKB:
-
Serine/threonine protein kinase Akt, protein kinase B
- BDNF:
-
Brain-derived neurotrophic factor
- CREB cAMP:
-
Response element-binding protein
- DIV:
-
Days in vitro
- ERK1/2:
-
Extracellularly regulated protein kinase 1/2
- LDH:
-
Lactate dehydrogenase
- LP:
-
Low-potassium
- MAPK:
-
Mitogen-activated protein kinase
- NMDA:
-
N-Methyl-d-aspartate
- NMDAR:
-
N-Methyl-d-aspartate receptor
- NR2B:
-
NMDA receptor 2B subunit
- PI3-K:
-
Phosphatidylinositol-3-OH kinase
- PKA:
-
Protein kinase A
- PKC:
-
Protein kinase C
- RA-SHSY5Y:
-
Retinoic acid-differentiated SH-SY5Y cells
- Sals:
-
Salsolinol, 1-methyl-6, 7-dihydroxy-1,2,3,4-tetrahydroisoquinoline hydrobromide
- St:
-
Staurosporine
- UN-SHSY5Y:
-
Undifferentiated SH-SY5Y cells
References
Wroblewski JT, Danysz W (1989) Modulation of glutamate receptors: molecular mechanisms and functional implications. Annu Rev Pharmacol Toxicol 29:441–474. doi:10.1146/annurev.pa.29.040189.002301
Parson CG, Quack G, Bresink I et al (1995) Comparison of the potency, kinetics and voltage-dependency of a series of uncompetitive NMDA receptor antagonists in vitro with anticonvulsive and motor impairment activity in vivo. Neuropharmacology 34:1239–1258. doi:10.1016/0028-3908(95)00092-K
Olney JW (1994) Excitatory transmitter neurotoxicity. Neurobiol Aging 15:259–260. doi:10.1016/0197-4580(94)90127-9
Durkin JP, Tremblay R, Chakravarthy B et al (1997) Evidence that the early loss of membrane protein kinase C is necessary step in the excitatory amino acid-induced death of primary cortical neurons. J Neurochem 68:1400–1412
Michaelis EK (1998) Molecular biology of glutamate receptors in the central nervous system and their role in excitotoxicity, oxidative stress and aging. Prog Neurobiol 54:369–415. doi:10.1016/S0301-0082(97)00055-5
Hoffmann H, Gremme T, Hatt H, Gottmann K (2000) Synaptic activity-dependent developmental regulation of NMDA receptor subunit expression in cultured neocortical neurons. J Neurochem 75:1590–1599. doi:10.1046/j.1471-4159.2000.0751590.x
Mattson MP (2003) Excitotoxic and excitoprotective mechanisms: abundant targets for the prevention and treatment of neurodegenerative disorders. Neuromolecular Med 3:65–94. doi:10.1385/NMM:3:2:65
Salinska E, Danysz W, Lazarewicz JW (2005) The role of excitotoxicity in neurodegeneration. Folia Neuropathol 43:322–339
Haberny KA, Paule MG, Scallet AC et al (2002) Ontogeny of the N-methyl-D-aspartate (NMDA) receptor system and susceptibility to neurotoxicity. Toxicol Sci 68:9–17. doi:10.1093/toxsci/68.1.9
Riedel G, Platt B, Micheau J (2003) Glutamate receptor function in learning and memory. Behav Brain Res 140:1–47. doi:10.1016/S0166-4328(02)00272-3
McDonald JW, Johnston MV (1990) Physiological and pathophysiological roles of excitatory amino acids during central nervous system development. Brain Res Brain Res Rev 15:41–70. doi:10.1016/0165-0173(90)90011-C
Ikonomidou C, Bosch F, Miksa M et al (1999) Blockade of NMDA receptors and apoptotic neurodegeneration in the developing brain. Science 283:70–74. doi:10.1126/science.283.5398.70
Olney JW (2002) New insights and new issues in developmental neurotoxicology. Neurotoxicology 23:659–668. doi:10.1016/S0161-813X(01)00092-4
Olney JW, Wozniak DF, Jevtovic-Todorovic V et al (2002) Drug-induced apoptotic neurodegeneration in the developing brain. Brain Pathol 12:488–498
Takadera T, Sakamoto Y, Ohyashiki T (2004) NMDA receptor 2B-selective antagonist ifenprodil-induced apoptosis was prevented by glycogen synthase kinase-3 inhibitors in cultured rat cortical neurons. Brain Res 1020:196–203. doi:10.1016/j.brainres.2004.06.035
Kajta M, Trotter A, Lasoń W, Beyer C (2005) Effect of NMDA on staurosporine-induced activation of caspase-3 and LDH release in mouse neocortical and hippocampal cells. Brain Res Dev Brain Res 160:40–52
Xifro X, Malagelada C, Minano A, Rodriguez-Alvarez J (2005) Brief exposure to NMDA produces long-term protection of cerebellar granule cells from apoptosis. Eur J NeuroSci 21:827–840. doi:10.1111/j.1460-9568.2005.03935.x
Zhu D, Wu X, Strauss KI, Lipsky RH et al (2005) N-methyl-D-aspartate and TrkB receptors protect neurons against glutamate excitotoxicity through an extracellular signal-regulated kinase pathway. J Neurosci Res 80:104–113. doi:10.1002/jnr.20422
Hack N, Hidaka H, Wakefield MJ, Balazs R (1993) Promotion of granule cell survival by high K+ or excitatory amino acid treatment and Ca2+/calmodulin-dependent protein kinase activity. Neuroscience 57:9–20. doi:10.1016/0306-4522(93)90108-R
Yao CJ, Lin CW, Lin-Shiau SY (1999) Altered intracellular calcium level in association with neuronal death induced by lithium chloride. J Formos Med Assoc 98:820–826
See V, Boutillier AL, Bito H, Loeffler JP (2001) Calcium/calmodulin-dependent protein kinase type IV (CaMKIV) inhibits apoptosis induced by potassium deprivation in cerebellar granule neurons. FASEB J 15:134–144. doi:10.1096/fj.00-0106com
Bazan-Peregrino M, GutiAcrrez-Kobeh L, Moran J (2007) Role of brain-derived neurotrophic factor in the protective action of N-methyl-D-aspartate in the apoptotic death of cerebellar granule neurons induced by low potassium. J Neurosci Res 85:332–341. doi:10.1002/jnr.21112
Bessho Y, Nakanishi S, Nawa H (1993) Glutamate receptor agonists enhance the expression of BDNF mRNA in cultured cerebellar granule cells. Brain Res Mol Brain Res 18:201–208. doi:10.1016/0169-328X(93)90190-Z
Favaron M, Manev RM, Rimland JM et al (1993) NMDA-stimulated expression of BDNF mRNA in cultured cerebellar granule neurones. NeuroReport 4:1171–1174. doi:10.1097/00001756-199307000-00033
Lipsky RH, Xu K, Zhu D et al (2001) Nuclear factor kappaB is a critical determinant in N-methyl-D-aspartate receptor-mediated neuroprotection. J Neurochem 78:254–264. doi:10.1046/j.1471-4159.2001.00386.x
Yamagishi S, Matsumoto T, Yokomaku D et al (2003) Comparison of inhibitory effects of brain-derived neurotrophic factor and insulin-like growth factor on low potassium-induced apoptosis and activation of p38 MAPK and c-Jun in cultured cerebellar granule neurons. Brain Res Mol Brain Res 119:184–191. doi:10.1016/j.molbrainres.2003.09.009
Xifro X, Falluel-Morel A, Minano A et al (2006) N-methyl-D-aspartate blocks activation of JNK and mitochondrial apoptotic pathway induced by potassium deprivation in cerebellar granule cells. J Biol Chem 281:6801–6812. doi:10.1074/jbc.M504571200
Zhang FX, Rubin R, Rooney TA (1998) N-methyl-D-aspartate inhibits apoptosis through activation of phosphatidylinositol 3-kinase in cerebellar granule neurons. A role for insulin receptor substrate-1 in the neurotrophic action of n-methyl-D-aspartate and its inhibition by ethanol. J Biol Chem 273:26596–26602. doi:10.1074/jbc.273.41.26596
Valera E, Sanchez-Martin FJ, Ferrer-Montiel AV et al (2008) NMDA-induced neuroprotection in hippocampal neurons is mediated through the protein kinase A and CREB (cAMP-response element-binding protein) pathway. Neurochem Int 53:148–154. doi:10.1016/j.neuint.2008.07.007
Singh J, Kaur G (2005) Neuroprotection mediated by subtoxic dose of NMDA in SH-SY5Y neuroblastoma cultures: activity-dependent regulation of PSA-NCAM expression. Brain Res Mol Brain Res 137:223–234. doi:10.1016/j.molbrainres.2005.03.012
Papadia S, Soriano FX, Léveillé F et al (2008) Synaptic NMDA receptor activity boosts intrinsic antioxidant defenses. Nat Neurosci 11:476–487. doi:10.1038/nn2071
Balazs R, Jorgensen OS, Hack N (1998) N-methyl-D-aspartate promotes the survival of cerebellar granule cells in culture. Neuroscience 27:437–451. doi:10.1016/0306-4522(88)90279-5
Contestabile A (2002) Cerebellar granule cells as a model to study mechanisms of neuronal apoptosis or survival in vivo and in vitro. Cerebellum 1:41–55. doi:10.1080/147342202753203087
Jantas D, Pytel M, Mozrzymas JW et al (2008) The attenuating effect of memantine on staurosporine-, salsolinol- and doxorubicin-induced apoptosis in human neuroblastoma SH-SY5Y cells. Neurochem Int 52:864–877. doi:10.1016/j.neuint.2007.10.003
Nagatsu T (1997) Isoquinoline neurotoxins in the brain and Parkinson’s disease. Neurosci Res 29:99–111. doi:10.1016/S0168-0102(97)00083-7
Maruyama W, Naoi M (2002) Cell death in Parkinson’s disease. J Neurol 242:6–10
Naoi M, Maruyama W, Akao Y, Yi H (2002) Dopamine-derived endogenous N-methyl-(R)-salsolinol: its role in Parkinson’s disease. Neurotoxicol Teratol 24:579–591. doi:10.1016/S0892-0362(02)00211-8
Antkiewicz-Michaluk L (2002) Endogenous risk factors in Parkinson’s disease: dopamine and tetrahydroisoquinolines. Pol J Pharmacol 54:567–572
Segura-Aguilar J, Kostrzewa RM (2004) Neurotoxins and neurotoxic species implicated in neurodegeneration. Neurotox Res 6:615–630
Brewer GJ (1995) Serum-free B27/neurobasal medium supports differentiated growth of neurones from the striatum, substantia nigra, septum, cerebral cortex, cerebellum and dentate gyrus. J Neurosci Res 42:674–683. doi:10.1002/jnr.490420510
Kajta M, Domin H, Grynkiewicz G, Lason W (2007) Genistein inhibits glutamate-induced apoptotic processes in primary neuronal cell cultures: an involvement of aryl hydrocarbon receptor and estrogen receptor/glycogen synthase kinase-3beta intracellular signaling pathway. Neuroscience 145:592–604. doi:10.1016/j.neuroscience.2006.11.059
Jantas-Skotniczna D, Kajta M, Lason W (2006) Memantine attenuates staurosporine-induced activation of caspase-3 and LDH release in mouse primary neuronal cultures. Brain Res 1069:145–153. doi:10.1016/j.brainres.2005.11.055
Jakubowicz-Gil J, Rzeski W, Zdzisinska B, Dobrowolski P, Gawron A (2008) Cell death and neuronal arborization upon quercetin treatment in rat neurons. Acta Neurobiol Exp (Warsz) 68:139–146
Pytlowany M, Strosznajder JB, Jeśko H, Cakała M, Strosznajder RP (2008) Molecular mechanism of PC12 cell death evoked by sodium nitroprusside, a nitric oxide donor. Acta Biochim Pol 55:339–347
Cagnoli CM, Kharlamov E, Atabay C et al (1996) Apoptosis induced in neuronal cultures by either the phosphatase inhibitor okadaic acid or the kinase inhibitor staurosporine is attenuated by isoquinolinesulfonamides H-7, H-8, and H-9. J Mol Neurosci 7:65–76. doi:10.1007/BF02736849
Armstrong RC, Aja TJ, Hoang KD et al (1997) Activation of the CED3/ICE-related protease CPP32 in cerebellar granule neurons undergoing apoptosis but not necrosis. J Neurosci 17:553–562
Polazzi E, Gianni T, Contestabile A (2001) Microglial cells protect cerebellar granule neurons from apoptosis: evidence for reciprocal signaling. Glia 36:271–280. doi:10.1002/glia.1115
Wigdal SS, Kirkland RA, Franklin JL, Haak-Frendscho M (2002) Cytochrome c release precedes mitochondrial membrane potential loss in cerebellar granule neuron apoptosis: lack of mitochondrial swelling. J Neurochem 82:1029–1038. doi:10.1046/j.1471-4159.2002.01049.x
Franco-Cea A, Valencia A, Sanchez-Armass S et al (2004) Role of ionic fluxes in the apoptotic cell death of cultured cerebellar granule neurons. Neurochem Res 29:227–238. doi:10.1023/B:NERE.0000010501.25627.0f
Eleuteri S, Polazzi E, Contestabile A (2008) Neuroprotection of microglia conditioned media from apoptotic death induced by staurosporine and glutamate in cultures of rat cerebellar granule cells. Neurosci Lett 448:74–78. doi:10.1016/j.neulet.2008.09.033
Maycotte P, Blancas S, Moran J (2008) Role of inhibitor of apoptosis proteins and Smac/DIABLO in staurosporine-induced cerebellar granule neurons death. Neurochem Res 33:1534–1540. doi:10.1007/s11064-008-9637-5
Lankiewicz S, Marc Luetjens C, Truc Bui N et al (2000) Activation of calpain I converts excitotoxic neuronal death into a caspase-independent cell death. J Biol Chem 275:17064–17071. doi:10.1074/jbc.275.22.17064
Slagsvold HH, Rosseland CM, Jacobs C et al (2003) High molecular weight DNA fragments are processed by caspase sensitive or caspase independent pathways in cultures of cerebellar granule neurons. Brain Res 984:111–121. doi:10.1016/S0006-8993(03)03119-6
Fonfria E, Dare E, Benelli M et al (2002) Translocation of apoptosis-inducing factor in cerebellar granule cells exposed to neurotoxic agents inducing oxidative stress. Eur J NeuroSci 16:2013–2016. doi:10.1046/j.1460-9568.2002.02269.x
Dawson VL, Dawson TM (2004) Deadly conversations: nuclear-mitochondrial cross-talk. J Bioenerg Biomembr 36:287–294. doi:10.1023/B:JOBB.0000041755.22613.8d
Yeste-Velasco M, Folch J, Jiménez A et al (2008) GSK-3 beta inhibition and prevention of mitochondrial apoptosis inducing factor release are not involved in the antioxidant properties of SB-415286. Eur J Pharmacol 588:239–243. doi:10.1016/j.ejphar.2008.04.019
Llansola M, Boscá L, Felipo V, Hortelano S (2003) Ammonia prevents glutamate-induced but not low K(+)-induced apoptosis in cerebellar neurons in culture. Neuroscience 117:899–907. doi:10.1016/S0306-4522(02)00957-0
Ikonomovic S, Kharlamov E, Manev H et al (1997) GABA and NMDA in the prevention of apoptotic-like cell death in vitro. Neurochem Int 31:283–290. doi:10.1016/S0197-0186(96)00159-3
Monyer H, Burnashev N, Laurie DJ et al (1994) Developmental and regional expression in the rat brain and functional properties of four NMDA receptors. Neuron 12:529–540. doi:10.1016/0896-6273(94)90210-0
Thompson CL, Drewery DL, Atkins HD et al (2000) Immunohistochemical localization of N-methyl-D-aspartate receptor NR1, NR2A, NR2B and NR2C/D subunits in the adult mammalian cerebellum. Neurosci Lett 283:85–88. doi:10.1016/S0304-3940(00)00930-7
Zhou M, Baudry M (2006) Developmental changes in NMDA neurotoxicity reflect developmental changes in subunit composition of NMDA receptors. J Neurosci 26:2956–2963. doi:10.1523/JNEUROSCI.4299-05.2006
Volbracht C, van Beek J, Zhu C, Blomgren K, Leist M (2006) Neuroprotective properties of memantine in different in vitro and in vivo models of excitotoxicity. Eur J NeuroSci 23:2611–2622. doi:10.1111/j.1460-9568.2006.04787.x
Alavez S, Blancas S, Moran J (2006) Effect of NMDA antagonists on the death of cerebellar granule neurons at different ages. J Neurosci Lett 398:241–245. doi:10.1016/j.neulet.2006.01.002
Alavez S, Blancas S, Moran J (2006) Effect of N-methyl-D-aspartate receptor blockade on caspase activation and neuronal death in the developing rat cerebellum. Neurosci Lett 404:176–181. doi:10.1016/j.neulet.2006.05.039
Klimaviciusa L, Safiulina D, Kaasik A et al (2008) The effects of glutamate receptor antagonists on cerebellar granule cell survival and development. Neurotoxicology 29:101–108. doi:10.1016/j.neuro.2007.09.006
Lin WW, Wang CW, Chuang DM (1997) Effects of depolarization and NMDA antagonists on the role survival of cerebellar granule cells: a pivotal role for protein kinase C isoforms. J Neurochem 68:2577–2586
Krapivinsky G, Krapivinsky L, Manasian Y et al (2003) The NMDA receptor is coupled to the ERK pathway by a direct interaction between NR2B and RasGRF1. Neuron 40:775–784
Kim MJ, Dunah AW, Wang YT, Sheng M (2005) Differential roles of NR2A- and NR2B-containing NMDA receptors in Ras-ERK signaling and AMPA receptor trafficking. Neuron 46:745–760. doi:10.1016/j.neuron.2005.04.031
Zhong J, Carrozza DP, Williams K et al (1995) Expression of mRNAs encoding subunits of the NMDA receptor in developing rat brain. J Neurochem 64:531–539
Tremblay R, Hewitt K, Lesiuk H et al (1999) Evidence that brain-derived neurotrophic factor neuroprotection is linked to its ability to reverse the NMDA-induced inactivation of protein kinase C in cortical neurons. J Neurochem 72:102–111. doi:10.1046/j.1471-4159.1999.0720102.x
Zheng X, Zhang L, Wang AP et al (1999) Protein kinase C potentiation of N-methyl-D-aspartate receptor activity is not mediated by phosphorylation of N-methyl-D-aspartate receptor subunits. Proc Natl Acad Sci USA 96:15262–15267. doi:10.1073/pnas.96.26.15262
Sala C, Rudolph-Correia S, Sheng M (2000) Developmentally regulated NMDA receptor-dependent dephosphorylation of cAMP response element-binding protein (CREB) in hippocampal neurons. J Neurosci 20:3529–3536
Dave JR, Williams AJ, Moffett JR et al (2003) Studies of neuronal apoptosis in primary forebrain cultures: neuroprotective/anti-apoptotic action of NR2B NMDA antagonists. Neurotox Res 5:255–264
Hetman M, Kharebava G (2006) Survival signaling pathways activated by NMDA receptors. Curr Top Med Chem 6:787–799. doi:10.2174/156802606777057553
Morikawa N, Naoi M, Maruyama W, Ohta S, Kotake Y, Kawai H, Niwa T, Dostert P, Mizuno Y (1998) Effects of various tetrahydroisoquinoline derivatives on mitochondrial respiration and the electron transfer complexes. J Neural Transm 105(6–7):677–688. doi:10.1007/s007020050087
Akao Y, Nakagawa Y, Maruyama W et al (1999) Apoptosis induced by an endogenous neurotoxin, N-methyl(R)salsolinol, is mediated by activation of caspase 3. Neurosci Lett 267:153–156. doi:10.1016/S0304-3940(99)00361-4
Naoi M, Maruyama W, Takahashi T et al (2000) Involvement of endogenous N-methyl(R)salsolinol in Parkinson’s disease: induction of apoptosis and protection by (-)deprenyl. J Neural Transm Suppl 58:111–121
Wanpen S, Govitrapong P, Shavali S et al (2004) Salsolinol, a dopamine-derived tetrahydroisoquinoline, induces cell death by causing oxidative stress in dopaminergic SH-SY5Y cells, and the said effect is attenuated by metallothionein. Brain Res 1005:67–76. doi:10.1016/j.brainres.2004.01.054
Marini AM, Paul SM (1992) N-methyl-D-aspartate receptor-mediated neuroprotection in cerebellar granule cells requires new RNA and protein synthesis. Proc Natl Acad Sci USA 89:6555–6559. doi:10.1073/pnas.89.14.6555
Acknowledgments
This study was supported by statutory funds of the Institute of Pharmacology Polish Academy of Sciences. We would like to thank Ms Barbara Korzeniak for her excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jantas, D., Lason, W. Different Mechanisms of NMDA-Mediated Protection Against Neuronal Apoptosis: A Stimuli-Dependent Effect. Neurochem Res 34, 2040–2054 (2009). https://doi.org/10.1007/s11064-009-9991-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-009-9991-y