Abstract
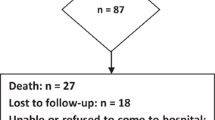
Brain tumors and anti-cancer treatments can cause a wide range of cognitive deficits that in turn, being a major cause of disability, significantly affect patients’ independence and quality of life. To evaluate the neurocognitive status of a non selected population of brain tumors outpatients, investigating the correlation with clinical and demographic variables. This prospective cross-sectional survey enrolled consecutive outpatients with a histopathologically confirmed diagnosis of brain tumor. All the patients were evaluated with a battery of standardized neuropsychological tests assessing language, memory, logical-executive functions, attention, visuo-constructional abilities. An univariate regression analysis was performed to assess the impact of socio-demographical and clinical variables on the presence of cognitive impairment. 147 patients (61F/86M, mean age 52.8 ± 13.3, mean schooling 12.7 ± 4 were enrolled into the study. Out of the 147 patients evaluated, 80 (54.4 %) showed cognitive impairment: 43 (53.75 %) presented a multidomain impairment, while 37 (46.25 %) patients revealed cognitive deficits limited respectively to language (n:13, 16.25 %), memory (n:11, 13.75 %), attention (n:7, 8.75 %), logical-executive functions (n:5, 6.25 %), visuo-spatial abilities (n:1, 1.25 %). At the regression analysis the variables significantly related to the development of cognitive impairment were age (p = 0.04), lesion side (p = 0.00), chemotherapy (p = 0.03). As advances in anti-cancer treatment have prolonged life expectancy of neuro-oncological patients, standard clinical endpoints can’t be limited to just survival or progression free survival, but have to consider clinical benefits on both motor and cognitive function and in general quality of life. Hence evaluation of new therapeutic strategies should routinely include longitudinal neuropsychological assessment.
Similar content being viewed by others
References
Bell KR, O’Dell MW, Barr K, Yablon SA (1998) Rehabilitation of the patient with brain tumor. Arch Phys Med Rehabil 79:S37–S46
Mukand JA, Blackinton DD, Crincoli MG, Lee JJ, Santos BB (2001) Incidence of neurologic deficits and rehabilitation of patients with brain tumors. Am J Phys Med Rehabil 80:346–350
Klein M, Heimans JJ, Aaronson NK, van der Ploeg HM, Grit J, Muller M, Postma TJ, Mooij JJ, Boerman RH, Beute GN, Ossenkoppele GJ, van Imhoff GW, Dekker AW, Jolles J, Slotman BJ, Struikmans H, Taphoorn MJ (2002) Effect of radiotherapy and other treatment-related factors on mild-term to long-term cognitive sequelae in low-grade gliomas: a comparative study. Lancet 360:1361–1368
Tucha O, Smely C, Preier M, Lange KW (2000) Cognitive deficits before treatment among patients with brain tumors. Neurosurgery 47:324–333
Klein M, Taphoorn MJ, Heimans JJ, van der Ploeg HM, Vandertop WP, Smit EF, Leenstra S, Tulleken CA, Boogerd W, Belderbos JS, Cleijne W, Aaronson NK (2001) Neurobehavioural status and health-related quality of life in newly diagnosed high-grade glioma patients. J Clin Oncol 19:4037–4047
Taphoorn MJB (2003) Neurocognitive sequelae in the treatment of low-grade gliomas. Semin Oncol 30(6 Suppl 19):45–48
Meyers CA, Smith JA, Bezjak A, Mehta MP, Liebmann J, Illidge T, Kunkler I, Caudrelier JM, Eisenberg PD, Meerwaldt J, Siemers R, Carrie C, Gaspar LE, Curran W, Phan SC, Miller RA, Renschler MF (2004) Neurocognitive function and progression in patients with brain metastases treated with whole-brain radiation and motexafin gadolinium: results of a randomized phase III trial. J Clin Oncol 22:157–165
van Nieuwenhuizen D, Klein M, Stalpers LJ, Leenstra S, Heimans JJ, Reijneveld JC (2007) Differential effect of surgery and radiotherapy on neurocognitive functioning and health-related quality of life in WHO grade I meningioma patients. J Neurooncol 84:271–278
Talacchi A, Santini B, Savazzi S, Gerosa M (2011) Cognitive effects of tumour and surgical treatment in glioma patients. J Neurooncol 103:541–549
Torres IJ, Mundt AJ, Sweeney PJ, Llanes-Macy S, Dunaway L, Castillo M, Macdonald RL (2003) A longitudinal neuropsychological study of partial brain radiation in adults with brain tumors. Neurology 60:1113–1118
Brown PD, Jensen AW, Felten SJ, Ballman KV, Schaefer PL, Jaeckle KA, Cerhan JH, Buckner JC (2006) Detrimental effects of tumor progression on cognitive function of patients with high-grade glioma. J Clin Oncol 24:5427–5433
Taphoorn MJ, Klein M (2004) Cognitive deficits in adult patients with brain tumours. Lancet Neurol 3(3):159–168
Biegler KA, Chaoul MA, Cohen L (2009) Cancer, cognitive impairment, and meditation. Acta Oncol 48(1):18–26
Locke DEC, Cerhan JH, Wu W, Malec JF, Clark MM, Rummans TA, Brown PD (2008) Cognitive rehabilitation and problem-solving to improve quality of life of patients with primary brain tumors: a pilot study. J Support Oncol 6:383–391
Hahn CA, Dunn RH, Logue PE, King JH, Edwards CL, Halperin EC (2003) Prospective study of neuropsychologic testing and quality-of-life assessment of adults with primary malignant brain tumors. Int J Radiat Oncol Biol Phys 55:992–999
Weitzner MA, Meyers CA (1997) Cognitive functioning and quality of life in malignant glioma patients: a review of the literature. Psychooncology 6:169–177
Gustaffson M, Edvardsson T, Ahlstrom G (2006) The relationship between function, quality of life and coping in patients with low-grade gliomas. Support Care Cancer 14:1205–1212
Giovagnoli AR, Silvani A, Colombo E, Boiardi A (2005) Facets and determinants of quality of life in patients with recurrent high grade glioma. J Neurol Neurosurg Psychiatry 76(4):562–568
Meyers CA, Hess KR (2003) Multifaceted end points in brain tumor clinical trials: cognitive deterioration precedes MRI progression. Neuro Oncol 5(2):89–95
Wefel JS, Witgert ME, Meyers CA (2008) Neuropsychological sequelae of non-central nervous system cancer and cancer therapy. Neuropsychol Rev 18(2):121–131
Giovagnoli AR (2012) Investigation of cognitive impairments in people with brain tumors. J Neurooncol 108(2):277–283
Triebel KL, Martin RC, Nabors LB, Marson DC (2009) Medical decision-making capacity in patients with malignant glioma. Neurology 73:2086–2092
Marson DC, Martin RC, Triebel KL, Nabors LB (2010) Capacity to consent to research participation in adults with malignant glioma. J Clin Oncol 28:3844–3850
Folstein MF, Folstein SE, Mc Hugh PR (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatry Res 12:189–198
Orsini A, Grossi D, Capitani E, Laiacona M, Papagno C, Vallar G (1987) Verbal and spatial immediate memory span: normative data from 1355 adults and 1112 children. Ital J Neurol Sci 8(6):539–548
Carlesimo GA, Caltagirone C, Gainotti G (1996) The Mental Deterioration Battery: normative data, diagnostic reliability and qualitative analyses of cognitive impairment. The Group for the Standardization of the Mental Deterioration Battery. Eur Neurol 36(6):378–384
Carlesimo GA, Buccione I, Fadda L, Graceffa A, Mauri M, Lo Russo S, Bevilacqua G, Caltagirone C (2002) Standardizzazione di due test di memoria per uso clinico: breve racconto e figura di rey. Nuova Rivista Neurol 12:1–13
Caffarra P, Vezzadini G, Dieci F, Zonato F, Venneri A (2002) Rey-Osterrieth complex figure: normative values in an Italian population sample. Neurol Sci 22:443–447
Caffarra P, Vezzadini G, Zonato F, Copelli S (1938) Venneri A (2003) A normative study of a shorter version of Raven’s progressive matrices. Neurol Sci 24(5):336–339
Apollonio I, Leone M, Isella V, Piamarta F, Consoli T, Villa ML, Forapani E, Russo A, Nichelli P (2005) The Frontal Assessment Battery (FAB): normative values in an Italian population sample. Neurol Sci 26:108–116
Giovagnoli AR, Del Pesce M, Mascheroni S, Simoncelli M, Laiacona M, Capitani E (1996) Trail making test: normative values from 287 normal adults controls. It J Neurol Sci 17:305–309
Novelli G, Papagno C, Capitani E, Laiacona M, Cappa SF, Vallar G (1986) Tre test clinici di ricerca e produzione lessicale. Taratura su soggetti normali. Archivio di Psicologia Neurologia Psichiatria 47:477–506
Correa DD (2006) Cognitive functions in brain tumor patients. Hematol Oncol Clin North Am 20:1363–1376
Dietrich J, Monje M, Wefel J, Meyers C (2008) Clinical patterns and biological correlates of cognitive dysfunction associated with cancer therapy. Oncologist 13(12):1285–1295
Heimans JJ, Reijneveld JC (2012) Factors affecting the cerebral network in brain tumor patients. J Neurooncol 108:231–237
Stern Y (2009) Cognitive reserve. Neuropsychologia 47(10):2015–2028
Steffener J, Stern Y (2012) Exploring the neural basis of cognitive reserve in aging. Biochim Biophys Acta 1822(3):467–473
Vataja R, Pohjasvaara T, Mantyla R, Ylikoski R, Leppavuori A, Leskela M, Kalska H, Hietanen M, Aronen HJ, Salonen O, Kaste M, Erkinjuntti T (2003) MRI correlates of executive disfunction in patients with ischaemic stroke. Eur J Neurol 10:625–631
Nys GMS, van Zandvoort MJE, de Kort PML, Jansen BPW, de Haan EHF, Kappelle LJ (2007) Cognitive disorders in acute stroke: prevalence and clinical determinants. Cerebrovasc Dis 23:408–416
Soussain C, Ricard D, Fike JR, Mazeron JJ, Psimaras D, Delattre JY (2009) CNS complications of radiotherapy and chemotherapy. Lancet 374:1639–1651
Argyriou AA, Assimakopoulos K, Iconomou G, Giannakopoulou F, Kalofonos HP (2010) Either called “chemobrain” or “chemofog,” the long-term chemotherapy-induced cognitive decline in cancer survivors is real. J Pain Symptom Manage 41:126–139
Mitchell T, Turton P (2011) ‘Chemobrain’: concentration and memory effects in people receiving chemotherapy–a descriptive phenomenological study. Eur J Cancer Care 20:539–548
Gong X, Schwartz PH, Linskey ME, Bota DA (2011) Neural stem/progenitors and glioma stem-like cells have differential sensitivity to chemotherapy. Neurology 76:1126–1134
Scheibel RS, Meyers CA, Levin AV (1996) Cognitive dysfunction following surgery for intracerebral glioma: influence of histopathology, lesion location, and treatment. J Neurooncol 30:61–69
Scoccianti S, Detti B, Cipressi S, Iannalfi A, Franzese C, Biti G (2012) Changes in neurocognitive functioning and quality of life in adult patients with brain tumors treated with radiotherapy. J Neurooncol 108(2):291–308
Sheline G, WaraWM Smith V (1980) Therapeutic irradiation and brain injury. Int J Radiat Oncol Biol Phys 6:1215–1228
Behin A, Delattre J-Y (2003) Neurologic sequelae of radiotherapy on the nervous system. In: Schiff D, Wen PY (eds) Cancer neurology in clinical practice. Humana Press Inc., Totowa, pp 173–191
Meyers CA, Hess KR, Yung WK, Levin VA (2000) Cognitive function as a predictor of survival in patients with recurrent malignant glioma. J Clin Oncol 18:646–650
Klein M, Postma TJ, Taphoorn MJB, Aaronson NK, Vandertop WP, Muller M, Van der Ploeg HM, Heimans JJ (2003) The prognostic value of cognitive functioning in the survival of patients with high-grade glioma. Neurology 61(12):1796–1799
Brown PD, Buckner JC, O’Fallon JR, Iturria NL, O’Neill BP, Brown CA, Scheithauer BW, Dinapoli RP, Arusell RM, Curran WJ, Abrams R, Shaw EG (2004) Importance of baseline mini-mental state examination as a prognostic factor for patients with low-grade glioma. Int J Radiat Oncol Biol Phys 59(1):117–125
Armstrong CL, Goldstein B, Shera D, Ledakis GE, Tallent EM (2003) The predictive value of longitudinal neuropsychological assessment in the early detection of brain tumor recurrence. Cancer 97:649–656
Meyers CA, Rock EP, Fine HA (2012) Refining endpoints in brain tumor clinical trials. J Neurooncol 108:227–230
Klein M, Duffau H, De Witt Hamer PC (2012) Cognition and resective surgery for diffuse infiltrative glioma: an overview. J Neurooncol 108:309–318
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zucchella, C., Bartolo, M., Di Lorenzo, C. et al. Cognitive impairment in primary brain tumors outpatients: a prospective cross-sectional survey. J Neurooncol 112, 455–460 (2013). https://doi.org/10.1007/s11060-013-1076-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-013-1076-8