Abstract
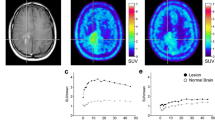
Amino acid transport and protein synthesis are important steps of tumor growth. We investigated the time course of tumor metabolism in low-grade gliomas (LGG) during temozolomide chemotherapy, and compared metabolic responses as measured with positron emission tomography (PET) with volume responses as revealed by magnetic resonance imaging (MR). A homogeneous population of 11 patients with progressive non-enhancing LGG was prospectively studied. Imaging was done at 6-months intervals starting six months, and in a second series starting three months after treatment initiation. F-18 fluoro-ethyl-l-tyrosine (FET) uptake was quantified with PET as metabolically active tumor volume, and was compared with the tumor volume on MR. Response was defined as ≥10% reduction of the initial tumor volume. Eight patients showed metabolic responses. Already 3 months after start of chemotherapy the active FET volumes decreased in 2 patients to a mean of 44% from baseline. First MR volume responses were noted at 6 months. Responders showed a volume reduction to 31 ± 23% (mean ± SD) from baseline for FET, and to 73 ± 26% for MR. The time to maximal volume reduction was 8.0 ± 4.4 months for FET, and 15.0 ± 3.0 months for MR. The initial metabolic response correlated with the best volume response on MR (Spearman Rank P = 0.011). Deactivation of amino acid transport represents an early indicator of chemotherapy response in LGG. Response assessment based on MR only has to be reconsidered. The time window obtained from PET may assist for individual treatment decisions in LGG patients.




Similar content being viewed by others
References
Schiff D, Brown PD, Giannini C (2007) Outcome in adult low-grade glioma: the impact of prognostic factors and treatment. Neurology 69:1366–1373. doi:10.1212/01.wnl.0000277271.47601.a1
Brada M, Viviers L, Abson C et al (2003) Phase II study of primary temozolomide chemotherapy in patients with WHO grade II gliomas. Ann Oncol 14:1715–1721. doi:10.1093/annonc/mdg371
Hoang-Xuan K, Capelle L, Kujas M et al (2004) Temozolomide as initial treatment for adults with low-grade oligodendrogliomas or oligoastrocytomas and correlation with chromosome 1p deletions. J Clin Oncol 22:3133–3138. doi:10.1200/JCO.2004.10.169
Kaloshi G, Benouaich-Amiel A, Diakite F et al (2007) Temozolomide for low-grade gliomas: predictive impact of 1p/19q loss on response and outcome. Neurology 68:1831–1836. doi:10.1212/01.wnl.0000262034.26310.a2
Pouratian N, Gasco J, Sherman JH et al (2007) Toxicity and efficacy of protracted low dose temozolomide for the treatment of low grade gliomas. J Neurooncol 82:281–288. doi:10.1007/s11060-006-9280-4
Quinn JA, Reardon DA, Friedman AH et al (2003) Phase II trial of temozolomide in patients with progressive low-grade glioma. J Clin Oncol 21:646–651. doi:10.1200/JCO.2003.01.009
Macdonald DR, Cascino TL, Schold SC Jr et al (1990) Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 8:1277–1280
Miyagawa T, Oku T, Uehara H et al (1998) “Facilitated” amino acid transport is upregulated in brain tumors. J Cereb Blood Flow Metab 18:500–509. doi:10.1097/00004647-199805000-00005
Kleihues P, Louis DN, Scheithauer BW et al (2002) The WHO classification of tumors of the nervous system. J Neuropathol Exp Neurol 61:215–225
EORTC (European Organization of Research and Treatment of Cancer) Intergroup study 22033-26033: Primary chemotherapy with temozolomide vs. radiotherapy in patients with low grade gliomas after stratification for genetic 1p loss: a phase III study (http://groups.eortc.be/brain/html/trials.html)
Weckesser M, Langen KJ, Rickert CH et al (2005) O-(2-[18F]fluorethyl)-l-tyrosine PET in the clinical evaluation of primary brain tumours. Eur J Nucl Med Mol Imaging 32:422–429. doi:10.1007/s00259-004-1705-8
Mikolajczyk K, Szabatin M, Rudnicki P et al (1998) A JAVA environment for medical image data analysis: initial application for brain PET quantitation. Med Inform (Lond) 23:207–214. doi:10.3109/14639239809001400
Wyss MT, Hofer S, Hefti M et al (2007) Spatial heterogeneity of low-grade gliomas at the capillary level: a PET study on tumor blood flow and amino acid uptake. J Nucl Med 48:1047–1052. doi:10.2967/jnumed.106.038489
Husstedt HW, Sickert M, Kostler H et al (2000) Diagnostic value of the fast-FLAIR sequence in MR imaging of intracranial tumors. Eur Radiol 10:745–752. doi:10.1007/s003300050997
Tang BN, Sadeghi N, Branle F et al (2005) Semi-quantification of methionine uptake and flair signal for the evaluation of chemotherapy in low-grade oligodendroglioma. J Neurooncol 71:161–168. doi:10.1007/s11060-004-9654-4
Herholz K, Holzer T, Bauer B et al (1998) 11C-methionine PET for differential diagnosis of low-grade gliomas. Neurology 50:1316–1322
Mandonnet E, Delattre JY, Tanguy ML et al (2003) Continuous growth of mean tumor diameter in a subset of grade II gliomas. Ann Neurol 53:524–528. doi:10.1002/ana.10528
Ricard D, Kaloshi G, Amiel-Benouaich A et al (2007) Dynamic history of low-grade gliomas before and after temozolomide treatment. Ann Neurol 61:484–490. doi:10.1002/ana.21125
Ribom D, Schoenmaekers M, Engler H et al (2005) Evaluation of 11C-methionine PET as a surrogate endpoint after treatment of grade 2 gliomas. J Neurooncol 71:325–332. doi:10.1007/s11060-004-2031-5
Nuutinen J, Sonninen P, Lehikoinen P et al (2000) Radiotherapy treatment planning and long-term follow-up with [(11)C]methionine PET in patients with low-grade astrocytoma. Int J Radiat Oncol Biol Phys 48:43–52. doi:10.1016/S0360-3016(00)00604-0
Voges J, Herholz K, Holzer T et al (1997) 11C-methionine and 18F-2-fluorodeoxyglucose positron emission tomography: a tool for diagnosis of cerebral glioma and monitoring after brachytherapy with 125I seeds. Stereotact Funct Neurosurg 69:129–135. doi:10.1159/000099864
Asano S, Kameyama M, Oura A et al (2007) l-type amino acid transporter-1 expressed in human astrocytomas, U343MGa. Biol Pharm Bull 30:415–422. doi:10.1248/bpb.30.415
Nawashiro H, Otani N, Shinomiya N et al (2006) l-type amino acid transporter 1 as a potential molecular target in human astrocytic tumors. Int J Cancer 119:484–492. doi:10.1002/ijc.21866
Roelcke U, Radu E, Ametamey S et al (1996) Association of rubidium and C-methionine uptake in brain tumors measured by positron emission tomography. J Neurooncol 27:163–171. doi:10.1007/BF00177480
Wienhard K, Herholz K, Coenen HH et al (1991) Increased amino acid transport into brain tumors measured by PET of l-(2-18F)fluorotyrosine. J Nucl Med 32:1338–1346
van den Bent MJ, Kros JM (2007) Predictive and prognostic markers in neuro-oncology. J Neuropathol Exp Neurol 66:1074–1081. doi:10.1097/nen.0b013e31815c39f1
Yip S, Iafrate AJ, Louis DN (2008) Molecular diagnostic testing in malignant gliomas: a practical update on predictive markers. J Neuropathol Exp Neurol 67:1–15. doi:10.1097/nen.0b013e31815f65fb
Acknowledgements
This work was supported by grants of the Swiss Group for Clinical Cancer Research (SAKK) and by Essex Chemie AG, Luzern, Switzerland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wyss, M., Hofer, S., Bruehlmeier, M. et al. Early metabolic responses in temozolomide treated low-grade glioma patients. J Neurooncol 95, 87–93 (2009). https://doi.org/10.1007/s11060-009-9896-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-009-9896-2