Abstract
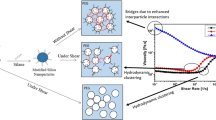
The dispersions was prepared by dispersing hydrophilic silica nanoparticles with an average diameter of 50 nm in 1-butyl-methylimidazolium tetrafluoroborate ([C4mim][BF4]), and investigated under the steady shear and oscillatory shear, respectively. Experimental results indicate that all of the dispersions present shear thinning, notable shear thickening, and shear thinning successively with increasing shear rate; the shear thickening behavior is derived from silica nanoparticle clusters and strongly controlled by silica nanoparticle content and temperature. The shear thickening fluid (STF) exhibits reversible property and transient response ability, and the time of transient response is no more than 100 ms. The conductivity of the STF is increased with increasing silica nanoparticle content, and the conductivity of 27 wt% dispersions is even two times as big as that of pure [C4mim][BF4]. The dispersions with high solid content dilate and present a phase transition changing from a liquid-like to a solid-like soft material in the shear thickening region. A theoretical model is developed to imitate the mechanism of shear thickening in the dispersions.










Similar content being viewed by others
References
Atkin R, Warr GG (2007) Structure in confined room-temperature ionic liquids. J Phys Chem C 111(13):5162–5168. doi:10.1021/jp067420g
Barnes HA (1989) Shear-thickening (“dilatancy”) in suspensions of nonaggregating solid particles dispersed in newtonian liquids. J Rheol (1978-present) 33(2):329–366. doi:10.1122/1.550017
Boersma WH, Laven J, Stein HN (1992) Viscoelastic properties of concentrated shear-thickening dispersions. J Colloid Interface Sci 149(1):10–22. doi:10.1016/0021-9797(92)90385-Y
Bossis G, Brady JF (1989) The rheology of brownian suspensions. J Chem Phys 91:1866–1874. doi:10.1063/1.457091
Brown E, Jaeger HM (2009) Dynamic jamming point for shear thickening suspensions. Phys Rev Lett 103:086001. doi:10.1103/PhysRevLett.103.086001
Cates ME, Wittmer JP, Bouchaud JP, Claudin P (1998) Jamming, force chains, and fragile matter. Phys Rev Lett 81:1841–1844. doi:10.1103/PhysRevLett.81.1841
Cheng X, McCoy JH, Israelachvili JN, Cohen I (2011) Imaging the microscopic structure of shear thinning and thickening colloidal suspensions. Science 333:1276–1279. doi:10.1126/science.1207032
Chow MK, Zukoski CF (1995) Nonequilibrium behavior of dense suspensions of uniform particles: volume fraction and size dependence of rheology and microstructure. J Rheol 39:33–59. doi:10.1122/1.550687
Decker MJ, Halbach CJ, Nam CH, Wagner NJ, Wetzel ED (2007) Stab resistance of shear thickening fluid (STF)-treated fabrics. Compos Sci Technol 67(3):565–578. doi:10.1016/j.compscitech.2006.08.007
Fall A, Huang N, Bertrand F, Ovarlez G, Bonn D (2008) Shear thickening of cornstarch suspensions as a reentrant jamming transition. Phys Rev Lett 100:018301. doi:10.1103/PhysRevLett.100.018301
Fernandez N et al. (2013) Microscopic mechanism for shear thickening of non-brownian suspensions. Phys Rev Lett 111:108301. doi:10.1103/PhysRevLett.111.108301
Foss DR, Brady JF (2000) Structure, diffusion and rheology of brownian suspensions by stokes dynamics simulation. J Fluid Mech 407:167–200. doi:10.1017/S0022112099007557
Fukushima T, Kosaka A, Ishimura Y, Yamamoto T, Takigawa T, Ishii N, Aida T (2003) Molecular ordering of organic molten salts triggered by Single-walled carbon nanotubes. Science 300:2072–2074. doi:10.1126/science.1082289
Heussinger C (2013) Shear thickening in granular suspensions: interparticle friction and dynamically correlated clusters. Phys Rev E 88(050201(R)):1–4. doi:10.1103/PhysRevE.88.050201
Hoffman RL (1972) Discontinuous and dilatant viscosity behavior in concentrated suspensions. I. Observation of a flow instability. Trans Soc Rheol 16:155–173. doi:10.1122/1.549250
Hoffman RL (1974) Discontinuous and dilatant viscosity behavior in concentrated suspensions. II. Theory and experimental tests. J Colloid Interface Sci 46:491–506. doi:10.1016/0021-9797(74)90059-9
Hoffman RL (1998) Explanations for the cause of shear thickening in concentrated colloidal suspensions. J Rheol 42:111–123. doi:10.1122/1.550884
Kalman DP, Schein JB, Houghton JM, Laufer CN, Wetzel ED, Wagner NJ (2007) Polymer dispersion based shear thickening fluid-fabrics for protective applications. In: Proceedings of SAMPE, Baltimore, pp 3–7
Kawauchi T, Kumaki J, Okoshi K, Yashima E (2005) Stereocomplex formation of isotactic and syndiotactic poly(methyl methacrylate) in ionic liquids leading to thermoreversible ion gels. Macromolecules 38:9155–9160. doi:10.1021/ma0517594
Lee YS, Wagner NJ (2003) Dynamic properties of shear thickening colloidal suspensions. Rheol Acta 42(3):199–208. doi:10.1007/s00397-002-0290-7
Lee YS, Wetzel ED, Wagner NJ (2003) The ballistic impact characteristics of Kevlar® woven fabrics impregnated with a colloidal shear thickening fluid. J Mater Sci 38(13):2825–2833. doi:10.1023/A:1024424200221
Leunissen ME, Christova CG et al (2005) Ionic colloidal crystals of oppositely charged particles. Nature 437:235–240. doi:10.1038/nature03946
Liu Y, Zhang Y, Wu G, Hu J (2006) Coexistence of liquid and solid phases of Bmim-PF6 ionic liquid on mica surfaces at room temperature. J Am Chem Soc 128:7456–7457. doi:10.1021/ja062685u
Lootens D, van Damme H, Hémar Y, Hébraud P (2005) Dilatant flow of concentrated suspensions of rough particles. Phys Rev Lett 95:268302. doi:10.1103/PhysRevLett.95.268302
MacFarlane DR, Pringle JM, Howlett PC, Forsyth M (2010) Ionic liquids and reactions at the electrochemical interface. Phys Chem Chem Phys 12:1659–1669. doi:10.1039/B923053J
Moosavi M, Daneshvar A (2014) Investigation of the rheological properties of two imidazolium-based ionic liquids. J Mol Liq 190:59–67. doi:10.1016/j.molliq.2013.10.024
Nathan CC, Lauren BP et al (2013) Shear thickening of corn starch suspensions: does concentration matter? J Colloid Interface Sci 396:83–89. doi:10.1016/j.jcis.2013.01.024
Nishida T, Tashiro Y, Yamamoto M (2003) Physical and electrochemical properties of 1-alkyl-3-methylimidazolium tetrafluoroborate for electrolyte. J Fluor Chem 120:135–141. doi:10.1016/S0022-1139(02)00322-6
Novak J, Britton MM (2013) Magnetic resonance imaging of the rheology of ionic liquid colloidal suspensions. Soft Matter 9:2730–2737. doi:10.1039/C3SM27409H
Raghavan SR, Khan SA (1997) Shear-thickening response of fumed silica suspensions under steady and oscillatory shear. J Colloid Interface Sci 185:57–67. doi:10.1006/jcis.1996.4581
Seto R, Mari R, Morris JF, Denn MM (2013) Discontinuous shear thickening of frictional hard-sphere suspensions. Phys Rev Lett 111:218301. doi:10.1103/PhysRevLett.111.218301
Srivastava A, Majumdar A, Butola BS (2011) Improving the impact resistance performance of Kevlar fabrics using silica based shear thickening fluid. Mater Sci Eng, A 529:224–229. doi:10.1016/j.msea.2011.09.021
Stokes JR, Frith WJ (2008) Rheology of gelling and yielding soft matter systems. Soft Matter 4:1133–1140. doi:10.1039/B719677F
Trappe V, Prasad V, Cipelletti L, Segre PN, Weitz DA (2001) Similarities between protein folding and granular jamming. Nature 411:772–775. doi:10.1038/ncomms2177
Ueno K, Watanabe M (2011) From colloidal stability in ionic liquids to advanced soft materials using unique media. Langmuir 27:9105–9115. doi:10.1021/la103942f
Ueno K, Hata K, Katakabe T, Kondoh M, Watanabe M (2008) Nanocomposite Ion Gels based on silica nanoparticles and an ionic liquid: ionic transport, viscoelastic properties, and microstructure. J Phys Chem B 112:9013–9019. doi:10.1021/jp8029117
Ueno K, Imaizumi S, Hata K, Watanabe M (2009) Colloidal interaction in ionic liquids: effects of ionic structures and surface chemistry on rheology of silica colloidal dispersions. Langmuir 25:825–831. doi:10.1021/la803124m
Ueno K, Sano Y, Inaba A, Kondoh M, Watanabe M (2010a) Soft glassy colloidal Arrays in an ionic liquid: colloidal glass transition, ionic transport, and structural color in relation to microstructure. J Phys Chem B 114:13095–13103. doi:10.1021/jp106872w
Ueno K, Tokuda H, Watanabe M (2010b) Ionicity in ionic liquids: correlation with ionic structure and physicochemical properties. Phys Chem Chem Phys 12:1649–1658. doi:10.1039/B921462N
Ueno K, Inaba A, Ueki T, Kondoh M, Watanabe M (2010c) Thermosensitive, soft glassy and structural colored colloidal array in ionic Liquid: colloidal glass to gel transition. Langmuir 26(23):18031–18038. doi:10.1021/la103716q
Wagner NJ, Brady JF (2009) Shear thickening in colloidal dispersions. Phys Today 10:27–32. doi:10.1063/1.3248476
Wakeham D, Hayes R, Warr GG, Atkin R (2009) Influence of temperature and molecular structure on ionic liquid solvation layers. J Phys Chem B 113(17):5961–5966. doi:10.1021/jp900815q
Wang P, Zakeeruddin SM, Comte P, Charvet R (2003) Enhance the performance of dye-sensitized solar cells by co-grafting amphiphilic sensitizer and hexadecylmalonic acid on TiO2 nanocrystals. J Phys Chem B 107:14336–14341. doi:10.1021/jp0365965
Welton T (1999) Room-temperature ionic liquids: solvents for synthesis and catalysis. Chem Rev 99:2071–2084. doi:10.1021/cr980032t
Wetzel ED, Lee YS, Egres RG, Kirkwood KM, Kirkwood JE, Wagner NJ (2004) The effect of rheological parameters on the ballistic properties of shear thickening fluid (STF)–Kevlar composites. AIP Conf Proc 712:288. doi:10.1063/1.1766538
Wittmar A, Ruiz-Abad D, Ulbricht M (2012) Dispersions of silica nanoparticles in ionic liquids investigated with advanced rheology. J Nanopart Res 14:651. doi:10.1007/s11051-011-0651-1
Wyart M, Cates ME (2014) Discontinuous shear thickening without inertia in dense non-brownian suspensions. Phys Rev Lett 112:089302. doi:10.1103/PhysRevLett.112.098302
Acknowledgments
We gratefully acknowledge the Lanzhou Institute of Chemical Physical for providing the 1-butyl-3-methylimidazolium tetrafluoroborate ([C4mim][BF4]) used in this work.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing financial interest.
Rights and permissions
About this article
Cite this article
Qin, J., Zhang, G., Shi, X. et al. Study of a shear thickening fluid: the dispersions of silica nanoparticles in 1-butyl-3-methylimidazolium tetrafluoroborate. J Nanopart Res 17, 333 (2015). https://doi.org/10.1007/s11051-015-3144-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-015-3144-9