Abstract
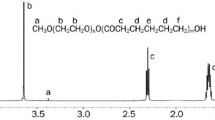
A new oral delivery system, polybutylcyanoacrylate nanoparticles (PBCNs), was introduced to improve the oral bioavailability of curcumin (CUR), a poorly soluble drug. The formulation was optimized by orthogonal design and the optimal PBCNs loading CUR exhibited a spherical shape under transmission electron microscopy with a range of 40–400 nm. Physicochemical state of CUR in PBCN was investigated by X-ray diffraction and the possible structure changes occurring in CUR after conjugating with polybutylcyanoacrylate were studied with FTIR. The results indicated that CUR in PBCN was in a non-crystalline state and CUR was encapsulated in PBCN without chemical reaction. The oral pharmacokinetic study was conducted in rats and the relative bioavailability of CUR encapsulated PBCNs to the crude CUR was more than 800%. The in situ absorption experiment in rat intestine indicated the absorption was first order with passive diffusion mechanism. The absorption results in various segments of intestine showed that the main absorption sites were ileum and colon. It can be concluded that PBCNs as an oral carrier can significantly improve the oral absorption of a poorly soluble drug.









Similar content being viewed by others
References
Alyautdin RN, Petrov VE, Langer K, Berthold A, Kharkevich DA, Kreuter J (1997) Delivery of loperamide across the blood–brain barrier with polysorbate 80-coated polybutylcyanoacrylate nanoparticles. Pharm Res 14:325–328. doi:10.1023/A:1012098005098
Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB (2007) Bioavailability of curcumin: problems and promises. Mol Pharm 4:807–818. doi:10.1021/mp700113r
Anand P, Nair HB, Sung B, Kunnumakkara AB, Yadav VR, Tekmal RR, Aggarwal BB (2010) Design of curcumin-loaded PLGA nanoparticles formulation with enhanced cellular uptake, and increased bioactivity in vitro and superior bioavailability in vivo. Biochem Pharmacol 79:330–338. doi:10.1016/j.bcp.2009.09.003
Behan N, Birkinshaw C (2000) Mechanism of polymerization of butylcyanoacrylate in aqueous dispersions. Macromol Rapid Commun 21:884–886. doi:10.1002/1521-3927(20000801
Behan N, Birkinshaw C, Clarke N (2001) Poly n-butyl cyanoacrylate nanoparticles: a mechanistic study of polymerisation and particle formation. Biomaterials 22:1335–1344. doi:10.1016/S0142-9612(00)00286-6
Chen RD, Ren F, Li GF, Liu SJ (2010) Preparation of paclitaxel-loaded polybutylanaocrylate nanoparticle. J South Med Univ 30:763–766
Cirri M, Muraa P, Corvi MP (2007) Liquid spray formulations of xibornol by using self-microemulsifying drug delivery systems. Int J Pharm 340:84–91. doi:10.1016/j.ijpharm.2007.03.021
Cui J, Yu B, Zhao Y, Zhu WW, Li HL, Lou HX, Zhai GX (2009) Enhancement of oral absorption of curcumin by self-microemulsifying drug delivery systems. Int J Pharm 371:148–155. doi:10.1016/j.ijpharm.2008.12.009
David W, Fryb J, White C, Goldman ID (1978) Rapid separation of low molecular weight solutes from liposomes without dilution. Anal Biochem 90:809–815. doi:10.1016/0003-2697(78)90172-0
Dembri A, Montisci MJ, Gantier JC, Chacun H, Ponchel G (2001) Targeting of 30-Azido 30-deoxythymidine (AZT)-loaded poly(isohexylcyanoacrylate) nanospheres to the gastrointestinal mucosa and associated lymphoid tissues. Pharm Res 18:467–474. doi:10.1023/A:1011050209986
Douglas SJ, Illum L, Davisand SS, Kreuter J (1984) Particle size and size distribution of poly(butyl-2-cyanoacrylate) nanoparticles. II. Influence of physicochemical factors. J Colloid Interface Sci 101:149–157. doi:10.1016/0021-9797(84)90015-8
Duchêne D, Ponchel G (1997) Bioadhesion of solid oral dosage forms, why and how? Eur J Pharm Biopharm 44:15–23. doi:10.1016/S0939-6411(97)00097-0
Duro R, Alvarez C, Martı′nez-Pacheco R, Go′mez-Amoza JL, Concheiro A, Souto C (1998) The adsorption of cellulose ethers in aqueous suspensions of pyrantel pamoate: effects on zeta potential and stability. Eur J Pharm Biopharm 45:181–188. doi:10.1016/S0939-6411(97)00103-3
Egan ME, Pearson M, Weiner SA, Rajendran V, Rubin D, Glockner-Pagel J, Canny S, Du K, Lukacs GL, Caplan MJ (2004) Curcumin, a major constituent of turmeric, corrects cystic fibrosis defects. Science 304:600–602. doi:10.1126/science.1093941
Eldridge JH (1990) Controlled release in the gut-associated lymphoid tissues. 1. Orally administered biodegradable microspheres target the Peyer’s patches. J Control Release 11:205–214. doi:10.1016/0168-3659(90)90133-E
Fawaz F, Guyot M, Lagueny AM, Devissaguet JP (1997) Ciprofloxacin-loaded poly (isobutylcyanoacrylate) nanoparticles: preparation and characterization. Int J Pharm 154:191–203
Gao XF, Deng YJ, Cao JN, Wang XM (2009a) Minicolumn centrifugation—HPLC determination of entrapment efficiency for liposomal formulation of altretamine. Chin J Pharm Anal 29:247–250
Gao Y, Wang YQ, Ma YK, Yu AH, Cai FQ, Shao W, Zhai GX (2009b) Formulation optimization and in situ absorption in rat intestinal tract of quercetin-loaded microemulsion. Colloids Surf B 71:306–314. doi:10.1016/j.colsurfb.2009.03.005
Gulyaev AE, Gelperina SE, Skidan IN, Antropov AS, Kivman GY, Kreuter J (1999) Significant transport of doxorubicin into the brain with polysorbate 80 coated nanoparticles. Pharm Res 16:1564–1570. doi:10.1023/A:1018983904537
He WL, Jiang XH, Zhang ZR (2008) Preparation and evaluation of poly-butylcyanoacrylate nanoparticles for oral delivery of thymopentin. J Pharm Sci 97:2250–2259. doi:10.1002/jps.21148
Hu YQ, Zheng LY, Qian CQ, Yu WH (1996) Absorption of phenol red from rat intestine. J Chin Pharm Univ 27:355–359
Huang CY, Lee YD (2006) Core–shell type of nanoparticles composed of poly [(n-butyl cyanoacrylate)-co-(2-octylcyanoacrylate)] copolymers for drug delivery application: synthesis, characterization and in vitro degradation. Int J Pharm 325:132–139. doi:10.1016/j.ijpharm.2006.06.008
Huang CY, Chen CM, Lee YD (2007) Synthesis of high loading and encapsulation efficient paclitaxel-loaded poly (n-butyl cyanoacrylate) nanoparticles via miniemulsion. Int J Pharm 338:267–275. doi:10.1016/j.ijpharm.2007.01.052
Huo DJ, Deng SH, Li LB, Ji JB (2005) Studies on the poly (lactic-co-glycolic) acid microspheres of cisplatin for lung-targeting. Int J Pharm 289:63–67. doi:10.1016/j.ijpharm.2004.10.017
Hussain N, Jaitley V, Florence AT (2001) Recent advances in the understanding of uptake of microparticulates across the gastrointestinal lymphatics. Adv Drug Deliv Rev 50:107–142. doi:10.1016/S0169-409X(01)00152-1
Johnson JJ, Mukhtar H (2007) Curcumin for chemoprevention of colon cancer. Cancer Lett 255:170–181. doi:10.1016/j.canlet.2007.03.005
Kakkar V, Singh S, Singla D, Kaur IP (2010) Exploring solid lipid nanoparticles to enhance the oral bioavailability of curcumin. Mol Nutr Food Res 54:1–9. doi:10.1002/mnfr.201000310
Lao C, Ruffin M, Normolle D, Heath D, Murray S, Bailey J (2006) Dose escalation of a curcuminoid formulation. BMC Complement Altern Med 6:10–15. doi:10.1186/1472-6882-6-10
Liu J, Zhu J, Du Z, Qin B (2005) Preparation and pharmacokinetic evaluation of Tashinone IIA solid lipid nanoparticles. Drug Dev Ind Pharm 31:551–556. doi:10.1080/03639040500214761
Lv QZ, Yu AH, Xi YW, Li HL, Song ZM, Cui J, Cao FL, Zhai GX (2009) Development and evaluation of penciclovir-loaded solid lipid nanoparticles for topical delivery. Int J Pharm 372:191–198. doi:10.1016/j.ijpharm.2009.01.014
Masaoka Y, Tanaka Y, Kataoka M, Sakuma S, Yamashita S (2006) Site of drug absorption after oral administration: assessment of membrane permeability and luminal concentration of drugs in each segment of gastrointestinal tract. Eur J Pharm Sci 29:240–250. doi:10.1016/j.ejps.2006.06.004
Mulik R, Mahadika K, Paradkarb A (2009) Development of curcuminoids loaded poly (butyl) cyanoacrylate nanoparticles: physicochemical characterization and stability study. Eur Pharm Sci 37:395–404. doi:10.1016/j.ejps.2009.03.009
Müller RH, Mäder K, Gohla S (2000) Solid lipid nanoparticles (SLN) for controlled drug delivery-are view of the state of the art. Eur J Pharm Biopharm 50:161–177. doi:10.1016/S0939-6411(00)00087-4
Murthy RSR, Reddy LH (2007) Poly (alkyl cyanocrylate) nanoparticles for delivery of anti-cancer drugs. Nanotechnology 251–288. doi:10.1201/9781420006636.ch15
Onoue S, Takahashi H, Kawabata Y, Seto Y, Hatanaka J, Timmermann B, Yamada S (2009) Formulation design and photochemical studies on nanocrystal solid dispersion of curcumin with improved oral bioavailability. J Pharm Sci 99:1871–1881. doi:10.1002/jps.21964
Pan MH, Huang TM, Lin JK (1999) Biotransformation of curcumin through reduction and glucuronidation in mice. Drug Metab Dispos 27:486–494. doi:0090-9556/99/2701-0486-494
Pappo J, Ermak TH (1989) Uptake and translocation of fluorescent latex particles by rabbit Peyer’s patch follicle epithelium: a quantitative model for Mcell uptake. Clin Exp Immunol 76:144–148
Shaikha J, Ankolab DD, Beniwal V, Singha D, RaviKumar MNV (2009) Nanoparticle encapsulation improves oral bioavailability of curcumin by at least 9-fold when compared to curcumin administered with piperine as absorption enhancer. Eur J Pharm Sci 37:223–230. doi:10.1016/j.ejps.2009.02.019
Shankar T, Shantha N, Ramesh H, Murthy I, Murthy V (1980) Toxicity studies on turmeric (Curcuma longa): acute toxicity studies in rats, guinea pigs and monkeys. Indian J Exp Biol 18:73–75
Shantha KTR, Chawla S, Nachaegari SK, Singh SK, Srinivas NR (2005) Validated HPLC analytical method with programmed wavelength UV detection for simultaneous determination of DRF-4367 and Phenol red in rat in situ intestinal perfusion study. J Pharm Biomed Anal 38:173–179. doi:10.1016/j.jpba.2004.11.063
Sharma RA, Steward WP, Gescher AJ (2007) Pharmacokinetics and pharmacodynamics of curcumin. Adv Exp Med Biol 595:453–470. doi:10.1007/978-0-387-46401-5_20
Simeonova M, Ivanova G, Enchev V, Markova N, Kamburov M, Petkov C, Devery A, Connor R, Brougham D (2009) Physicochemical characterization and in vitro behavior of daunorubicin-loaded poly(butylcyanoacrylate) nanoparticles. Acta Biomater 5:2109–2121
Singh B, Mehta G, Kumar R, Bhatia A, Ahuja N, Katare OP (2005) Design, development and optimization of nimesulide-loaded liposomal systems for topical application. Curr Drug Deliv 2:143–153
Sun M, Gao Y, Guo CY, Cao FL, Song ZM, Xi YW, Yu AH, Li AG, Zhai GX (2010) Enhancement of transport of curcumin to brain in mice by poly(n-butylcyanoacrylate) nanoparticle. J Nanopart Res 12:3111–3122. doi:10.1007/s11051-010-9907-4
Takahashi M, Uechi S, Takara K, Asikin Y, Wada K (2009) Evaluation of an oral carrier system in rats: bioavailability and antioxidant properties of liposome-encapsulated curcumin. J Agric Food Chem 57:9141–9146. doi:10.1021/jf9013923
Tonnesen HH, Masson M, Loftsson T (2002) Studies of curcumin and curcuminoids. XXVII. Cyclodextrin complexation: solubility, chemical and photochemical stability. Int J Pharm 244:127–135. doi:10.1016/S0378-5173(02)00323-X
Venkatesan N, Uchino K, Amagase K, Ito Y, Shibata N, Takada K (2006) Gastrointestinal patch system for the delivery of erythropoietin. J Control Release 111:19–26. doi:10.1016/j.jconrel.2005.11.009
Wang YJ, Pan MH, Cheng AL, Lin LI, Ho YS, Hsieh CY, Lin JK (1997) Stability of curcumin in buffer solutions and characterization of its degradation products. J Pharm Biomed 15:1867–1876. doi:10.1016/S0731-7085(96)02024-9
Yang XX, Chen JH, Guo D (2005) Study of biocompatible of polybutylcyanoacrylate nanoparticles. J First Mil Med Univ 25:1261–1263. doi:http://www.j-smu.com/pdf2/200510/2005101261.pdf
Yang KY, Lin LC, Tseng TY, Wang SC, Tsai TH (2007) Oral bioavailability of curcumin in rat and the herbal analysis from Curcuma longa by LC-MS/MS. J Chromatogr B Anal Technol Biomed Life Sci 853:183–189. doi:10.1016/j.jchromb.2007.03.010
Ye JS, Wang Q, Zhou XF, Zhang Na (2008) Injectable actarit-loaded solid lipid nanoparticles as passive targeting therapeutic agents for rheumatoid arthritis. Int J Pharm 319:162–168. doi:10.1016/j.ijpharm.2007.10.014
Yuan H, Chen J, Du YZ, Hu FQ, Zeng S, Zhao HL (2007) Studies on oral absorption of stearic acid SLN by a novel fluorometricmethod. Colloids Surf B Biointerfaces 58:157–164. doi:10.1016/j.colsurfb.2007.03.002
Zakeri-Milania P, Valizadeha H, Tajerzadehc H, Azarmia Y, Islambolchilara Z, Barzegara S, Barzegar-Jalalia M (2007) Predicting human intestinal permeability using single-pass intestinal perfusion in rat. J Pharm Pharm Sci 10:368–379
Zhu WW, Yu AH, Wang WH, Dong RQ, Wu J, Zhai GX (2008) Formulation design of microemulsion for dermal delivery of penciclovir. Int J Pharm 360:184–190. doi:10.1016/j.ijpharm.2008.04.008
Acknowledgment
This work is partly supported by the National Natural Science Foundation of China (No. 30973646).
Author information
Authors and Affiliations
Corresponding author
Additional information
Min Sun and Lixia Zhao contributed equally to this work.
Rights and permissions
About this article
Cite this article
Sun, M., Zhao, L., Guo, C. et al. Evaluation of an oral carrier system in rats: bioavailability and gastrointestinal absorption properties of curcumin encapsulated PBCA nanoparticles. J Nanopart Res 14, 705 (2012). https://doi.org/10.1007/s11051-011-0705-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-011-0705-4