Abstract
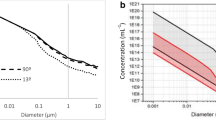
Before commencing any nanotoxicological study, it is imperative to know the state of the nanoparticles to be used and in particular their size and size distribution in the appropriate test media is particularly important. Particles satisfying standards can be commercially purchased; however, these invariably cannot be used directly and need to be dispersed into the relevant biological media. Often such changes in the environment or ionic strength, or a change in the particle concentration, results in some aggregation or a shift in the particle size distribution. Such unexpected aggregation, dissolution or plating out, if unaccounted for, can have a significant effect on the available nanoparticle dose and on interpretation of any results obtained thereafter. Here, we demonstrate the application of characterisation instrumentation that sizes nanoparticles based on their Brownian motion in suspension. Unlike classical light-scattering techniques, the nanoparticle tracking and analysis (NTA) technique allows nanoparticles to be sized in suspension on a particle-by-particle basis allowing higher resolution and therefore better understanding of aggregation than ensemble methods (such as dynamic light scattering (DLS) and differential centrifugation sedimentation (DCS)). Results will be presented from gold (standard) nanoparticles in biologically relevant media that emphasise the importance of characterisation of the nanoparticle dispersion. It will be shown how the NTA technique can be extended to multi-parameter analysis, allowing for characterization of particle size and light scattering intensity on an individual basis. This multi-parameter measurement capability allows sub-populations of nanoparticles with varying characteristics to be resolved in a complex mixture. Changes in one or more of such properties can be followed both in real time and in situ.






Similar content being viewed by others
References
Cedervall T, Lynch I, Lindman S, Berggård T, Thulin E, Nilsson H, Dawson KA, Linse S (2007a) Understanding the nanoparticle protein corona using methods to quantify exchange rates and affinities of proteins for nanoparticles. Proc Natl Acad Sci USA 104:2050–2055
Cedervall T, Lynch I, Foy M, Berggård T, Donelly S, Cagney G, Linse S, Dawson KA (2007b) Detailed identification of plasma proteins absorbed to copolymer nanoparticles. Angew Che Intl Ed 46:5754–5756
Chithrani BD, Chan WC (2007) Elucidating the mechanism of cellular uptake and removal of protein-coated gold nanoparticles of different sizes and shapes. Nano Lett 7:1542–1550
Clift MJ, Rothen-Rutishauser B, Brown DM, Duffin R, Donaldson K, Proudfoot L, Guy K, Stone V (2008) The impact of different nanoparticle surface chemistry and size on uptake and toxicity in a murine macrophage cell line. Toxicol Appl Pharmacol 232:418–427
Hackley V (2008) Enabling risk assessment, benchmarking, calibration and method validation, NanoImpactNet, Dublin 2008
Lundqvist M, Stigler J, Cedervall T, Elia G, Lynch I, Dawson K (2008) Nanoparticle size and surface properties determine the protein corona with possible implications for biological impacts. Proc Natl Acad Sci USA 105:14265–14270
Malloy A, Carr B (2006) Nanoparticle tracking analysis—the Halo ™ system. Part Part Syst Charact 23:197–204
National Institute of Standards & Technology (2007) Report of investigation, Reference material 8013, Gold nanoparticles, Nominal 60 nm diameter 13/12/07
Oberdorster G, Sharp Z, Atudorei V, Elder A, Gelein R, Kreyling W, Cox C (2004) Translocation of inhaled ultrafine particles to the brain. Inhal Toxicol 16:437–445
Royal Society of Chemistry and Royal Academy of Engineering (2005) Nanoscience and Nanotechnologies: opportunities and uncertainties. Royal Society, London
Saveyn H, De Baets B, Hole P, Smith J, Van der Meeren P (2008) Accurate particle size distribution determination by nanoparticle tracking analysis based on 2-D Brownian dynamics simulation. In Proceedings of the Particulate systems analysis, Stratford, 2008
Acknowledgements
Funding is gratefully acknowledged from EU FP6 project NanoInteract, NMP4-CT-2006-033231 (IL, KD), and SFI Research Frontiers Projects, PHY0033 and CHP031 (DW, IM-B).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Montes-Burgos, I., Walczyk, D., Hole, P. et al. Characterisation of nanoparticle size and state prior to nanotoxicological studies. J Nanopart Res 12, 47–53 (2010). https://doi.org/10.1007/s11051-009-9774-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-009-9774-z