Abstract
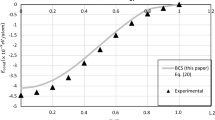
A self-consistent statistical method is used to describe size effects on melting of free nanocrystals. The melting transition is assumed to be directly related to evolution of high-temperature instability of the phonon subsystem of the crystal, caused by strong anharmonicity of atomic vibrations. We show that depression of the melting temperature of small free particles is mainly due to presence of surface atoms which are bound to smaller numbers of atoms than those of the interior. The melting temperatures of spherical nanocrystals of Ar and Au were calculated as functions of the inverse of their radii and compared with experimental and molecular dynamics data.




Similar content being viewed by others
References
Alavi S, Thompson DL (2006) Molecular dynamics simulation of the melting of aluminum nanoparticles. J Phys Chem A 110:1518–1523
Beaglehole D (1991) Surface melting of small particles, and the effects of surface impurities. J Cryst Growth 112:663–669
Ben David T, Lereah Y, Deutscher G, Kofman R, Cheyssac P (1995) Solid–liquid trtansition in ultra-fine lead particles. Phil Mag A 71:1135–1143
Böttger H (1983) Principles of the theory of lattice dynamics. Academie-Verlag, Berlin
Buffat Ph, Borel J-P (1976) Size effect on the melting temperature of gold particles. Phys Rev A 13:2287–2298
Celestini F, Pellenq RJ-M, Bordarier P, Rousseau B (1996) Melting of Lennard-Jones clusters in confined geometries. Z Phys D 37:49–53
Choquard PF (1967) The anharmonic crystal. Benjamin, New York
Couchman PR (1979) The Lindemann hypothesis and the size-dependence of melting temperature. II. Phil Mag A 40:637–643
Couchman PR, Jesser WA (1977) Thermodynamic theory of size dependence of melting temperature in metals. Nature (London) 269:481–483
Couchman PR, Ryan CL (1978) The Lindemann hypothesis and the size-dependence of melting temperature. Phil Mag A 37:369–373
Delogu F (2005) Structural and energetic properties of unsupported Cu nanoparticles from room temperature to the melting point: molecular dynamics simulations. Phys Rev B 72:205418
Ercolessi F, Andreoni W, Tosatti E (1991) Melting of small gold particles: mechanism and size effect. Phys Rev Lett 66:911–914
Hanszen K-J (1960) Theoretische Untersuchungen über den Schmelzpunkt kleiner Kügelchen. Ein Beitrag zur Thermodynamik der Grenzflächen. Z Phys 157:523–553
Hoshino K, Shimamura S (1979) A simple model for the melting of fine particles. Phil Mag A 40:137–141
Karasevskii AI, Lubashenko VV (2002) Binary distribution functions of atoms of simple crystals. Phys Rev B 66:054302
Karasevskii AI, Lubashenko VV (2004) Calculation of thermodynamic properties of Cu and Ag using a self-consistent statistical method. Phys Stat Sol (b) 241:1274–1280
Karasevskii AI, Lubashenko VV (2005) Role of anharmonicity of atomic vibrations in formation of vacancies in the rare gas crystals. Phys Rev B 71:012107
Kittel C (1986) Introduction to solid state physics. Wiley, New York
Lindemann FA (1910) Über die Berechnung molekularer Eigenfrequenzen. Z Phys 11:609–612
Maradudin AA, Flinn PA, Coldwell-Horsfall RA (1961) Anharmonic contributions to vibrational thermodynamical properties of solids. Part I. General formulation and application to the linear chain. Ann Phys (NY) 15:337–359
Nanda KK (1998) Size-dependent melting of small particles: a classical approach. Eur J Phys 19:471–472
Nanda KK, Sahu SN, Behera SN (2002) Liquid-drop model for the size-dependent melting of low-dimensional systems. Phys Rev A 66:013208
Nishiguchi N, Sakuma T (1981) Vibrational spectum and specific heat of fine particles. Solid State Commun 38:1073–1077
Olson EA, Efremov MYu, Zhang M, Allen LH (2005) Size-dependent melting of Bi nanoparticles. J Appl Phys 97:034304
Pawlow P (1909) Über die Abhängigkeit des Schmelzpunktes von den Oberflächenenergie eines festen Körpers. Z Phys Chem 65:1–35
Qi Y, Çağin T, Johnson WL, Goddard III WA (2001) Melting and crystallization in Ni nanoclusters: the mesoscale regime. J Chem Phys 115:385–394
Qi WH, Wang MP, Zhou M, Shen XQ, Zhang XF (2006) Modeling cohesive energy and melting temperature of nanocrystals. J Phys Chem Solids 67:851-855
Saka H, Nishikawa Y, Imura T (1988) Melting temperature of In particles embedded in an Al matrix. Phil Mag A 57:895–906
Sambles JR (1971) An electron microscope study of evaporating gold particles: the Kelvin equation for liquid gold and the lowering of the melting point of solid gold particles. Proc R Soc Lond A 324:339–351
Shandiz MA, Safaei A, Sanjabi S, Barber ZH (2007) Modeling size dependence of melting temperature of metallic nanoparticles. J Phys Chem Solids 68:1396–1399
Skripov VP, Koverda V P, Skokov V N (1981) Size effect on melting of small particles. Phys Stat Sol (a) 66:109–118
Takagi M (1954) Electron-diffraction study of liquid-solid transition of thin metal films. J Phys Soc Jpn 9:359–363
Tamura A, Nigeta K, Ichinokawa T (1982) Lattice vibrations and specific heat of a small particle. J Phys C 15:4975–4991
Wautelet M (1990) Size effect on the melting (or disordering) temperature of small particles. Solid State Commun 74:1237–1239
Yang CC, Li S (2007) Investigation of cohesive energy effects on size-dependent physical and chemical properties of nanocrystals. Phys Rev B 75:165413
Zhang L, Jin ZH, Zhang LH, Sui ML, Lu K (2000a) Superheating of confined Pb thin films. Phys Rev Lett 85:1484–1487
Zhang Z, Li JC, Jiang Q J (2000b) Modelling for size-dependent and dimension-dependent melting of nanocrystals. Phys D 33:2653–2656
Acknowledgements
This study was supported in part by Award no. 28/09-H within the framework of the Complex Program of Fundamental Investigations “Nanosized systems, nanomaterials, nanotechnology” of National Academy of Sciences of Ukraine.
Author information
Authors and Affiliations
Corresponding author
Appendix: Distribution functions of atoms in a simple crystal
Appendix: Distribution functions of atoms in a simple crystal
Here, we give expressions for distribution functions of atomic displacements in a simple perfect crystal consisting of N atoms of mass M (Karasevskii and Lubashenko 2002). In the harmonic approximation, an N-particle normalized function f N can be represented as a product of 3N independent real normal coordinates. In the coordinate representation, it is written as
where C N is a normalization factor, q nβ is the βth Cartesian coordinate of displacement \({\mathbf q}_{n}\) of atom n from its equilibrium position \({\mathbf R}_n, \) and coefficients B ββ′nn′ are given by
Here \(e_{j}({\mathbf k})\) is a polarization vector, summation over \({\mathbf k}\) is taken over the first Brillouin zone. A diagonal element of the matrix (14), B ββ nn , determines vibrational amplitudes of atom n in the β direction, and off-diagonal elements (n ≠n′) characterize correlations between displacements of different atoms.
For fcc and bcc lattices, \( B_{nn}^{\beta \beta{^\prime}}=\gamma \delta_{\beta \beta{^\prime}}, \) where
is the inverse square of the distribution width of atomic displacements which is independent of n and β. It was also found that all the off-diagonal elements of the matrix (14) are negligibly small except for that describing correlations between longitudinal components q nx and qn′x of displacements of two neighbouring atoms. Introducing a dimensionless correlation parameter ζ such that B xxnn′ = −γζ (0≤ζ < 1), we obtain
where the x axis goes through the sites \({\mathbf R}_n\) and \({\mathbf R}_{n{^\prime}}. \) Neglecting all the other off-diagonal elements, we get the N-particle distribution function of atomic coordinates in the form
with subscript n′ running over the first coordination sphere of atom n.
In order to calculate the average potential energy of pairwise interatomic interaction, we need a normalized two-particle (binary) distribution function \(f_2({\mathbf q}_n,{\mathbf q}_{n{^\prime}})\) of neighbouring atoms which is obtained by averaging the function f N over coordinates of all the atoms except n and n′. The result is given by
where factors g l(ζ), g t(ζ) and χ(ζ) (all positive and ≤1) determine effective reduction of parameters of the distribution function (17) due to interatomic correlations. Their functional form is defined by the lattice type. For example, for the fcc lattice, these functions appear as
The average energy of interaction of neighbouring atoms n and n′ is expressed as
where u(r nn′) is interatomic potential. For the Morse potential (5), the integration in (19) can be done analytically, and so we obtain
with q(ζ) = 2/(g l + ζχ). Since contribution of the transverse displacements into \(\langle U \rangle\) (the fractions in the denominators) is negligible for both RGC and metals (\(\alpha/(\gamma g_{\rm t} R) \sim 10^{-2}), \) we discard it in computation of the average potential energy and use Eq. 6.
Rights and permissions
About this article
Cite this article
Lubashenko, V.V. Size-dependent melting of nanocrystals: a self-consistent statistical approach. J Nanopart Res 12, 1837–1844 (2010). https://doi.org/10.1007/s11051-009-9743-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-009-9743-6