Abstract
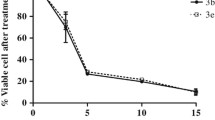
Paracoccidioides brasiliensis is the agent of paracoccidioidomycosis, the most prevalent deep mycosis in Latin America. Production of eicosanoids during fungal infections plays a critical role on fungal biology as well as on host immune response modulation. The purpose of our study was to assess whether P. brasiliensis strains with different degree of virulence (Pb18, Pb265, Bt79, Pb192) produce prostaglandin Ex (PGEx). Moreover, we asked if P. brasiliensis could use exogenous sources of arachidonic acid (AA), as well as metabolic pathways dependent on cyclooxygenase (COX) enzyme, as reported for mammalian cells. A possible association between this prostanoid and fungus viability was also assessed. Our results showed that all strains, independently of their virulence, produce high PGEx levels on 4 h culture that were reduced after 8 h. However, in both culture times, higher prostanoid levels were detected after supplementation of medium with exogenous AA. Treatment with indomethacin, a COX inhibitor, induced a reduction on PGEx, as well as in fungus viability. The data provide evidence that P. brasiliensis produces prostaglandin-like molecules by metabolizing either endogenous or exogenous AA. Moreover, the results suggest the involvement of these mediators on fungal viability.



Similar content being viewed by others
References
Brock TG, Peters-Golden M. Activation and regulation of cellular eicosanoid biosynthesis. Scientific World Journal. 2007;7:1273–84.
Jakobsson PJ, Thorén S, Morgenstern R, Samuelsson B. Identification of human prostaglandin E synthase: a microsomal, glutathione-dependent, inducible enzyme, constituting a potential novel drug target. Proc Natl Acad Sci USA. 1999;96:7220–5.
Betz M, Fox BS. Prostaglandin E2 inhibits production of Th1 lymphokines but not Th2 lymphokines. J Immunol. 1991;146:108–13.
Phipps RP, Stein SH, Roper RL. A new view of prostaglandin E regulation of the immune response. Immunol Today. 1991;12:349–52.
Rocca B, FitzGerald GA. Cyclooxygenases and prostaglandins: shaping up the immune response. Int Immunopharmacol. 2002;2:603–30.
Harizi H, Gualde N. Pivotal role of PGE2 and IL-10 in the cross-regulation of dendritic cell-derived inflammatory mediators. Cell Mol Immunol. 2006;3:271–7.
San-Blás G. Paracoccidioides brasiliensis: cell wall glucans, pathogenicity and dimorphism. Curr Top Med Mycol. 1985;1:235–57.
Franco M, Mendes RP, Moscardi-Bacchi M, Rezkallah-Iwasso MT, Montenegro MR. Paracoccidioidomycosis. Bailliere’s Clin Trop Med Commun Dis. 1989;4:185–220.
Franco M, Montenegro MR, Mendes RP, Marques SA, Dillon NL, Mota NG. Paracoccidioidomycosis: a recently proposed classification of its clinical forms. Rev Soc Bras Med Trop. 1987;20:129–32.
Brummer E, Castaneda E, Restrepo A. Paracoccidioidomycosis: an update. Clin Microbiol Rev. 1993;6:89–117.
Mendes RP. The gamut of clinical manifestations. In: Franco MF, Lacaz CS, Restrepo A, Del Negro G, editors. Paracoccidioidomycosis. Boca Raton: CRC Press; 1994. p. 233–52.
Benard G. An overview of the immunopathology of human Paracoccidioidomycosis. Mycopathologia. 2008;165:209–21.
Bordon AP, Dias-Melicio LA, Acorci MJ, Biondo GA, Fecchio D, Peraçoli MT, et al. Prostaglandin E(2) production by high and low virulent strains of Paracoccidioides brasiliensis. Mycopathologia. 2007;163:129–35.
Soares AMVC, Calvi SA, Peraçoli MTS, Fernandez AC, Dias LA, Dos Anjos AR. Modulatory effect of prostaglandins on human monocyte activation for killing of high- and low-virulence strains of Paracoccidioides brasiliensis. Immunology. 2001;102:480–5.
Noverr NC, Phare SM, Toews GB, Coffey MJ, Huffnagle GB. Pathogenic yeasts Cryptococcus neoformans and Candida albicans produce immunomodulatory prostaglandins. Infect Immun. 2001;69:2957–63.
Singer-Vermes LM, Ciavaglia MC, Casino SS, Burger E, Calich VLG. The source of the growth-promoting factor(s) affects the plating efficiency of Paracoccidioides brasiliensis. J Med Vet Mycol. 1992;30:261–4.
Harizi H, Gualde N. The impact of eicosanoids on the crosstalk between innate and adaptive immunity: the key roles of dendritic cells. Tissue Antigens. 2005;65:507–14.
Boyce JA. Eicosanoids in asthma, allergic inflammation, and host defense. Curr Mol Med. 2008;8:335–49.
Bordon AP, Dias-Melicio LA, Acorci MJ, Calvi SA, Serrão Peraçoli MT, Soares AM. Prostaglandin E2 inhibits Paracoccidioides brasiliensis killing by human monocytes. Microbes Infect. 2007;9:744–7.
Kurokawa CS, Lopes CR, Sugizaki MF, Kuramae EE, Franco MF, Peraçoli MT. Virulence profile of ten Paracoccidioides brasiliensis isolates: association with morphologic and genetic patterns. Rev Inst Med Trop São Paulo. 2005;47:257–62.
Noverr MC, Toews GB, Huffnagle GB. Production of prostaglandins and leukotrienes by pathogenic fungi. Infect Immun. 2002;70:400–2.
Gangopadhyay P, Thadepalli H, Roy I, Ansari A. Identification of species of Candida, Cryptococcus and Torulopsis by gas-liquid chromatography. J Infect Dis. 1979;140:952–8.
Marumo K, Aoki Y. Discriminant analysis of cellular fatty acids of Candida species, Torulopsis glabrata and Cryptococcus neoformans determined by gas-liquid chromatography. J Clin Microbiol. 1990;28:1509–13.
Botha A, Kock JL, Coetzee DJ, Botes PJ. Physiological properties and fatty acid composition in Mucor circinelloides f. circinelloides. Antonie Van Leeuwenhoek. 1997;71:201–6.
Lamacka M, Sajbidor J. The content of prostaglandins and their precursors in Mortierella and Cunninghamella species. Lett Appl Microbiol. 1998;26:224–6.
Pohl CH, Botha A, Kock JL, Coetzee DJ, Botes PJ, Schewe T, et al. Oxylipin formation in fungi: biotransformation of arachidonic acid to 3-hydroxy-5, 8-tetradecadienoic acid by Mucor genevensis. Biochem Biophys Res Commun. 1998;253:703–6.
Botha A, Paul I, Roux C, Kock JL, Coetzee DJ, Strauss T, et al. An isolation procedure for arachidonic acid producing Mortierella species. Antonie Van Leeuwenhoek. 1999;75:253–6.
Noverr MC, Erb-Downward JR, Huffnagle GB. Production of eicosanoids and other oxylipins by pathogenic eukaryotic microbes. Clin Microbiol Rev. 2003;16:517–33.
Erb-Downward JR, Huffnagle GB. Cryptococcus neoformans produces authentic prostaglandin E2 without a cyclooxygenase. Eukaryot Cell. 2007;6:346–50.
Erb-Downward JR, Noverr MC. Characterization of prostaglandin E2 production by Candida albicans. Infect Immun. 2007;75:3498–505.
Tsitsigiannis DI, Bok JW, Andes D, Nielsen KF, Frisvad JC, Keller NP. Aspergillus cyclooxygenase-like enzymes are associated with prostaglandin production and virulence. Infect Immun. 2005;73:4548–59.
Acknowledgments
Conflict of interest statement
There is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Biondo, G.A., Dias-Melicio, L.A., Bordon-Graciani, A.P. et al. Paracoccidioides brasiliensis Uses Endogenous and Exogenous Arachidonic Acid for PGEx Production. Mycopathologia 170, 123–130 (2010). https://doi.org/10.1007/s11046-010-9301-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-010-9301-x