Abstract
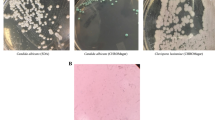
The aim of this study was to evaluate the prevalence of Candida spp., and particularly C. dubliniensis, among oral isolates from Brazilian HIV-positive patients correlating these results with CD4 cell counts and viral load. Forty-five individuals (23 female and 22 male) diagnosed as HIV-positive by ELISA and Western-blot, under anti-retroviral therapy for at least 1 year and without oral candidosis signals were included in the study. The control group was constituted by 45 healthy individuals, matched to the test group in relation to age, gender, and oral conditions. Oral rinses were collected and the identification was performed by phenotypic tests. The existence of C. dubliniensis among the isolates was analyzed using a validated multiplex PCR assay. Candida spp. were detected at significantly higher number in the oral cavity of HIV-positive patients in relation to the controls (P = 0.0008). C. albicans was the most frequently isolated species in both groups. In the HIV group, C. glabrata, C. lipolytica, C. krusei, C. guilliermondii, and C. parapsilosis were also identified. In the control group, we additionally identified C. tropicalis and C. dubliniensis. Two isolates (1.9%, 2/108) from control individuals were identified as C. dubliniensis and this species was not verified in the HIV group. Candida spp. counts were statistically lower (P = 0.0230) in the oral cavity of patients with low viral load (<400 copies/mm³). Candida spp. counts did not differ statistically among groups with different levels of CD4 cells counts (P = 0.1068).

Similar content being viewed by others
References
Samaranayake LP, et al. Fungal infections associated with HIV infection. Oral Dis. 2002;8(Suppl 2):151–60.
Aas JA, Paster BJ, Stokes LN, Olsen I, Dewhirst FE. Defining the normal bacterial flora of the oral cavity. J Clin Microbiol. 2005;43:5721–32. doi:10.1128/JCM.43.11.5721-5732.2005.
Eggimann P, Garbino J, Pittet D. Epidemiology of Candida species infections in critically ill non-immunossupressed patients. Lancet Infect Dis. 2003;3:658–720.
Coleman DC, Rinaldi MG, Haynes KA, Rex JH, Summerbell RC, Anaissie EJ, et al. Importance of Candida species other than Candida albicans as opportunistic pathogens. Med Mycol. 1998;36:156–65.
Lin AL, et al. Salivary anticandidal activity and saliva composition in a HIV-infected cohort. Oral Microbiol Immunol. 2001;16(5):270–8. doi:10.1034/j.1399-302x.2001.016005270.x.
Al-Abeid HM, Khaled HA, Elkarmi AZ, Hamad MA. Isolation. characterization of Candida spp. in Jordanian cancer patients: prevalence, pathogenic determinants, and antifungal sensitivity. Jpn J Infect Dis. 2004;57:279–84.
Costa EMB, et al. Heterogeneity of metallo and serine extracellular proteinases in oral clinical isolates of Candida albicans in HIV-positive and healthy children from Rio de Janeiro, Brazil. FEMS Immunol Med Microbiol. 2003;38(2):173–80. doi:10.1016/S0928-8244(03)00145-7.
Capoluongo E, et al. Heterogeneity of oral isolates of Candida albicans in HIV-positive patients: correlation between candidal carriage, karyotype and disease stage. J Med Microbiol. 2000;49(11):985–91.
Li SY, et al. Molecular epidemiology of long-term colonization of Candida albicans strain from HIV-infected patients. Epidemiol Infect. 2006;134(2):265–96. doi:10.1017/S0950268805004905.
Campisi G, Pizzo G, Milici ME. Candidal carriage in the oral cavity of human immunodeficiency virus-infected subjects. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2002;93(3):281–6. doi:10.1067/moe.2002.120804.
Barchiesi F, Maracci M, Radi B, Arzeni D, Baldassarri I, Giacometti A, et al. Point prevalence, microbiology and fluconazole susceptibility patterns of yeast isolates colonizing the oral cavities of HIV-infected patients in the era of highly active antiretroviral therapy. J Antimicrob Chem. 2002;50:999–1002. doi:10.1093/jac/dkf233.
Sánchez-Vargas LO, et al. Oral isolates colonyzing infecting human immunodeficiency virus-infected and healthy persons in Mexico. J Clin Microbiol. 2005;43(8):4159–62. doi:10.1128/JCM.43.8.4159-4162.2005.
Sullivan DJ, Moran GP, Pinjon E, Al-Mosaid A, Stokes C, Vaughan C, et al. Comparison of the epidemiology, drug resistance mechanisms, and virulence of Candida dubliniensis and Candida albicans. FEMS Yeast Res. 2004;4:369–76.
Alves SH, Milan EP, Branchini ML, Nishimura K, Fukushima K, Oliveira LO, et al. First isolation of Candida dubliniensis in Rio Grande do Sul, Brazil. Diagn Microbiol Infect Dis. 2001;39:165–8. doi:10.1016/S0732-8893(01)00220-6.
Milan EP, Laet Sant’Ana P, Melo ASA, Sullivan DJ, Coleman DC, Lewi D, et al. Multicenter prospective surveillance of oral Candida dubliniensis among adult Brazilian human immunodeficiency virus-positive and AIDS patients. Diagn Microbiol Infect Dis. 2001;41:29–35. doi:10.1016/S0732-8893(01)00290-5.
Chavasco JK, Paula CR, Hirata MY, Aleva NA, Melo CE, Gambale W, et al. Molecular identification of Candida dubliniensis isolated from oral lesions of HIV-positive and HIV-negative patients in São Paulo, Brazil. Rev Inst Med Trop Sao Paulo. 2006;48(1):1–26. doi:10.1590/S0036-46652006000100005.
Portela MB, Souza IPR, Costa EMMB, Hagler AN, Soares RMA, Santos ALS. Differential recovery of Candida species from subgingival sites in human immunodeficiency virus-positive and healthy children from Rio de Janeiro, Brazil. J Clin Microbiol. 2004;42(12):5925–7. doi:10.1128/JCM.42.12.5925-5927.2004.
Samaranayake LP, et al. A comparison or oral rinse and imprint sampling techniques for detection of yeast, coliform and Staphylococcus aureus carriage in the oral cavity. J Oral Pathol. 1986;15:386–8. doi:10.1111/j.1600-0714.1986.tb00646.x.
Sandven P. Laboratory identification and sensitivity testing of yeast isolates. Acta Odontol Scand. 1990;48:27–36. doi:10.3109/00016359009012731.
Williams DW, Lewis MAO. Isolation and identification of Candida from the oral cavity. Oral Dis. 2000;6:3–11.
Donnely SM, Sullivan DJ, Shanley DB, Coleman DC. Phylogenetic analysis and rapid identification of Candida dubliniensis based on analysis of ACT1 intron and exon sequences. Microbiology. 1999;145:1871–82.
Mähnß B, Stehr F, Schäfer W, Neuber K. Comparison of standard phenotypic assays with a PCR method to discriminate Candida albicans and Candida dubliniensis. Mycoses. 2005;46:55–61. doi:10.1111/j.1439-0507.2004.01054.x.
Center for disease control and prevention. Reported CD4+ T-lymphocyte results for adults and adolescents with HIV/AIDS–33 states, 2005. HIV/AIDS Surveillance Supplemental Report. 2005;11(2):1–31.
Center for disease control and prevention. Guidelines for using antiretroviral agents among HIV-infected adults and adults and adolescents. Recommendations of the panel of clinical practices for treatment of HIV. Morb Mortal Wkly Rep. 2002;nRR-7:1–64.
Nittayananta W, Jealeae S, Winn T. Oral Candida in HIV-infected heterosexuals and intravenous drug users in Thailand. J Oral Pathol Med. 2001;30(6):347–54. doi:10.1034/j.1600-0714.2001.300604.x.
Gonçalves LS, et al. Association of T CD4 lymphocyte levels and chronic perodontitis in HIV-infected brazilian patients undergoing highly active anti-retroviral therapy: clinical results. J Periodontol. 2005;76(6):915–22. doi:10.1902/jop.2005.76.6.915.
Perezous LF, et al. Colonization of Candida species in denture wearers with emphasis on HIV infection: a literature review. J Prosthet Dent. 2005;93(3):288–93. doi:10.1016/j.prosdent.2004.11.015.
Jabra-Rizk MA, Falkler WA, Enwoncu CO, Onwujekwe DI, Merz G, Meiller TF. Prevalence of yeast among children in Nigeria and the United States. Oral Microbiol Immunol. 2001;16:383–5. doi:10.1034/j.1399-302X.2001.160611.x.
Koga-Ito CY, Lyon JP, Vidotto V, Resende MA. Virulence factors and antifungal susceptibility of Candida albicans isolates from oral candidosis patients and control individuals. Mycopathologia. 2006;161:219–23. doi:10.1007/s11046-005-0001-x.
Tsang CSP, Samaranayake LP. Oral yeasts and coliforms in HIV-infected individuals in Hong Kong. Mycoses. 2000;43(7–8):303–8. doi:10.1046/j.1439-0507.2000.00584.x.
Ahmad S, Khan Z, Mokaddas E, Khan ZU. Isolation and molecular identification of Candida dubliniensis from non-human immunodeficiency virus-infected patients in Kuwait. J Med Microbiol. 2004;53:633–7. doi:10.1099/jmm.0.05315-0.
Gugnani HC, Becker K, Fegeler W, Basu S, Chattopadhya D, Baveja U, et al. Oropharyngeal carriage of Candida species in HIV-infected patients in India. Mycoses. 2003;46:281–8. doi:10.1046/j.1439-0507.2003.00896.x.
Blignaut E, Messer S, Hollis RJ, Pfaller MA. Antifungal susceptibility of South African oral yeast isolates from HIV/AIDS patients and healthy individuals. Diagn Microbiol Infect Dis. 2002;44:169–74. doi:10.1016/S0732.08893(02)00440.6.
Mosca CO, Moragues MD, Brena S, Rosa AC, Pontón J. Isolation of Candida dubliniensis in a teenager with denture stomatitis. Med Oral Patol Oral Cir Bucal. 2005;10:25–31.
Sant’Ana PL, et al. Multicenter brazilian study of oral Candida species isolated from AIDS patients. Mem Inst Oswaldo Cruz. 2002;97(2):253–7. doi:10.1590/S0074.02762002000200019.
Bodey GP, et al. The epidemiology of Candida glabrata and Candida albicans fungemia in immunocompromised patients with cancer. Am J Med. 2002;112(5):380–385. doi:10.1016/S0002-9343(01)01130-5.
Acknowledgement
The authors would like to thank Dr. F. G. Nóbrega for suggestions and experimental support, and Fundação de Amparo à Pesquisa do Estado de São Paulo-FAPESP (2004/14088-8; 2004/12381-6) for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Back-Brito, G.N., Mota, A.J., Vasconcellos, T.C. et al. Frequency of Candida spp. in the Oral Cavity of Brazilian HIV-Positive Patients and Correlation with CD4 Cell Counts and Viral Load. Mycopathologia 167, 81–87 (2009). https://doi.org/10.1007/s11046-008-9153-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-008-9153-9