Abstract
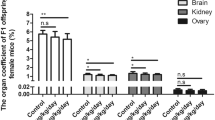
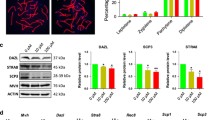
Diethylhexyl phthalate (DEHP) is an estrogen-like compound widely used as a plasticizer in commercial products and is present in medical devices, and common household items. It is considered an endocrine disruptor since studies on experimental animals clearly show that exposure to DEHP can alter epigenetics of germ cells. This study was designed to assess the effects of DEHP on DNA methylation of imprinting genes in germ cells from fetal and adult mouse. Pregnant mice were treated with DEHP at doses of 0 and 40 μg DEHP/kg body weight/day from 0.5 to 18.5 day post coitum. The data revealed DEHP exposure significantly reduced the percentage of methylated CpG sites in Igf2r and Peg3 differentially methylated regions (DMRs) in primordial germ cells from female and male fetal mouse, particularly, in the oocytes of 21 dpp mice (F1), which were produced by the pregnant micetreated with DEHP. More surprisingly, the modification of the DNA methylation of imprinted genes in F1 mouse oocytes was heritable to F2 offspring which exhibit lower percentages of methylated CpG sites in imprinted genes DMRs. In conclusion, DEHP exposure can affect the DNA methylation of imprinting genes not only in fetal mouse germ cells and growing oocytes, but also in offspring’s oocytes.





Similar content being viewed by others
References
Singh S, Li SS (2012) Epigenetic effects of environmental chemicals bisphenol a and phthalates. Int J Mol Sci 13(8):10143–10153
Zhang T, Li L, Qin XS, Zhou Y, Zhang XF, Wang LQ, De Felici M, Chen H, Qin GQ, Shen W (2013b). Di-(2-ethylhexyl) phthalate and bisphenol A exposure impairs mouse primordial follicle assembly in vitro. Environ Mol Mutagen
Zhang XF, Zhang LJ, Li L, Feng YN, Chen B, Ma JM, Huynh E, Shi QH, De Felici M, Shen W (2013) Diethylhexyl phthalate exposure impairs follicular development and affects oocyte maturation in the mouse. Environ Mol Mutagen 54(5):354–361
Zhang XF, Zhang T, Wang L, Zhang HY, Chen YD, Qin XS, Feng YM, Feng YN, Shen W, Li L (2013) Effects of diethylhexyl phthalate (DEHP) given neonatally on spermatogenesis of mice. Mol Biol Rep 40:6509–6517
Ambruosi B, Uranio MF, Sardanelli AM, Pocar P, Martino NA, Paternoster MS, Amati F, Dell’Aquila ME (2009) In vitro acute exposure to DEHP affects oocyte meiotic maturation, energy and oxidative stress parameters in a large animal model. PLoS One 6(11):e27452
Braun JM, Smith KW, Williams PL, Calafat AM, Berry K, Ehrlich S, Hauser R (2012) Variability of urinary phthalate metabolite and bisphenol A concentrations before and during pregnancy. Environ Health Perspect 120(5):739–745
Carnevali O, Tosti L, Speciale C, Peng C, Zhu Y, Maradonna F (2010) DEHP impairs zebrafish reproduction by affecting critical factors in oogenesis. PLoS One 5(4):e10201
Dalman A, Eimani H, Sepehri H, Ashtiani SK, Valojerdi MR, Eftekhari-Yazdi P, Shahverdi A (2008) Effect of mono-(2-ethylhexyl) phthalate (MEHP) on resumption of meiosis, in vitro maturation and embryo development of immature mouse oocytes. BioFactors 33(2):149–155
Lovekamp TN, Davis BJ (2001) Mono-(2-ethylhexyl) phthalate suppresses aromatase transcript levels and estradiol production in cultured rat granulosa cells. Toxicol Appl Pharmacol 172(3):217–224
Zhang GL, Zhang XF, Feng YM, Li L, Sun XF, Sun ZY, Shen W (2013) Exposure to bisphenol A results in a decline in mouse spermatogenesis. Reprod Fertil Dev 25(6):847–859
Song ZH, Min LJ, Pan QJ, Shi QH, Shen W (2009) Maternal imprinting during mouse oocyte growth in vivo and in vitro. Biochem Biophys Res Commun 387:800–805
Chao HH, Zhang XF, Chen B, Pan B, Zhang LJ, Li L, Sun XF, Shi QH, Shen W (2012) Bisphenol A exposure modifies methylation of imprinted genes in mouse oocytes via the estrogen receptor signaling pathway. Histochem Cell Biol 137(2):249–259
Eichenlaub-Ritter U, Vogt E, Cukurcam S, Sun F, Pacchierotti F, Parry J (2008) Exposure of mouse oocytes to bisphenol A causes meiotic arrest but not aneuploidy. Mutat Res 651(1–2):82–92
Hunt PA, Lawson C, Gieske M, Murdoch B, Smith H, Marre A, Hassold T, VandeVoort CA (2012) Bisphenol A alters early oogenesis and follicle formation in the fetal ovary of the rhesus monkey. Proc Natl Acad Sci USA 109(43):17525–17530
Anderson OS, Nahar MS, Faulk C, Jones TR, Liao C, Kannan K, Weinhouse C, Rozek LS, Dolinoy DC (2012) Epigenetic responses following maternal dietary exposure to physiologically relevant levels of bisphenol A. Environ Mol Mutagen 53(5):334–342
Susiarjo M, Sasson I, Mesaros C, Bartolomei MS (2013) Bisphenol A exposure disrupts genomic imprinting in the mouse. PLoS Genet 9(4):e1003401
Zhang HQ, Zhang XF, Zhang LJ, Chao HH, Pan B, Feng YM, Li L, Sun XF, Shen W (2012) Fetal exposure to bisphenol A affects the primordial follicle formation by inhibiting the meiotic progression of oocytes. Mol Biol Rep 39(5):5651–5657
Zhang XF, Zhang LJ, Feng YN, Chen B, Feng YM, Liang GJ, Li L, Shen W (2012) Bisphenol A exposure modifies DNA methylation of imprint genes in mouse fetal germ cells. Mol Biol Rep 39(9):8621–8628
Pesce M, De Felici M (1995) Purification of mouse primordial germ cells by MiniMACS magnetic separation system. Dev Biol 170:722–725
Bromer JG, Zhou Y, Taylor MB, Doherty L, Taylor HS (2010) Bisphenol-A exposure in utero leads to epigenetic alterations in the developmental programming of uterine estrogen response. FASEB J 24(7):2273–2280
Kundakovic M, Gudsnuk K, Franks B, Madrid J, Miller RL, Perera FP, Champagne FA (2013) Sex-specific epigenetic disruption and behavioral changes following low-dose in utero bisphenol A exposure. Proc Natl Acad Sci USA 110(24):9956–9961
Kundakovic M, Champagne FA (2011) Epigenetic perspective on the developmental effects of bisphenol A. Brain Behav Immun 25:1084–1093
Dolinoy DC, Huang D, Jirtle RL (2007) Maternal nutrient supplementation counteracts bisphenol A-induced DNA hypomethylation in early development. Proc Natl Acad Sci USA 104(32):13056–13061
Yaoi T, Itoh K, Nakamura K, Ogi H, Fujiwara Y, Fushiki S (2008) Genome-wide analysis of epigenomic alterations in fetal mouse forebrain after exposure to low doses of bisphenol A. Biochem Biophys Res Commun 376:563–567
Pathak S, D’Souza R, Ankolkar M, Gaonkar R, Balasinor NH (2010) Potential role of estrogen in regulation of the insulin-like growth factor2-H19 locus in the rat testis. Mol Cell Endocrinol 15:110–117
Pathak S, Kedia-Mokashi N, Saxena M, D’Souza R, Maitra A, Parte P, Gill-Sharma M, Balasinor N (2009) Effect of tamoxifen treatment on global and insulin-like growth factor 2–H19 locus-specific DNA methylation in rat spermatozoa and its association with embryo loss. Fertil Steril 91:2253–2263
Doshi T, D’souza C, Vanage G (2013) Aberrant DNA methylation at Igf2-H19 imprinting control region in spermatozoa upon neonatal exposure to bisphenol A and its association with post implantation loss. Mol Biol Rep 40(8):4747–4757
Acknowledgments
This work is supported by National Basic Research Program of China (973 Program, 2012CB944401), National Nature Science Foundation (31201120 and 31001010) and Program for New Century Excellent Talents in University (NCET-12-1026), Foundation of Distinguished Young Scholars (JQ201109), and Nature Science Foundation of Hubei Province (2011CDB230).
Conflict of interest
The authors fully declare any financial or other potential conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, L., Zhang, T., Qin, XS. et al. Exposure to diethylhexyl phthalate (DEHP) results in a heritable modification of imprint genes DNA methylation in mouse oocytes. Mol Biol Rep 41, 1227–1235 (2014). https://doi.org/10.1007/s11033-013-2967-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-013-2967-7