Abstract
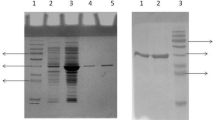
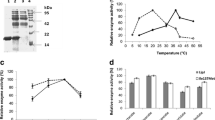
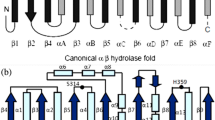
A lip gene from a Bacillus isolate was cloned and expressed in E. coli. By thermal denaturation analysis, T1/2 of lipase was observed to be 7 min at 50°C with less than 10% activity after 1 h incubation at 50°C. To expand the functionality of cloned lipase, attempts have been made to create thermostable variants of lip gene. A lipase variant with an isoleucine to threonine amino acid substitution at the protein surface was isolated that demonstrated higher thermostability than its wild type predecessor. To explore the structure–function relationship, the lip gene product of wild type (WT) and mutant was characterized in detail. The mutation enhanced the specific activity of enzyme by 2-folds when compared with WT. The mutant enzyme showed enhanced T1/2 of 21 min at 50°C. The kinetic parameters of the mutant enzyme were significantly altered. The mutant enzyme displayed higher affinity for substrate (decreased K m ) in comparison to the wild type. The k cat and catalytic efficiency (k cat/K m ) of mutant were also enhanced by two and five times, respectively, as compared with the WT. The mutation resides on the part of helix which is exposed to the solvent and away from the catalytic triad. The replacement of a solvent exposed hydrophobic residue (Ile) in WT with a hydrophilic residue (Thr) in mutant might impart thermostability to the protein structure.





Similar content being viewed by others
References
Brockman HW, Mornsen WE, Tsuijita T (1988) The biology, biochemistry and technology of lipases. J Am Oil Chem Soc 65:891–896
Leow TC, Rahman RN, Basri M, Salleh B (2004) High level expression of thermostable lipase from Geobacillus sp Strain T1. Biosci Biotechnol Biochem 68:96–103
Ewis HE, Abdelal AT, Lu C (2002) Molecular cloning and characterization of two thermostable carboxyl esterases from G. stearothermophilus. Gene 329:187–195
Nawani N, Khurana J, Kaur J (2006) A thermostable lipolytic enzyme from a thermophilic Bacillus sp: purification and characterization. Mol Cell Biochem 290:17–22
Castro-Ochoa LD, Rodriguez-Gomez C, Valerio-Alfaro G, Ros RO (2005) Screening, purification and characterization of the thermoalkalophilic lipase produced by Bacillus thermoleovorans CCR11. Enzym Microb Technol 37:648–654
Nawani N, Kaur J (2007) Studies on lipolytic isoenzymes from a thermophilic Bacillus sp: production, purification and biochemical characterization. Enzym Microb Technol 40:881–887
Giver L, Gershenson A, Freskgard P, Arnold FH (1998) Directed evolution of a thermostable esterase. Proc Natl Acad Sci USA 95:15609–15613
Kumar S, Tsai CJ, Nussinov R (2000) Factors enhancing protein thermostability. Protein Eng 13:179–191
Eijsink V, Bjørk A, Gaseidnes S, Sirevag R, Synstad B, Burg B, Vriend G (2004) Rational engineering of enzyme stability. J Biotechnol 113:105–120
Khan MIH, Kousuke I, Hyeung K, Hiroyuki A, Takahiro I, Hitoshi S, Yoshihiro S (2005) Molecular properties and enhancement of thermostability by random mutagenesis of glutamate dehydrogenase from Bacillus subtilis. Biosci Biotechnol Biochem 69:1861–1870
Dalby PA (2003) Optimizing enzyme function by directed evolution. Curr Opin Struct Biol 13:500–505
Koutsioulis D, Wang E, Tzanodaskalaki M, Nikiforaki D, Deli A, Feller G, Heikinheimo P, Bouriotis V (2008) Directed evolution on the cold adapted properties of TAB 5 alkaline phosphatase. Protein Eng Des Sel 21:319–327
Kuchner O, Arnold FH (1997) Directed evolution of enzyme catalysts. Trends Biotechnol 15:523–530
Zhao HM, Arnold FH (1999) Directed evolution converts subtilisin E into a functional equivalent of thermitase. Protein Eng 12:47–53
Reetz MT (2004) Controlling the enantioselectivity of enzymes by directed evolution: practical and theoretical ramifications. Proc Natl Acad Sci USA 101:5716–5722
Leung DW, Chen E, Goeddel DV (1989) A method for random mutagenesis of a defined DNA segment using a modified polymerase chain reaction. Technique 1:11–15
Sigurgisladottirr S, Konraosdottir M, Jonson A (1993) Lipase activity of thermophilic bacteria from Icelandic hot springs. Biotechnol Lett 15:361–366
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular Cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Peitsch MC (1996) ProMod and Swiss-Model: Internet-based tools for automated comparative protein modeling. Biochem Soc Trans 24:274–279
Acharya P, Rajakumara E, Sankaranarayanan R, Rao NM (2004) Structural Basis of selection and thermostability of laboratory evolved Bacillus subtilis lipase. J Mol Biol 341:1271–1561
Ahmad SA, Kamal MZ, Sankaranarayanan R, Rao NM (2008) Thermostable Bacillus subtilis lipases: in-vitro evolution and structural insight. J Mol Biol 381:324–340
Kawamura S, Abe Y, Ueda T, Masumoto K, Imoto T, Yamasaki N, Kimura M (1998) Investigation of the structural basis for thermostability of DNA-binding protein HU from Bacillus stearothermophilus. J Biol Chem 273:19982–19987
Eggert T, Pencreac’h G, Douchet I, Verger R, Jaeger KE (2000) A novel esterase from B. subtilis and its conversion to a monoacylglycerol hydrolase. Eur J Biochem 267:6459–6469
Eggert T, Pouderoyen G, Dijkstra BW, Jaeger KE (2001) Lipolytic enzymes LipA and LipB from Bacillus subtilis differ in regulation of gene expression, biochemical properties and three dimensional structure. FEBS Lett 502:89–92
Ruiz C, Pastor FIJ, Diaz P (2003) Isolation and characterization of Bacillus sp BP-6 LipA, a ubiquitous lipase among mesophilic Bacillus species. Lett Appl Microbiol 37:354–359
Ruiz C, Pastor FIJ, Diaz P (2002) Analysis of Bacillus megaterium lipolytic system and cloning of LipA, a novel subfamily 1.4 bacterial lipase. FEMS Microbiol Lett 217:263–267
Moeller B, Vetter R, Wilke D, Foullois B (1992) Alkane lipases from Bacillus and the cloning of their genes. Patent number WO9116422. Applicant(s) Kali Chemie A-G
Dartois V, Baulard A, Schanck K, Colson C (1992) Cloning, nucleotide sequence and expression in E. coli of a lipase gene from Bacillus subtilis 168. Biochim Biophys Acta 1131:253–260
Uchiyama H, Inaoka T, Ohkuma-Soyejima T, Togaine H, Shibanaka Y, Yoshimoto T, Eokubo T (2000) Directed evolution to improve the thermostability of prolyl-endopeptidase. J Biochem 156:441–447
Johannes TW, Woodyer RD, Zhao H (2005) Directed evolution of a thermostable phosphite dehydrogenase for NAD(P)H regeneration. Appl Environ Microbiol 71:5734–5756
Schreiber G, Buckle AM, Fersht AR (1994) Stability and function: two constraints in the evolution of barstar and other proteins. Structure 2:945–951
Patkar S, Vind J, Kelstrup E, Christensen MW, Svendsen A, Borch K, Kirk O (1998) Effect of mutations in Candida antarctica B lipase. Chem Phys Lipids 93:95–101
Van GP, Eggert T, Jaeger KE, Dijkstra BW (2001) The crystal structure of Bacillus subtilis lipase: a minimal alpha/beta hydrolase fold enzyme. J Mol Biol 309:215–226
Vogt G, Woell S, Argos P (1997) Protein thermal stability, hydrogen bonds and ion pairs. J Mol Biol 269:631–643
Acknowledgment
This study was supported by financial assistance to JK by Department of Science and Technology, New Delhi, India.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khurana, J., Singh, R. & Kaur, J. Engineering of Bacillus lipase by directed evolution for enhanced thermal stability: effect of isoleucine to threonine mutation at protein surface. Mol Biol Rep 38, 2919–2926 (2011). https://doi.org/10.1007/s11033-010-9954-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-010-9954-z