Abstract
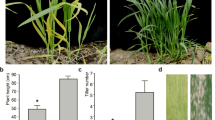
Leaf color-related genes play key roles in chloroplast development and photosynthetic pigment biosynthesis and affect photosynthetic efficiency and grain yield in crops. In this study, a recessive homozygous individual displaying yellow leaf color (yl1) was identified in the progeny population derived from a cross between wheat cultivars Xingmai1 (XM1) and Yunong3114 (YN3114). Phenotypic identification showed that yl1 exhibited the yellow character state over the entire growth period. Compared with XM1, yl1 plants had significantly lower chlorophyll content and net photosynthetic rate, and similar results were found between the green-type lines and yellow-type lines in the BC2F3 XM1 × yl1 population. Gene mapping via the bulked segregant exome capture sequencing (BSE-seq) method showed that the target gene TaYL1 was located within the region of 582,556,971–600,837,326 bp on chromosome 7D. Further analysis by RNA-seq suggested TraesCS7D02G469200 as a candidate gene for yellow leaf color in common wheat, which encodes a protein containing the AP2 domain. Moreover, comparative transcriptome profiling revealed that most differentially expressed genes (DEGs) were enriched in chlorophyll metabolism and photosynthesis pathways. Together, these results indicate that TaYL1 potentially affects chlorophyll synthesis and photosynthesis. This study further elucidates the biological mechanism of chlorophyll synthesis, metabolism, and photosynthesis in wheat and provides a theoretical basis for high photosynthetic efficiency in wheat breeding.




Similar content being viewed by others
Data availability
Data used in the manuscript as electronic supplementary materials. The research materials used in this study can be obtained by request.
References
Blum A (1985) Photosynthesis and transpiration in leaves and ears of wheat and barley varieties. J Exp Bot 36(3):432–440
Cao L, Wang H, Sun D, Yi F, Li X, Min D (2010) Chloroplast thylakoid protein composition and characteristics of chlorophyll biosynthesis in a novel aurea mutant of wheat. J Triticeae Crops 30(4):638–643
Dong C, Zhang L, Chen Z, Xia C, Gu Y, Wang J, Li D, Xie Z, Zhang Q, Zhang X, Gui L, Liu X, Kong X (2020) Combining a new exome capture panel with an effective varBScore algorithm accelerates BSA-based gene cloning in wheat. Front Plant Sci 11:1249
Ferreira FJ, Guo C, Coleman JR (2008) Reduction of plastid-localized carbonic anhydrase activity results in reduced Arabidopsis seedling survivorship. Plant Physiol 147(2):585–594
Gaudinier A, Rodriguez-Medina J, Zhang L, Olson A, Liseron-Monfils C, Bågman AM, Foret J, Abbitt S, Tang M, Li B, Runcie DE, Kliebenstein DJ, Shen B, Frank MJ, Ware D, Brady SM (2018) Transcriptional regulation of nitrogen-associated metabolism and growth. Nature 563(7730):259–264
Hou D, Xu H, Du G, Lin J, Duan M, Guo A (2009) Proteome analysis of chloroplast proteins in stage albinism line of winter wheat (triticum aestivum) FA85. BMB Rep 42(7):450–455
Hussain W, Baenziger PS, Belamkar V, Guttieri MJ, Venegas JP, Easterly A, Sallam A, Poland J (2017) Genotyping-by sequencing derived high-density linkage map and its application to QTL mapping of flag leaf traits in bread wheat. Sci Rep 7(1):16394
Kosuge K, Watanabe N, Kuboyama T (2011) Comparative genetic mapping of homoeologous genes for the chlorine phenotype in the genus Triticum. Euphytica 179(2):257–263
Lawlor DW, Paul MJ (2014) Source/sink interactions underpin crop yield: the case for trehalose 6-phosphate/SnRK1 in improvement of wheat. Front Plant Sci 5:418
Li N, Jia J, Xia C, Liu X, Kong X (2013) Characterization and mapping of novel chlorophyll deficient mutant genes in durum wheat. Breed Sci 63(2):169–175
Liu X, Xu H, Zhang J, Liang G, Liu Y, Guo A (2012) Effect of low temperature on chlorophyll biosynthesis in albinism line of wheat (Triticum aestivum) FA85. Physiol Plant 145(3):384–394
Liu Y, Tao Y, Wang Z, Guo Q, Wu F, Yang X, Deng M, Ma J, Chen G, Wei Y, Zheng Y (2018) Identification of QTL for flag leaf length in common wheat and their pleiotropic effects. Mol Breed 38:11
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2 (−Delta Delta C(T)) method. Methods 25(4):402–408
Lu Q, Lu C (2004) Photosynthetic pigment composition and photoststem II photochemistry of wheat ears. Plant Physiol Bioch 42(5):395–402
Maekawa M, Hase Y, Shikazono N, Tanaka A (2003) Induction of somatic instability in stable yellow leaf mutant of rice by ion beam irradiation. Nucl Instrum Methods Phys Res 206:579–585
Makino A (2011) Photosynthesis, grain yield, and nitrogen utilization in rice and wheat. Plant Physiol 155(1):125–129
Ma S, Wang M, Wu J, Guo W, Chen Y, Li G, Wang Y, Shi W, Xia G, Fu D, Kang Z, Ni F (2021) WheatOmics: A platform combining multiple omics data to accelerate functional genomics studies in wheat. Mol Plant 14(12):1965–1968
Murchie E, Kefauver S, Araus JL, Muller O, Rascher U, Flood PJ, Lawson T (2018) Measuring the dynamic photosynthome. Ann Bot 122(2):207–220
Neatby KW (1933) A chlorophyll mutation in wheat. J Hered 24(4):159–162
Rudnik R, Bulcha JT, Reifschneider E, Ellersiek U, Baier M (2017) Specificity versus redundancy in the RAP2.4 transcription factor family of Arabidopsis thaliana: transcriptional regulation of genes for chloroplast peroxidases. BMC Plant Biol 17(1):144
Sears LMS, Sears ER (1968) The mutants chlorina-1 and Hermsen’s virescent. In: Proceedings of the 3rd International Wheat Genetics Symposium, Eds: KW Finlay and KW Shepherd, Australian Academy of Sciences, Canberra, Australia, pp 299–304
Tambussi EA, Bort J, Guiamet JJ, Nogues S, Araus JL (2007) The photosynthetic role of ears in C3 cereals: Metabolism, water use efficiency and contribution to grain yield. Crit Rev Plant Sci 26:1–16
Tanaka A, Tanaka R (2006) Chlorophyll metabolism. Curr Opin Plant Biol 9(3):248–255
Thimm O, Bläsing O, Gibon Y, Nagel A, Meyer S, Krüger P, Selbig J, Müller LA, Rhee SY, Stitt M (2004) MAPMAN: a user-driven tool to display genomics data sets onto diagrams of metabolic pathways and other biological processes. Plant J 37(6):914–939
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28(5):511–515
Walker BJ, Vanloocke A, Bernacchi CJ, Ort DR (2016) The costs of photorespiration to food production now and in the future. Annu Rev Plant Biol 67:107–129
Wang Y, Xi W, Wang Z, Wang B, Xu X, Han M, Zhou S, Zhang Y (2016) Contribution of ear photosynthesis to grain yield under rainfed and irrigation conditions for winter wheat cultivars released in the past 30 years in north China plain. J Integr Agric 15(10):2247–2256
Watanabe N, Koval SF (2003) Mapping of chlorina mutant genes on the long arm of homoeologous group 7 chromosomes in common wheat with partial deletion lines. Euphytica 129(3):259–265
Yang Y, Xu J, Rao Y, Zeng Y, Liu H, Zheng T, Zhang G, Hu J, Guo L, Qian Q, Zeng D (2016) Cloning and functional analysis of pale-green leaf (PGL10) in rice (Oryza sativa L.). Plant Growth Regul 78:69–77
Yan X, Zhao L, Ren Y, Zhang N, Dong Z, Chen F (2020) Identification of genetic loci and a candidate gene related to flag leaf traits in common wheat by genome-wide association study and linkage mapping. Mol Breed 40:58
Zhang L, Dong C, Chen Z, Gui L, Chen C, Li D, Xie Z, Zhang Q, Zhang X, Xia C, Liu X, Kong X, Wang J (2021) WheatGmap: a comprehensive platform for wheat gene mapping and genomic studies. Mol Plant 14(2):187–190
Zhang L, Liu C, An X, Wu H, Feng Y, Wang H, Sun D (2017) Identification and genetic mapping of a novel incompletely dominant yellow leaf color gene, Y1718, on chromosome 2BS in wheat. Euphytica 213:141
Zhao L, Tan L, Zhu Z, Xiao L, Xie D, Sun C (2015) PAY1 improves plant architecture and enhances grain yield in rice. Plant J 83(3):528–536
Zhao L, Wang C, Wang T, Liu J, Qiao Q, Yang Y, Hu P, Zhang L, Zhao S, Chen D, Ren Y, Zhang N, Dong Z, Chen F (2023a) Identification of the candidate gene controlling tiller angle in common wheat through genome-wide association study and linkage analysis. Crop J. https://doi.org/10.1016/j.cj.2023.01.004
Zhao L, Zheng Y, Wang Y, Wang S, Wang T, Wang C, Chen Y, Zhang K, Zhang N, Dong Z, Chen F (2023b) A HST1-like gene controls tiller angle through regulating endogenous auxin in common wheat. Plant Biotechnol J 21(1):122–135
Zheng S, Ye C, Lu J, Liufu J, Lin L, Dong Z, Li J, Zhuang C (2021) Improving the rice photosynthetic efficiency and yield by editing OsHXK1 via CRISPR/Cas9 system. Int J Mol Sci 22(17):9554
Zhou B, Serret MD, Elazab A, Bort Pie J, Araus JL, Aranjuelo I, Sanz-Sáez Á (2016) Wheat ear carbon assimilation and nitrogen remobilization contribute significantly to grain yield. J Integr Plant Biol 58(11):914–926
Funding
This work was funded by the National Natural Science Foundation (31971947 and 32072057), Henan Major Science and Technology Projects (201300110800 and 201300111600) and Key Scientific and Technological Project of Henan Province (212102110246 and 222102110026) of China.
Author information
Authors and Affiliations
Contributions
LZ designed the experiment and organized the manuscript. LZ, YY, PH, and QQ performed BSE-Seq analysis and identified the function of TaYL1 gene. GL, JL, LL, and JW investigated agronomic traits. YR and ZD provided experimental materials. FC participated in the supervision of the overall work and contributed to the finalization of the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, L., Yang, Y., Hu, P. et al. Genetic mapping and analysis of candidate leaf color genes in common winter wheat (Triticum aestivum L.). Mol Breeding 43, 48 (2023). https://doi.org/10.1007/s11032-023-01395-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11032-023-01395-z