Abstract
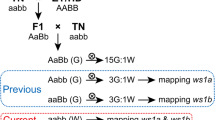
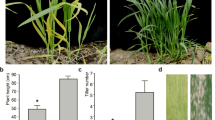
Leaf color is an indicator of chlorophyll (Chl) level, and isolating leaf color mutants can facilitate the understanding of Chl metabolism regulation. Here, we describe an ethyl methanesulfonate-induced light color mutant white stem 1 (ws1) in common tobacco (Nicotiana tabacum L.) that shows a phenotype highly similar to burley tobacco (Nicotiana tabacum L.), a type of air-cured tobacco that has light-colored leaves with white veins. Compared with the wild type, the light green stem of ws1 gradually became pale white along with growth, while ws1 leaves lost green color rapidly, which was positively correlated with the decline of Chl levels. A series of genetic analyses indicated that the ws1 mutant phenotype was controlled by two recessive nuclear genes ws1a and ws1b which were preliminarily mapped to the intervals of tobacco simple sequence repeat markers linkage groups 5 and 24 using the BC1F2 populations, respectively. The allelism test further revealed that the same two genes controlled the burley character in burley tobacco. Based on the Chl-deficient phenotype of ws1 and the locations of the two genes, we hypothesized that ws1a and ws1b were paralogs of each other probably originated from the ancestral species N. sylvestris and N. tomentosiformis, respectively. Both genes might share similar biological functions and expression patterns, and play key roles in the regulation of Chl biosynthesis. These results laid a solid foundation for marker-assisted selection breeding and gene function analysis of the burley character in tobacco.




Similar content being viewed by others
References
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15. doi:10.1104/pp.24.1.1
Babiychuk E, Muller F, Eubel H, Braun HP, Frentzen M, Kushnir S (2003) Arabidopsis phosphatidylglycerophosphate synthase 1 is essential for chloroplast differentiation, but is dispensable for mitochondrial function. Plant J 33(5):899–909. doi:10.1046/j.1365-313X.2003.01680.x
Barry CS, McQuinn RP, Chung M-Y, Besuden A, Giovannoni JJ (2008) Amino acid substitutions in homologs of the STAY-GREEN protein are responsible for the green-flesh and chlorophyll retainer mutations of tomato and pepper. Plant Physiol 147(1):179–187. doi:10.1104/pp.108.118430
Bassam BJ, Caetano-Anollés G, Gresshoff PM (1991) Fast and sensitive silver staining of DNA in polyacrylamide gels. Anal Biochem 196(1):80–83. doi:10.1016/0003-2697(91)90120-I
Beale SI (2005) Green genes gleaned. Trends Plant Sci 10(7):309–312. doi:10.1016/j.tplants.2005.05.005
Bindler G, Plieske J, Bakaher N, Gunduz I, Ivanov N, Van der Hoeven R, Ganal M, Donini P (2011) A high density genetic map of tobacco (Nicotiana tabacum L.) obtained from large scale microsatellite marker development. Theor Appl Genet 123(2):219–230. doi:10.1007/s00122-011-1578-8
Chen G, Bi YR, Li N (2005) EGY1 encodes a membrane-associated and ATP-independent metalloprotease that is required for chloroplast development. Plant J 41(3):364–375. doi:10.1111/j.1365-313X.2004.02308.x
Clausen RE, Cambron DR (1944) Inheritance in Nicotiana Tabacum. XVIII. Monosomic analysis. Genetics 29:447–477
Duncan EN, Pate JB (1967) Inheritance and use of golden crown virescence in cotton. J Hered 58:237–239
Gray JC, Sullivan JA, Wang JH, Jerome CA, MacLean D (2003) Coordination of plastid and nuclear gene expression. Philos Trans R Soc B Biol Sci 358(1429):135–144. doi:10.1098/rstb.2002.1180
Greene EA, Codomo CA, Taylor NE, Henikoff JG, Till BJ, Reynolds SH, Enns LC, Burtner C, Johnson JE, Odden AR, Comai L, Henikoff S (2003) Spectrum of chemically induced mutations from a large-scale reverse-genetic screen in Arabidopsis. Genetics 164(2):731–740
Henika FS (1932) The inheritance of the White Burley character in tobacco. J Agric Res 44:477–493
Henikoff S, Comai L (2003) Single-nucleotide mutations for plant functional genomics. Annu Rev Plant Biol 54:375–401. doi:10.1143/annurev.arplant.54.031902.135009
Jiang H, Li M, Liang N, Yan H, Wei Y, Xu X, Liu J, Xu Z, Chen F, Wu G (2007) Molecular cloning and function analysis of the stay green gene in rice. Plant J 52(2):197–209. doi:10.1111/j.1365-313X.2007.003221.x
Jung KH, Hur J, Ryu CH, Choi Y, Chung YY, Miyao A, Hirochika H, An GH (2003) Characterization of a rice chlorophyll-deficient mutant using the T-DNA gene-trap system. Plant Cell Physiol 44(5):463–472. doi:10.1093/pcp/pcg064
Keskitalo J, Bergquist G, Gardestrom P, Jansson S (2005) A cellular timetable of autumn senescence. Plant Physiol 139(4):1635–1648. doi:10.1104/pp.105.066845
Kim Y, Schumaker KS, Zhu J-K (2006) EMS mutagenesis of Arabidopsis. In: Salinas J, SanchezSerrano JJ (eds) Methods in molecular biology, vol 323. Humana Press Inc, 999 Riverview Dr, Ste 208, Totowa, NJ 07512-1165 USA, pp 101–103
Kurata N, Miyoshi K, Nonomura K, Yamazaki Y, Ito Y (2005) Rice mutants and genes related to organ development, morphogenesis and physiological traits. Plant Cell Physiol 46(1):48–62. doi:10.1093/pcp/pci506
Kusaba M, Ito H, Morita R, Iida S, Sato Y, Fujimoto M, Kawasaki S, Tanaka R, Hirochika H, Nishimura M, Tanaka A (2007) Rice NON-YELLOW COLORING1 is involved in light-harvesting complex II and grana degradation during leaf senescence. Plant Cell 19(4):1362–1375. doi:10.1105/tpc.106.042911
Larkin RM, Alonso JM, Ecker JR, Chory J (2003) GUN4, a regulator of chlorophyll synthesis and intracellular signaling. Science 299(5608):902–906. doi:10.1126/science.1079978
Lei S, Yao X, Yi B, Chen W, Ma C, Tu J, Fu T (2007) Towards map-based cloning: fine mapping of a recessive genic male-sterile gene (BnMs2) in Brassica napus L. and syntenic region identification based on the Arabidopsis thaliana genome sequences. Theor Appl Genet 115(5):643–651. doi:10.1007/s00122-007-0594-1
Leitch IJ, Hanson L, Lim KY, Kovarik A, Chase MW, Clarkson JJ, Leitch AR (2008) The ups and downs of genome size evolution in polyploid species of Nicotiana (Solanaceae). Ann Botany 101(6):805–814. doi:10.1093/aob/mcm326
Leon P, Arroyo A, Mackenzie S (1998) Nuclear control of plastid and mitochondrial development in higher plants. Ann Rev Plant Physiol Plant Mol Biol 49:453–480. doi:10.1146/annurev.arplant.49.1.453
Li H, Ribaut J-M, Li Z, Wang J (2008) Inclusive composite interval mapping (ICIM) for digenic epistasis of quantitative traits in biparental populations. Theor Appl Genet 116(2):243–260. doi:10.1007/s00122-007-0663-5
Li N, Jia J, Xia C, Liu X, Kong X (2013) Characterization and mapping of novel chlorophyll deficient mutant genes in durum wheat. Breed Sci 63(2):169–175. doi:10.1270/jsbbs.63.169
Lim KY, Matyasek R, Kovarik A, Leitch AR (2004) Genome evolution in allotetraploid Nicotiana. Biol J Linn Soc 82(4):599–606. doi:10.1111/j.1095-8312.2004.00344.x
Lim KY, Kovarik A, Matyasek R, Chase MW, Clarkson JJ, Grandbastien MA, Leitch AR (2007) Sequence of events leading to near-complete genome turnover in allopolyploid Nicotiana within five million years. New Phytol 175(4):756–763. doi:10.1111/j.1469-8137.2007.02121.x
Liu W, Fu Y, Hu G, Si H, Zhu L, Wu C, Sun Z (2007) Identification and fine mapping of a thermo-sensitive chlorophyll deficient mutant in rice (Oryza sativa L.). Planta 226(3):785–795. doi:10.1007/s00425-007-0525-z
Matile P (2000) Biochemistry of Indian summer: physiology of autumnal leaf coloration. Exp Gerontol 35(2):145–158. doi:10.1016/s0531-5565(00)00081-4
Mochizuki N, Brusslan JA, Larkin R, Nagatani A, Chory J (2001) Arabidopsis genomes uncoupled 5 (GUN5) mutant reveals the involvement of Mg-chelatase H subunit in plastid-to-nucleus signal transduction. Proc Natl Acad Sci USA 98(4):2053–2058. doi:10.1073/pnas.98.4.2053
Moulin M, McCormac AC, Terry MJ, Smith AG (2008) Tetrapyrrole profiling in Arabidopsis seedlings reveals that retrograde plastid nuclear signaling is not due to Mg-protoporphyrin IX accumulation. Proc Natl Acad Sci USA 105(39):15178–15183. doi:10.1073/pnas.0803054105
Nagata N, Tanaka R, Satoh S, Tanaka A (2005) Identification of a vinyl reductase gene for chlorophyll synthesis in Arabidopsis thaliana and implications for the evolution of Prochlorococcus species. Plant Cell 17(1):233–240. doi:10.1105/tpc.104.027276
Nakamura T, Yamamori M, Hirano H, Hidaka S, Nagamine T (1995) Production of waxy (amylose-free) wheats. Mol Gen Genet 248(3):253–259. doi:10.1007/bf02191591
Park S-Y, Yu J-W, Park J-S, Li J, Yoo S-C, Lee N-Y, Lee S-K, Jeong S-W, Seo HS, Koh H-J, Jeon J-S, Park Y-I, Paek N-C (2007) The senescence-induced staygreen protein regulates chlorophyll degradation. Plant Cell 19(5):1649–1664. doi:10.1105/tpc.106.044891
Pruzinska A, Tanner G, Anders I, Roca M, Hortensteiner S (2003) Chlorophyll breakdown: pheophorbide a oxygenase is a Rieske-type iron-sulfur protein, encoded by the accelerated cell death 1 gene. Proc Natl Acad Sci USA 100(25):15259–15264. doi:10.1073/pnas.2036571100
Rissler HM, Collakova E, DellaPenna D, Whelan J, Pogson BJ (2002) Chlorophyll biosynthesis. Expression of a second Chl I gene of magnesium chelatase in Arabidopsis supports only limited chlorophyll synthesis. Plant Physiol 128(2):770–779. doi:10.1104/pp0010625
Rzeznicka K, Walker CJ, Westergren T, Kannangara CG, von Wettstein D, Merchant S, Gough SP, Hansson M (2005) Xantha-l encodes a membrane subunit of the aerobic Mg-protoporphyrin IX monomethyl ester cyclase involved in chlorophyll biosynthesis. Proc Natl Acad Sci USA 102(16):5886–5891. doi:10.1073/pnas.0501784102
Sato Y, Morita R, Nishimura M, Yamaguchi H, Kusaba M (2007) Mendel’s green cotyledon gene encodes a positive regulator of the chlorophyll-degrading pathway. Proc Natl Acad Sci USA 104(35):14169–14174. doi:10.1073/pnas.0705521104
Sawers RJH, Viney J, Farmer PR, Bussey RR, Olsefski G, Anufrikova K, Hunter CN, Brutnell TP (2006) The maize Oil yellow1 (Oy1) gene encodes the I subunit of magnesium chelatase. Plant Mol Biol 60(1):95–106. doi:10.1007/s11103-005-2880-0
Sierro N, Battey JND, Ouadi S, Bovet L, Goepfert S, Bakaher N, Peitsch MC, Ivanov NV (2013) Reference genomes and transcriptomes of Nicotiana sylvestris and Nicotiana tomentosiformis. Genome Biol 14(6):R60. doi:10.1186/gb-2013-14-6-r60
Spano G, Di Fonzo N, Perrotta C, Platani C, Ronga G, Lawlor DW, Napier JA, Shewry PR (2003) Physiological characterization of ‘stay green’ mutants in durum wheat. J Exp Bot 54(386):1415–1420. doi:10.1093/jxb/erg150
Stines BJ, Mann TJ (1960) Diploidization in Nicotiana tabacum. J Hered 51:222–227
Terry MJ, Kendrick RE (1999) Feedback inhibition of chlorophyll synthesis in the phytochrome chromophore-deficient aurea and yellow-green-2 mutants of tomato. Plant Physiol 119(1):143–152. doi:10.1104/pp.119.1.143
Terry MJ, Wahleithner JA, Lagarias JC (1993) Biosynthesis of the plant photoreceptor phytochrome. Arch Biochem Biophys 306(1):1–15. doi:10.1006/abbi.1993.1473
Tong Z, Yang Z, Chen X, Jiao F, Li X, Wu X, Gao Y, Xiao B, Wu W (2012) Large-scale development of microsatellite markers in Nicotiana tabacum and construction of a genetic map of flue-cured tobacco. Plant Breed 131(5):674–680. doi:10.1111/j.1439-0523.2012.01984.x
Watanabe S, Mizoguchi T, Aoki K, Kubo Y, Mori H, Imanishi S, Yamazaki Y, Shibata D, Ezura H (2007) Ethylmethanesulfonate (EMS) mutagenesis of Solanum lycopersicum cv. Micro-Tom for large-scale mutant screens. Plant Biotechnol 24(1):33–38
Wu DX, Shen SQ, Cui HR, Xia YW, Shu QY (2003) A novel thermo/photoperiod-sensitive genic male-sterile (T/PGMS) rice mutant with green-revertible albino leaf color marker induced by gamma irradiation. Field Crops Res 81(2–3):141–147. doi:10.1016/s0378-4290(02)00218-6
Wu Z, Zhang X, He B, Diao L, Sheng S, Wang J, Guo X, Su N, Wang L, Jiang L, Wang C, Zhai H, Wan J (2007) A chlorophyll-deficient rice mutant with impaired chlorophyllide esterification in chlorophyll biosynthesis. Plant Physiol 145(1):29–40. doi:10.1104/pp.107.100321
Wu FN, Eannetta NT, Xu YM, Plieske J, Ganal M, Pozzi C, Bakaher N, Tanksley SD (2010) COSII genetic maps of two diploid Nicotiana species provide a detailed picture of synteny with tomato and insights into chromosome evolution in tetraploid N. tabacum. Theor Appl Genet 120(4):809–827. doi:10.1007/s00122-009-1206-z
Xu P, Lv Z, Zhang X, Wang X, Pu Y, Wang H, Yi B, Wen J, Ma C, Tu J, Fu T, Shen J (2013) Identification of molecular markers linked to trilocular gene (mc1) in Brassica juncea L. Mol Breed 1–10. doi:10.1007/s11032-013-9960-7
Yao FY, Xu CG, Yu SB, Li JX, Gao YJ, Li XH, Zhang QF (1997) Mapping and genetic analysis of two fertility restorer loci in the wild-abortive cytoplasmic male sterility system of rice (Oryza sativa L.). Euphytica 98(3):183–187. doi:10.1023/a:1003165116059
Yaronskaya E, Ziemann V, Walter G, Averina N, Borner T, Grimm B (2003) Metabolic control of the tetrapyrrole biosynthetic pathway for porphyrin distribution in the barley mutant albostrians. Plant J 35(4):512–522. doi:10.1046/j.1365-313X.2003.01825.x
Yi B, Chen YN, Lei SL, Tu JX, Fu TD (2006) Fine mapping of the recessive genic male-sterile gene (Bnms1) in Brassica napus L. Theor Appl Genet 113(4):643–650. doi:10.1007/s00122-006-0328-9
Acknowledgments
We thank Prof. Yongfeng Guo of Tobacco Research Institute, Chinese Academy of Agricultural Sciences for critically reading this manuscript. This work was supported by the China National Tobacco Company (Grant Nos. 110201201003 (JY-03) and 110201201004 (JY-04)).
Author information
Authors and Affiliations
Corresponding author
Additional information
Qingzhang Wu, Xinru Wu, and Xuefeng Zhang contributed equally to this article.
Rights and permissions
About this article
Cite this article
Wu, Q., Wu, X., Zhang, X. et al. Mapping of two white stem genes in tetraploid common tobacco (Nicotiana tabacum L.). Mol Breeding 34, 1065–1074 (2014). https://doi.org/10.1007/s11032-014-0097-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11032-014-0097-0