Abstract
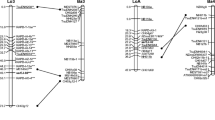
Loquat [Eriobotrya japonica (Thunb.) Lindl.] is a Rosaceae fruit species of growing interest as an alternative to the main fruit crops. However, only a few genetic studies have been carried out on this species. This paper reports the construction of the first genetic maps of two loquat cultivars based on AFLP and microsatellite markers from Malus, Eriobotrya, Pyrus and Prunus genera. An F1 population consisting of 81 individuals, derived from the cross between ‘Algerie’ and ‘Zaozhong-6’ cultivars, was used to construct both maps. A total of 111 scorable simple sequence repeat (SSR) loci resulted from the testing of 440 SSR primer pairs in the analyzed progeny and the SSR transferability to Eriobotrya was found to be 74% from apple, 58% from pear and 49% from Prunus spp. In addition, 183 AFLP polymorphic bands were produced using 42 primer combinations. The ‘Algerie’ map was organized in 17 linkage groups covering a distance of 900 cM and comprising 177 loci (83 SSRs and 94 AFLPs) with an average marker distance of 5.1 cM. Self-incompatibility trait was mapped at the distal part of the LG17 linkage group, as previously reported in Malus and Pyrus. The ‘Zaozhong-6’ map covered 870 cM comprising 146 loci (64 SSRs and 82 AFLPs) with an average marker distance of 5.9 cM. The 44 SSRs and the 48 AFLPs share in common by both maps were essentially collinear and, moreover, the order of the 75% of apple and pear SSRs mapped in Eriobotrya was shown to be consistent across the Maloideae subfamily. As a whole, these maps represent a useful tool to facilitate loquat breeding and an interesting framework for map comparison in the Rosaceae.



Similar content being viewed by others
References
Aranzana MJ, García-Mas J, Carbó J, Arús P (2002) Development and variability analysis of microsatellite markers in peach. Plant Breed 121:87–92. doi:10.1046/j.1439-0523.2002.00656.x
Bassam BJ, Caetano-Anollés G, Gresshoff PM (1991) Fast and sensitive silver staining of DNA in polyacrylamide gels. Anal Biochem 196:80–83. doi:10.1016/0003-2697(91)90120-I
Bratteler M, Lexer C, Widmer A (2006) A genetic linkage map of Silene vulgaris based on AFLP markers. Genome 49:320–327. doi:10.1139/G05-114
Campbell CS, Evans RC, Morgan DR, Dickinson TA, Arsenault MP (2007) Phylogeny of subtribe Pyrinae (formerly the Maloideae, Rosaceae): limited resolution of a complex evolutionary history. Plant Syst Evol 266:119–145. doi:10.1007/s00606-007-0545-y
Chevreau E, Lespinasse Y, Gallet M (1985) Inheritance of pollen enzymes and polyploid origin of apple (Malus x domestica Borkh.). Theor Appl Genet 71:268–277
Dirlewanger E, Cosson P, Tavaud M, Aranzana MJ, Poizat C, Zanetto A, Arús P, Laigret F (2002) Development of microsatellite markers in peach [Prunus persica (L.) Batsch] and their use in genetic diversity analysis in peach and sweet cherry (Prunus avium L.). Theor Appl Genet 105:127–138. doi:10.1007/s00122-002-0867-7
Dirlewanger E, Graziano E, Joobeur T, Garriga-Calderé F, Cosson P, Howad W, Arús P (2004) Comparative mapping and marker-assisted selection in Rosaceae fruit crops. Proc Natl Acad Sci USA 26:9891–9896. doi:10.1073/pnas.0307937101
Doyle JJ, Doyle JL (1987) A rapid isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Ellis JR, Burke JM (2007) EST-SSRs as a resource for population genetic analysis. Heredity 99:125–132. doi:10.1038/sj.hdy.6801001
Fernández-Fernández F, Evans KM, Clarke JB, Govan CL, James CM, Maric S, Tobutt KR (2008) Development of an STS map of an interspecific progeny of Malus. Tree Genet Genomes 4:469–479. doi:10.1007/s11295-007-0124-y
Gianfranceschi L, Seglias N, Tarchini R (1998) Simple sequence repeats for the genetic analysis of apple. Theor Appl Genet 96:1069–1076. doi:10.1007/s001220050841
Gisbert AD, López-Capuz I, Soriano JM, Llácer G, Romero C, Badenes ML (2008) Development of microsatellite markers from loquat (Eriobotrya japonica (Thunb.) Lindl.). Mol Ecol Res (in press)
Grattapaglia D, Sederoff RR (1994) Genetic linkage maps of Eucalyptus grandis and E. urophylla using a pseudotest-cross strategy and RAPD markers. Genetics 137:1121–1137
Guilford P, Prakash S, Zhu JM, Rikkerink E, Gardiner S, Basset H, Forster R (1997) Microsatellites in Malus x domestica (apple): abundance, polymorphism and cultivar identification. Theor Appl Genet 94:249–254. doi:10.1007/s001220050407
Hemmat M, Weeden NF, Manganaris AG, Lawson DM (1994) Molecular marker linkage map for apple. J Hered 85:4–11
Hokanson SC, Szewc-McFadden AK, Lamboy WF, McFerson JR (1998) Microsatellite (SSR) markers reveal genetic identities, genetic diversity and relationships in a Malus x domestica Borkh. core subset collection. Theor Appl Genet 97:671–683. doi:10.1007/s001220050943
Howad W, Yamamoto T, Dirlewanger E, Testolin R, Cosson P, Cipriani G, Monforte AJ, Georgi L, Abbott AG, Arús P (2005) Mapping with a few plants: using selective mapping for microsatellite saturation of the Prunus reference map. Genetics 171:1305–1309. doi:10.1534/genetics.105.043661
Huang JS, Xu XT, Fang JQ (1990) A new, stable, productive loquat variety, ‘Changhong No. 3’. China Fruits 1990(2):26–27
Kenis K, Keulemans J (2005) Genetic linkage maps of two apple cultivars (Malus x domestica Borkh.) based on AFLP and microsatellite markers. Mol Breed 15:205–219. doi:10.1007/s11032-004-5592-2
Kosambi DD (1944) The estimation of map distance from recombination values. Ann Eugen 12:172–175
Liebhard R, Gianfranceschi L, Koller B, Ryder CD, Tarchini R, Van de Weg E, Gessler C (2002) Development and characterisation of 140 new microsatellites in apple (Malus x domestica Borkh.). Mol Breed 10:217–241. doi:10.1023/A:1020525906332
Liebhard R, Koller B, Gianfrancechi L, Gessler C (2003) Creating a saturated reference map for the apple (Malus x domestica Borkh.) genome. Theor Appl Genet 106:1497–1508
Lin S, Sharpe RH, Janick J (1999) Loquat: botany and horticulture. In: Janick J (ed) Horticultural reviews, vol 23. Wiley, New York, pp 233–276
Llácer G, Martínez-Calvo J, Gisbert AD, Romero C, Badenes ML (2008) Del germoplasma a la mejora genética: el caso del níspero japonés. Actas Horticultura (in press)
Lopes MS, Sefc KM, Laimer M, Machado Da Câmara (2002) Identification of microsatellite loci in apricot. Mol Ecol Notes 2:24–26. doi:10.1046/j.1471-8286.2002.00132.x
Lu LX, Lin SQ (eds) (1995) An introduction on reproductive biology in fruit trees. China Agr Press, Beijing (in Chinese)
Maliepaard C, Alston FH, van Arkel G, Brown LM, Chevreau E, Dunemann Evans KM, Gardiner S, Guilford P, van Heusden AW, Janse J, Laurens F, Lynn JR, Manganaris AG, den Nijs APM, Periam N, Rikkerink E, Roche P, Ryder C, Sansavini S, Schmidt H, Tartarini S, Verhaegh JJ, Vrielink-van Ginkel M, King GJ (1998) Aligning male and female linkage maps of apple (Malus pumila Mill.) using multi-allelic markers. Theor Appl Genet 97:60–73. doi:10.1007/s001220050867
Martínez-Calvo J, Badenes ML, Llácer G (2000) Descripción de variedades de níspero japonés. Serie de divulgación técnica n 47. Consellería de Agricultura. Pesca y Alimentación de la Generalitat Valenciana, Valencia
Mnejja M, García-Mas J, Howad W, Badenes ML, Arús P (2004) Simple-sequence repeat (SSR) markers of Japanese plum (Prunus salicina Lindl.) are highly polymorphic and transferable to peach and almond. Mol Ecol Notes 4:163–166. doi:10.1111/j.1471-8286.2004.00603.x
Mnejja M, García-Mas J, Howad W, Arús P (2005) Development and transportability across Prunus species of 42 polymorphic almond microsatellites. Mol Ecol Notes 5:531–535. doi:10.1111/j.1471-8286.2005.00977.x
Morton JF (1987) Loquat. In: Fruits of warm climate. Creative Resource Systems, Winterville, pp 103–108
Pierantoni L, Cho KH, Shin LS, Chiodini R, Tartarini S, Dondini L, Kang SJ, Sansavini S (2004) Characterisation and transferability of apple SSRs to two European pear F1 populations. Theor Appl Genet 109:1519–1524. doi:10.1007/s00122-004-1775-9
Raspé O, Kohn JK (2002) S-allele diversity in Sorbus aucuparia and Crataegus monogyna (Rosaceae: Maloideae). Heredity 88:458–465. doi:10.1038/sj.hdy.6800079
Rodríguez A (1983) El cultivo del níspero en el Valle del Algar-Guadalest. Sociedad Cooperativa de Crédito de Callosa de Ensarriá, Alicante
Silfverberg-Dilworth E, Matasci CL, Van de Weg WE, Van Kaauwen MPW, Walser M, Kodde LP, Soglio V, Gianfranceschi L, Durel CE, Costa F, Yamamoto T, Koller B, Gessler C, Patocchi A (2006) Microsatellite markers spanning the apple (Malus x domestica Borkh.) genome. Tree Genet Genomes 2:202–224. doi:10.1007/s11295-006-0045-1
Soriano JM, Romero C, Vilanova S, Llácer G, Badenes ML (2005) Genetic diversity of loquat germplasm (Eriobotrya japonica (Thunb.) Lindl.) assessed by SSR markers. Genome 48:108–114. doi:10.1139/g04-101
Sosinski B, Gannavarapu M, Hager LD, Beck LE, King GJ, Ryder CD, Rajapakse S, Baird WV, Ballard RE, Abbott AG (2000) Characterization of microsatellite markers in peach. Theor Appl Genet 101:421–428. doi:10.1007/s001220051499 (Prunus persica (L.) Batsch)
Van Ooijen JW, Voorrips RE (2001) JoinMap®3.0 Software for the calculation of genetic linkage maps. Plant Research International, Wageningen
Vinatzer BA, Patocchi A, Tartarini S, Gianfranceschi L, Sansavini S, Gessler C (2004) Isolation of two microsatellite markers from BAC clones of the Vf scab resistance region and molecular characterization of scab accessions in Malus germplasm. Plant Breed 123:321–326. doi:10.1111/j.1439-0523.2004.00973.x
Vos P, Hogers R, Bleeker M, Reijans M, Van de Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M, Zabeau M (1995) AFLP: a new technique for DNA fingerprinting. Nucleic Acids Res 23:4407–4414. doi:10.1093/nar/23.21.4407
Yamamoto T, Kimura T, Sawamura Y, Kotobuki K, Ban Y, Hayashi T, Matsuta N (2001) SSRs isolated from apple can identify polymorphism and genetic diversity in pear. Theor Appl Genet 102:865–870. doi:10.1007/s001220000524
Yamamoto T, Kimura T, Shoda M, Ban Y, Hayashi T, Matsuta N (2002a) Development of microsatellite markers in the Japanese pear (Pyrus pyrifolia Nakai). Mol Ecol Notes 2:14–16. doi:10.1046/j.1471-8286.2002.00128.x
Yamamoto T, Kimura T, Shoda M, Imai T, Saito T, Sawamura Y, Kotobuki K, Hayashi T, Matsuta N (2002b) Genetic linkage maps constructed by using an interspecific cross between Japanese and European pears. Theor Appl Genet 106:9–18
Yamamoto T, Kimura T, Sawamura Y, Manabe T, Kotobuki K, Hayashi T, Ban Y, Matsuta N (2002c) Simple sequence repeats for genetic analysis in pear. Euphytica 124:129–137. doi:10.1023/A:1015677505602
Yamamoto T, Mochida K, Imai T, Shi YZ, Ogiwara I, Hayashi T (2002d) Microsatellite markers in peach [Prunus persica (L.) Batsch] derived from an enriched genomic and cDNA libraries. Mol Ecol Notes 2:298–301. doi:10.1046/j.1471-8286.2002.00242.x
Yamamoto T, Kimura T, Saito T, Kotobuki K, Matsuta N, Liebhard R, Gessler C, Van de Weg WE, Hayashi T (2004) Genetic linkage maps of Japanese and European pears aligned to the apple consensus map. Acta Hortic 663:51–56
Zhang HZ, Peng SA, Cai LH, Fang DQ (1990) The germplasm resources of the genus Eriobotrya with special reference on the origin of E. japonica Lindl. Acta Hortic Sin 17:5–12
Acknowledgments
The IVIA germplasm collection is funded by a grant from the Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria (INIA RF2004-017) and this work was also partly supported by the growers association from Callosa d’en Sarrià (Alicante, Spain). The authors also thank Laura Ramirez and Gregoire Marandel (INRA-Bordeaux, France) for their technical contributions. A. D. G. was funded by a fellowship from the IVIA. All the experiments described in this paper comply with the current laws of Spain.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gisbert, A.D., Martínez-Calvo, J., Llácer, G. et al. Development of two loquat [Eriobotrya japonica (Thunb.) Lindl.] linkage maps based on AFLPs and SSR markers from different Rosaceae species. Mol Breeding 23, 523–538 (2009). https://doi.org/10.1007/s11032-008-9253-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11032-008-9253-8