Abstract
Sirtuin3 (SIRT3) is a deacetylase that plays an important role in normal physiological activities by regulating a variety of substrates. Considerable evidence has shown that the content and activity of SIRT3 are altered in neurological diseases. Furthermore, SIRT3 affects the occurrence and development of neurological diseases. In most cases, SIRT3 can inhibit clinical manifestations of neurological diseases by promoting autophagy, energy production, and stabilization of mitochondrial dynamics, and by inhibiting neuroinflammation, apoptosis, and oxidative stress (OS). However, SIRT3 may sometimes have the opposite effect. SIRT3 can promote the transfer of microglia. Microglia in some cases promote ischemic brain injury, and in some cases inhibit ischemic brain injury. Moreover, SIRT3 can promote the accumulation of ceramide, which can worsen the damage caused by cerebral ischemia–reperfusion (I/R). This review comprehensively summarizes the different roles and related mechanisms of SIRT3 in neurological diseases. Moreover, to provide more ideas for the prognosis of neurological diseases, we summarize several SIRT3-mediated rehabilitation training methods.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Sirtuins family contains seven members, which are marked as SIRT1 to SIRT7 (Chang and Guarente 2014; Imai and Guarente 2014; Kane and Sinclair 2018). The members of this family participate in a variety of physiological activities and pathological reactions by causing different types of post-translational modifications of various substrate proteins (Kida and Goligorsky 2016; Morigi et al. 2018). The location of each type of sirtuin in the cell is not the same. SIRT2 is mainly located in the cytoplasm; SIRT1, SIRT6, and SIRT7 are mainly located in the nucleus; and SIRT3, SIRT4, and SIRT5 are mainly located in mitochondria (Chen et al. 2021a, b). As a deacetylase located in mitochondria, SIRT3 must have a relationship with mitochondrial function. According to the available literature, SIRT3 can directly regulate about 100 downstream proteins, and most of the substrate proteins are activated by SIRT3 deacetylation (Yang et al. 2016). The related physiological functions include regulation of material metabolism, maintenance of mitochondrial stability, apoptosis, and autophagy (Zhang et al. 2020a, b, c, d).
At present, neurological diseases are increasingly affecting the quality of life of the elderly, and mitochondrial function is closely related to neurological diseases (Carrì et al. 2018; Chan 2020; Todorova and Blokland 2017). The current research mainly concentrates on SIRT1 and SIRT2, and there are relatively few studies on SIRT3. Therefore, in this review, we summarize the different roles and related mechanisms of SIRT3 in neurological diseases and summarize several rehabilitation training methods related to SIRT3 that can improve the prognosis of neurological diseases.
Deacetylation activity and function of SIRT3
SIRT3 and substance metabolism
The main function of SIRT3 is to participate in the regulation of substance metabolism (Sidorova-Darmos et al. 2018) (Table 1). Normal material metabolism maintains the stability of the body, and its main function is to provide energy for life activities and generate metabolic wastes that are easy to eliminate through biotransformation.
Glucose metabolism, lipid metabolism, and oxidative phosphorylation (OXPHOS) provide most of the energy required for life activities. During anaerobic oxidation of sugars, SIRT3 deacetylates cyclophilin D (CypD) (Wei et al. 2013a, b) and lactate dehydrogenase A (LDHA) (Cui et al. 2015), which in turn inhibit glycolysis and promote lactate production, respectively. During aerobic oxidation of sugars, SIRT3 can deacetylate and activate mitochondrial pyruvate carrier 1 (MPC1) (Liang et al. 2015) and pyruvate dehydrogenase (PDH) (Jing et al. 2013), which promote the entry of pyruvate into mitochondria and the production of acetyl-CoA, respectively. The activation of pyruvate dehydrogenase phosphatase 1 (PDP1) by SIRT3 can also activate PDH (Fan et al. 2014). SIRT3 promotes aerobic oxidation by activating multiple key enzymes in the tricarboxylic acid cycle (TCA) cycle, such as citrate synthase (CS) (Cui et al. 2017), isocitrate dehydrogenase 2 (IDH2) (Smolková et al. 2020), and succinate dehydrogenase (SDH) (Li et al. 2016a, b); in contrast, deacetylation of aconitase (Aco) by SIRT3 inhibits its activity and thus inhibits aerobic oxidation (Fernandes et al. 2015).
When the body is in a state of starvation, energy generated by fatty acid metabolism compensates for the lack of energy caused by insufficient sugar supply and maintains the body's homeostasis. First, acyl-CoA synthase family member 3 (ACSF3) is involved in the activation of fatty acids to produce acyl-CoA (Sloan et al. 2011), which is transported to mitochondria under the action of carnitine/acylcarnitine transporter (CACT). The deacetylation activation of ACSF3 and CACT by SIRT3 promotes this process (Giangregorio et al. 2017; Sun et al. 2020). Acyl-CoA in mitochondria undergoes β-oxidation to generate acetyl-CoA, which in turn participates in the TCA cycle. SIRT3 deacetylates and activates long-chain acyl-CoA dehydrogenase (LCAD) (Hirschey et al. 2010), very long-chain acyl-CoA dehydrogenase (VLCAD) (Zhang et al. 2015), β-hydroxyacyl-CoA dehydrogenase (β-HAD) (Alrob et al. 2014), and enoyl-CoA hydratase-1 (ECHS1) (Zhang et al. 2017) during β-oxidation. In addition, SIRT3 can activate acetyl-CoA carboxylase (ACC1) to promote fatty acid synthesis (Xu et al. 2020), which is a beneficial way of energy storage when nutrients are plentiful.
OXPHOS is the process by which electron transfer chain (ETC) converts the metabolites NADH and FADH2 to ATP (Nolfi-Donegan et al. 2020). There are five complexes involved in this process, which are labeled as complexes I to V. SIRT3 can deacetylate and activate the NADH dehydrogenase (ubiquinone) 1 alpha subcomplex 9 (NDUFA9) subunit on complex I (Ahn et al. 2008), succinate dehydrogenase flavoprotein (SDHA) subunit on complex II (Cimen et al. 2010), ubiquinol cytochrome c reductase core protein 1 (UQCRQ) subunit on complex III (Sun et al. 2019), cytochrome c oxidase-1 (COX-1) subunit on complex IV (Tu et al. 2019), and ATP synthase β subunit on complex V (Rahman et al. 2014), hereby promoting the generation of ATP. Furthermore, deacetylation of p53 by SIRT3 inhibits the expression-repressive effect of p53 on ND2 and ND4 genes, which encode key subunits of complex I (Lee et al. 2018). SIRT3 also activates leucine-containing protein 130 (LRP130), which promotes the process of OXPHOS (Liu et al. 2014). However, it has been reported that deacetylation of ceramide synthases 1 (CerS1), CerS2, and CerS6 by SIRT3 promotes ceramide accumulation and inhibits complex III activity (Novgorodov et al. 2016).
Amino acid metabolism can also generate small amounts of energy, and SIRT3 can deacetylate and activate glutamate dehydrogenase (GDH) (Choi et al. 2016). This enzyme facilitates the conversion of glutamate to alpha-ketoglutarate, which enters the TCA cycle for energy production. This process produces the toxic metabolite ammonia (NH3), and the urea cycle is the main metabolic route of intracellular NH3. Activation of ornithine transcarbamoylase (OTC) (Hallows et al. 2011) and carbamoyl phosphate synthase 1 (CPS1) (Li et al. 2016a, b) by SIRT3 in the urea cycle can promote urea synthesis and inhibit the toxic effects of NH3 on the body.
SIRT3 also has a partial effect on the metabolism of nonnutrients. SIRT3 can deacetylate and inhibit aldehyde dehydrogenase 2 (ALDH2) activity and thereby inhibit the conversion of acetaldehyde to acetate (Wei et al. 2013a, b), which may cause acetaldehyde toxicity. SIRT3 can also deacetylate acetyl-CoA synthase 2 (AceCS2) to convert acetate to acetyl-CoA (Hallows et al. 2006), and then participate in the TCA cycle.
SIRT3 and mitochondrial dynamics
Mitochondrial dynamics include two groups of events: mitochondrial fusion and mitochondrial fission, and mitochondrial biogenesis and mitochondrial degradation, including mitophagy (Meyer et al. 2017). Together, these four processes maintain the stability of mitochondrial number, shape, and function in cells (Forte et al. 2021). SIRT3 regulates the activity of key enzymes in mitochondrial dynamics through deacetylation, thereby regulating the stabilization of mitochondrial function in cells.
Optic atrophy 1 (Opa1) and mitofusin (Mfn) are major proteins involved in mitochondrial fusion, namely in promoting fusion of the inner and outer mitochondrial membranes, respectively. There are two isoforms of Mfn, namely Mfn1 and Mfn2 (van der Bliek et al. 2013). SIRT3 can directly deacetylate Opa1 at lysine 926 and 931 and promote its activity (Samant et al. 2014). However, SIRT3 does not directly deacetylate Mfn, but it can deacetylate and activate liver kinase B1 (LKB1) (Pillai et al. 2010), thereby promoting mitochondrial fusion through the LKB1–AMP activated protein kinase (AMPK)–Mfn pathway (Dong et al. 2019; Fan et al. 2019; Wu et al. 2020a, b, c). The proteins involved in mitochondrial fission are mainly dynamin related protein 1 (DRP1) and mitochondrial fission protein 1 (Fis1). There is no direct deacetylation of these two proteins by SIRT3, and deacetylation of forkhead box O3a (FOXO3a) by SIRT3 can upregulate the expression levels of DRP1 and Fis1, which are involved in the regulation of mitochondrial dynamics (Tseng et al. 2013). However, one experiment has shown that reduced levels of SIRT3 in brain tissue do not affect DRP1 expression, but promote DRP1 phosphorylation and translocation from the cytoplasm to mitochondria, thereby promoting mitochondrial fission (Park et al. 2020). This is most likely mediated by the LKB1–AMPK pathway (Mao et al. 2022; Xue et al. 2019).
Peroxisome proliferator-activated receptor gamma coactivator-1alpha (PGC-1α) is a core protein that causes mitochondrial biogenesis, which can be positively regulated by the LKB1–AMPK–PGC-1α pathway, and participates in mitochondrial biogenesis through the PGC-1α– nuclear respiratory factor (NRF)–transcription factor A (TFAM) pathway; thus, deacetylation modification of LKB1 by SIRT3 also promotes mitochondrial biogenesis (Fu et al. 2012; Li et al. 2017). TFAM can activate RNA polymerase in mitochondria and directly promote transcription of the genome in mitochondria. In kidney tumor cells, SIRT3 deacetylates and activates TFAM, which in turn promotes mitochondrial biogenesis (Liu et al. 2018a, b, c). E3 ubiquitin ligase Parkin-mediated mitophagy is important for the maintenance of mitochondrial homeostasis and is an important clearance mechanism for mitochondria. This process is regulated by FOXO3a, and SIRT3 is one of the important regulatory enzymes of FOXO3a. FOXO3a is activated upon deacetylation and promotes Parkin expression (Ma et al. 2018; Yu et al. 2017).
SIRT3 and oxidative stress
OS is an intracellular damage state, which is closely related to the occurrence and development of many diseases. Excessive intracellular production of reactive oxygen species (ROS) is an important cause of OS. The abnormal working state of the oxidative respiratory chain causes a large amount of electron leakage, which is one of the important reasons for the excess of ROS. The effects of SIRT3 on the five complexes in the oxidative respiratory chain can affect the production of ROS (Ahn et al. 2008; Cimen et al. 2010; Lee et al. 2018; Novgorodov et al. 2016; Sun et al. 2019; Tu et al. 2019). Impaired ROS scavenging is another cause of OS, and endogenous antioxidant enzymes can scavenge superoxide in cells to inhibit OS. SIRT3 directly regulates the activities of antioxidant enzymes such as manganese superoxide dismutase (MnSOD) (Qiu et al. 2010), catalase (CAT) (Wang et al. 2014), and peroxiredoxin3 (PRDX3) (Wang et al. 2020a, b) through deacetylation, thereby inhibiting OS. SIRT3 also indirectly activates MnSOD by mediating the deacetylation of FOXO3a and forkhead box O1 (FOXO1), promoting ROS scavenging (Tseng et al. 2014; Zhang et al. 2013). In addition, deacetylated FOXO3a also promotes CAT activity (Zhang et al. 2018a, b). ROS scavenging by glutathione peroxidase (Gpx) can also be enhanced by its deacetylation by SIRT3 (Yoon and Kim 2016). Glutathione (GSH) acts as a cofactor for Gpx to scavenge ROS, while nicotinamide adenine dinucleotide phosphate (NADPH) is a necessary factor to generate GSH. Deacetylation activation of IDH2 (Someya et al. 2010) and mitochondrial methylenetetrahydrofolate dehydrogenase/cyclohydrolase (MTHFD2) (Wan et al. 2020) by SIRT3 can promote NADPH production, thereby inhibiting OS. However, one study came to the opposite conclusion; i.e., that deacetylation of glutamate oxaloacetate transaminases2 (GOT2) by SIRT3 inhibits NADPH production, which in turn leads to tumor cell death under conditions of OS (Yang et al. 2015a, b).
SIRT3 and apoptosis
The relationship between SIRT3 and apoptosis has only been explored in a few experiments. In general, inhibition of OS by SIRT3 can inhibit the occurrence of apoptosis. In addition, SIRT3 can regulate apoptosis by regulating the deacetylation of the following proteins. First, activation of glycogen synthase kinase 3β (GSK-3β) by SIRT3 can promote the expression of BCL2-associated protein X (Bax), which in turn promotes apoptosis (Song et al. 2016). he inhibitory effect of SIRT3 on apoptosis can be achieved by activating 8-oxoguanine DNA glycosylase 1 (OGG1) (Cheng et al. 2013), Ku70 (Sundaresan et al. 2008), and 17-β-hydroxysteroid dehydrogenase 10 (HSD17B10) (Liu et al. 2020a, b, c), and inhibiting the activity of CypD (Liu et al. 2018a, b, c). OGG1 can repair DNA damage caused by OS; Ku70 can inhibit Bax-induced apoptosis; both the increased activity of HSD17B10 and the decreased activity of CypD inhibit mitochondrial dysfunction.
SIRT3 and autophagy
Autophagy is a highly conserved eukaryotic cell cycle process. Subcellular structures such as organelles are degraded under the action of certain mechanisms to form decomposition products that can be recycled. Autophagy thus plays an important role in maintaining cellular homeostasis (Parzych and Klionsky 2014). In addition to regulating mitophagy by regulating the activity of FOXO3a, SIRT3 can also regulate autophagy through the LKB1–AMPK–mammalian/mechanistic target of the rapamycin (mTOR) axis. mTOR inhibits autophagy in general, and activated AMPK can inhibit mTOR activity, so deacetylation of LKB1 by SIRT3 can promote autophagy (Zhang et al. 2018a, b). The Atg12–Atg5–Atg16 complex is an important regulatory system during autophagy (Yang and Klionsky 2009), and deacetylation of ATG5 by SIRT3 ensures autophagosome maturation (Liu et al. 2018a, b, c).
Chronic neurodegenerative diseases
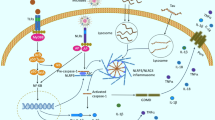
Chronic neurological diseases are closely related to age and mostly occur in the elderly. In this section, we mainly focus on Alzheimer's disease (AD), Parkinson's disease (PD), Huntington's disease (HD) and amyotrophic lateral sclerosis (ALS) (Fig. 1).
SIRT3 and chronic neurodegenerative diseases. The occurrence and development of chronic neurodegenerative diseases are related to the accumulation of intracellular toxic substances, programmed cell death and mitochondrial homeostasis. SIRT3 activates a range of substrates by deacetylation to reduce intracellular Tau content, promote energy production, inhibit OS and apoptosis, and promote autophagy. In addition, SIRT3 also maintains stable mitochondrial dynamics. The green proteins represent the substrates of SIRT3. Brown circles represent acetyl group
SIRT3 and Alzheimer's disease
AD is a very common chronic neurodegenerative disease characterized by memory impairment, executive dysfunction, and personality and behavior abnormalities, which seriously affect the quality of life of the elderly in the late stage. The etiology of AD is very complex and has not yet been fully elucidated. OS, neuronal apoptosis, inflammation, and cellular senescence can all promote the occurrence and development of AD.
It has been found that the level of SIRT3 in mitochondria of the cerebral cortex of AD mice is decreased (Yang et al. 2015a, b) and that the level of SIRT3 mRNA in the brain tissue of AD patients is reduced (Song et al. 2020). These results indicate that the presence of SIRT3 may be very important to inhibit the occurrence and development of AD. In the early stage of AD, there is an excessive accumulation of Tau protein in the brain tissue (Li et al. 2019). However, SIRT3 can deacetylate Tau and reduce its protein content (Li et al. 2019; Yin et al. 2018), which may help inhibit the further development of AD (Villemagne et al. 2015). SIRT3 can also inhibit the acetylation level of p53, thereby restoring the normal expression levels of ND2 and ND4 genes in the brain tissue. This is vital for maintaining the normal progress of mitochondrial OXPHOS and inhibiting AD caused by neuronal damage (Lee et al. 2018). Reducing the content of ROS in the cell may be an effective method to treat AD. One of the most important substrates of SIRT3 is MnSOD, and the activity of MnSOD is enhanced after deacetylation. MnSOD can convert excessive ROS into hydrogen peroxide (H2O2). H2O2 is converted into harmless oxygen and water under the action of CAT. This function of SIRT3 has been confirmed in multiple experiments (Li et al. 2020; Liu et al. 2020a, b, c; Ramesh et al. 2018). SIRT3 can promote the deacetylation and activity of 17-β-hydroxysteroid dehydrogenase 10 (HSD17B10), thereby promoting the antiapoptotic ability of cells (Liu et al. 2020a, b, c). Although there is currently no evidence that the effect of SIRT3 on HSD17B10 can inhibit AD, the inhibition of HSD17B10 activity by amyloid-β (Aβ) can lead to neuronal dysfunction associated with AD (Oppermann et al. 1999; Yan et al. 1997). Therefore, it is credible that HSD17B10 mediates the inhibitory effect of SIRT3 on AD. SIRT3 can also inhibit neuronal apoptosis by inhibiting the expression of CypD, and thus alleviates the symptoms of AD (Jiang et al. 2017). In addition, the inhibitory effect of SIRT3 on AD can be achieved by promoting the canonical autophagy pathway LKB1–AMPK–mTOR (Shu et al. 2020; Zhang et al. 2020a, b, c, d).
SIRT3 and Parkinson’s disease
Among neurodegenerative diseases, PD is the second by incidence (Risiglione et al. 2021; Tysnes and Storstein 2017). PD is characterized by progressive degeneration of dopaminergic neurons in the substantia nigra striatum (Liu et al. 2015). The main clinical manifestations of the disease are motor dysfunction including tremors at rest, rigidity, postural instability, and bradykinesia (Huang et al. 2021; Li et al. 2021a, b; Tolosa et al. 2006).
In several chemical drug trials, it has been found that SIRT3 agonists such as theacrine (Duan et al. 2020) and trans-(-)-ε-viniferin (ε-viniferin) (Zhang et al. 2020a, b, c, d) can inhibit the occurrence and development of PD. When SIRT3 inhibitors such as dipeptidyl peptidase-4 (DPP-4), P2X7 purinoceptors (P2X7R) (Jamali-Raeufy et al. 2020), and miR-494-3p (Geng et al. 2018) are inhibited, PD and related clinical symptoms improve. Hence, SIRT3 has a positive effect on PD inhibition.
In current research, the inhibitory effect of SIRT3 on PD is mainly achieved by inhibiting the accumulation of ROS in cells and consequent inhibition of OS (Dai et al. 2014), which is similar to the effect of SIRT3 on AD. SIRT3 can promote the activity of MnSOD (Jiang et al. 2019; Zhang et al. 2016) as well as the expression level of MnSOD in cells by promoting the deacetylation level and activity of FOXO3a (Rangarajan et al. 2015). FOXO3a can also promote the expression of CAT (Rangarajan et al. 2015). Therefore, under this series of actions, SIRT3 strongly eliminates the content of ROS in neuronal cells. In addition, it has been shown that the promotion of autophagy by SIRT3 can inhibit the occurrence and development of PD (Zhang et al. 2018a, b). SIRT3 can increase the deacetylation level and activity of LKB1 (Woods et al. 2003), which in turn promotes the phosphorylation level and activity of AMP activated protein kinase (AMPK) (Pillai et al. 2010) and inhibits the activity of mTOR, thereby promoting autophagy (Jung et al. 2010). SIRT3 also promotes the activities of ATP synthase β (Zhang et al. 2016) and CS (Cui et al. 2017) to promote OXPHOS and the TCA cycle, respectively. The resulting large amount of energy inhibits neuronal damage caused by insufficient energy. Recent studies have shown that SIRT3's inhibition of DRP1 phosphorylation can maintain normal mitochondrial dynamics, which can positively regulate the treatment and prognosis of PD (Park et al. 2020).
SIRT3 and Huntington's disease
Unlike AD and PD, the incidence of HD is low and the pathogenesis is clearer. The disease is an autosomal dominant genetic disease (Wild and Tabrizi 2017). The mutated chromosomes can cause the neurotoxic mutant huntingtin (mHTT) to accumulate in the body (Ross and Tabrizi 2011). The most common clinical sign of the disease is dyskinesia, and patients often show weird dance-like movements (Jimenez-Sanchez et al. 2017). In addition, many patients have different levels of psychiatric symptoms, such as depression and obsessive–compulsive disorder (Rosenblatt 2007). This may be caused by the strange behavior, or it may be caused by the organic disease directly caused by mHTT.
At present, treatments for HD related to SIRT3 mostly focus on how to maintain the normal mitochondrial biogenesis because abnormal mitochondrial biogenesis is one of the important factors in the development of HD. After SIRT3 is activated, the LKB1–AMPK pathway is also activated and enhances mitochondrial biogenesis (Fu et al. 2012). Moreover, AMPK can also promote the activity of SIRT3 by increasing the NAD+/NADH ratio, thereby forming a positive feedback pathway (Duan et al. 2016). In addition, the inhibitory effect of SIRT3 on ROS can inhibit the damage of mHTT to neurons (Fu et al. 2012). It has also been shown that the ability of SIRT3 to inhibit the accumulation of DRP1 in mitochondria and thus inhibit mitochondrial fission contributes to mitochondrial elongation, which in turn promotes cell viability (Naia et al. 2021; Oliver and Reddy 2019). Although there are few related studies, the agonist of SIRT3 used in the above two studies is viniferin, and viniferin has a good inhibitory effect on HD.
SIRT3 and amyotrophic lateral sclerosis
ALS is a neurodegenerative disease for which the cause has not yet been identified. The disease is characterized by the degeneration of motor neurons (MN) in the brain and spinal cord, and the resulting muscle dysfunction such as limb weakness and difficulty swallowing (Taylor et al. 2016). Most patients eventually die from respiratory failure caused by respiratory muscle dysfunction (Brown and Al-Chalabi 2017). Although the incidence of the disease is relatively low, patients endure great pain during the illness. Therefore, it is necessary to improve the clinical symptoms and cure the disease.
At present, the relationship between SIRT3 and ALS has been relatively poorly studied. However, since mitochondrial morphology is altered in ALS disease models (Magrané et al. 2009; Sasaki and Iwata 2007), inhibition of mitochondrial fission by SIRT3 may inhibit the development of ALS (Song et al. 2013). CypD inhibition by SIRT3 prevents the transition of mitochondrial permeability and thus inhibits apoptosis, which also plays an important role in the inhibition of ALS (Song et al. 2013). MN of ALS patients has specific metabolic characteristics such as reduced mitochondrial respiration and elevated glycolysis. Therefore, the reversal of the clinical phenotype of ALS by SIRT3 agonists is inseparable from the function of SIRT3 to promote OXPHOS (Hor et al. 2021). This is one of the reasons why SIRT3 agonists are the preferred treatment strategy for ALS in some treatment regimens (Harlan et al. 2020).
Acute neurodegenerative diseases
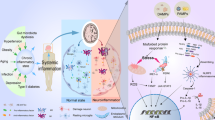
Acute neurological diseases are mostly related to abnormal discharge of cerebral nerves and cerebral vascular lesions, and are often accompanied by serious sequelae. In this section, we will mainly discuss how SIRT3 regulates its substrates to regulate the occurrence and development of acute neurological diseases (Fig. 2).
SIRT3 and acute neurodegenerative diseases. In the vast majority of cases, SIRT3 has a positive effect on the treatment of acute neurodegenerative diseases. Inhibition of apoptosis and OS, as well as promotion of energy production, autophagy, and stabilization of mitochondrial dynamics by SIRT3 can also inhibit the progression of acute neurodegenerative diseases. SIRT3 can also alleviate the clinical manifestations of the diseases by inhibiting neuroinflammation, glial Scars and thrombosis, promoting angiogenesis and nneurogenesis. However, it is worth noting that deacetylation of CerS1, CerS2, and CerS6 by SIRT3 accelerates the accumulation of ceramides, which induces OS that further exacerbates cerebral I/R injury. The green proteins represent the substrates of SIRT3. Brown circles represent acetyl group
SIRT3 and epilepsy (EP)
Simply put, EP is a disease caused by excessive discharge of central nervous system neurons (Marchi et al. 2014). The etiology of the disease is very complex, and EP is generally divided into the following four categories: idiopathic, symptomatic, provoked, and cryptogenic (Shorvon 2011). Effective treatment measures depend on the cause of EP.
The expression of SIRT3 is reduced in a chronic EP disease model (Gano et al. 2018), which suggests that SIRT3 may become a target protein for the treatment of EP. SIRT3 can promote the activity of FOXO3a, MnSOD, and IDH2, and can also increase the expression of uncoupling protein 2 (UCP2) in neurons, which all contribute to the inhibition of neuronal OS damage (Cho et al. 2019; Gano et al. 2018; Hasan-Olive et al. 2019). CypD is deacetylated by SIRT3 and its activity is inhibited, which limits the opening of mitochondrial membrane permeability transition pore (mPTP) and inhibits neuronal apoptosis, thereby protecting neurons from excitotoxicity (Cheng et al. 2016). The promotion of autophagy by SIRT3 during seizures also reduces brain damage, but it has not been determined whether the relevant mechanism involves the LKB1–AMPK–mTOR pathway, which is closely linked to autophagy (Wang et al. 2018; Wu et al. 2020a, b, c). The deacetylation of NDUFA9 by SIRT3 can promote the activity of complex I, thereby inhibiting mitochondrial dysfunction and promoting ATP production (Ahn et al. 2008; Gano et al. 2018). OS damage and abnormal energy metabolism are important mechanisms leading to the occurrence and deterioration of EP (Pauletti et al. 2019; Pearson-Smith and Patel 2017). Therefore, SIRT3 agonists may become effective drugs for the treatment of EP. In addition, inflammation seriously affects the occurrence and development of EP (Vezzani et al. 2011), and SIRT3 can also effectively inhibit the inflammatory response in a variety of ways (Almalki et al. 2021; Dikalova et al. 2020; Huang et al. 2019; Palomer et al. 2020; Song et al. 2019). However, the relationship between the three and related mechanisms has not yet been fully clarified.
SIRT3 and ischemic stroke
When the cerebral blood vessels are ruptured or blocked, blood cannot flow into the brain in time, so the brain tissue is damaged to different degrees, which is known as a stroke. Depending on the cause, stroke is divided into ischemic stroke and hemorrhagic stroke (Broderick et al. 2017). The incidence of the former is greater than that of the latter. Regardless of the type of stroke, if patients are not treated in time, they often suffer from serious sequelae. At present, due to the rapid progression of the disease, the prevention of stroke occupies a more important position than the treatment of stroke. However, when patients are not treated in time, powerful and effective drugs can alleviate the brain damage caused by stroke, which provides great help to improve the patients' quality of life.
Several investigators have reported that SIRT3 expression in mouse hippocampal neurons is decreased after acute ischemic stroke and that acute ischemia-induced neuronal damage is attenuated under conditions of SIRT3 overexpression (Fan et al. 2021). This suggests that SIRT3 may contribute to the treatment of nerve damage caused by an ischemic stroke. As previously mentioned, SIRT3 promotes the phosphorylation level of AMPK and then inhibits the phosphorylation level of mTOR to promote autophagy. This mechanism also plays a protective role against ischemia-induced brain injury (Dai et al. 2017; Li et al. 2021a, b). However, it remains unknown whether this effect involves the activation of LKB1 by SIRT3 in this disease condition. Activated AMPK can also activate SIRT3 by activating PGC-1α, which in turn forms a positive feedback pathway (Gao et al. 2018). The function of SIRT3 to activate FOXO3a and MnSOD to inhibit OS can also alleviate the damage caused by cerebral ischemia (Wang et al. 2020a, b; Wang et al. 2015; Yin et al. 2015). Glial scars produced after a cerebral ischemic injury can inhibit neuronal repair (Bao et al. 2012; Barres 2008; Jeong et al. 2012; Ridet et al. 1997). Inhibition of Notch1 expression by SIRT3 inhibits astrocyte activation, which in turn inhibits glial scar formation (Yang et al. 2017). However, excessive inhibition of the Notch signaling pathway may inhibit SIRT3 (Guo et al. 2020). Therefore, it is very important to adjust the balance between SIRT3 and Notch1. One of the sequelae of cerebral ischemic injury is cognitive dysfunction (CD). SIRT3 can also promote the phosphorylation level of advanced protein kinase B (AKT) and extracellular signal-regulated kinases (ERK) and the expression of vascular endothelial growth factor (VEGF), thereby promoting neurogenesis and angiogenesis in the injured area. In addition, phosphorylated AKT and ERK can also promote the expression of VEGF (Yang et al. 2018a, b). SIRT3 can also promote the transfer of microglia to cerebral ischemic areas by promoting the expression of fractalkine receptor (CX3CR1) (Cao et al. 2019). However, due to different disease conditions, microglia can both relieve ischemic damage and promote ischemic damage (Block et al. 2007; Fu et al. 2014; Nakajima and Kohsaka 2004). Therefore, the role of SIRT3 in this aspect needs to be further explored.
From the perspective of prevention, SIRT3 has an inhibitory effect on neutrophil extracellular traps (NETs) and plasma tissue factor (TF). This can inhibit the formation of thrombi, thereby reducing the incidence of ischemic brain injury (Gaul et al. 2018).
SIRT3 and hemorrhagic stroke
Intracerebral hemorrhage (ICH) and subarachnoid hemorrhage (SAH) are the two most representative subtypes of hemorrhagic stroke (Luo et al. 2019; Zhao et al. 2018a, b). The former is characterized by the accumulation of blood in the brain tissue (Zhao et al. 2018a, b), which accounts for 10%–20% of all stroke events worldwide (Qureshi et al. 2009; Sporns et al. 2021; van Asch et al. 2010); the latter is characterized by the accumulation of blood in the subarachnoid space (Zhao et al. 2018a, b) and accounts for 5%–15% of all strokes (Liao et al. 2020).
Inhibition of ROS production by SIRT3 inhibits both OS injury and the expression of NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome and interleukin-1beta (IL-1β) and the resulting neuroinflammation (Ma et al. 2014; Zheng et al. 2018; Zhou et al. 2010). Inhibition of neuroinflammation by SIRT3 can also be achieved by activating NRF2. NRF2 promotes the expression of heme oxygenase-1 (HO-1), which in turn inhibits the production of inflammatory factors such as tumor necrosis factor-α (TNF-α) and IL-1β (Dai et al. 2022). Furthermore, activation of the NRF1–TFAM pathway by SIRT3 promotes mitochondrial biogenesis, which maintains stable mitochondrial dynamics (Zheng et al. 2018). The above effects can effectively improve secondary brain injury (SBI) caused by ICH (Zheng et al. 2018).
Similar to the impact of SIRT3 on ICH, SIRT3 also has a protective effect on SAH-induced brain damage (Huang et al. 2016). Some researchers have found that SIRT3 in SAH model mice is inhibited, and the ability of neurons to resist OS is reduced (Zhang et al. 2020a, b, c, d). After SIRT3 is activated, it can be found that the degree of neuronal OS damage and the level of neuronal apoptosis are reduced (Wu et al. 2020a, b, c; Yang et al. 2018a, b; Zhang et al. 2019). SIRT3 can also promote the expression of Mfn1 and Mfn2 by activating AMPK, thereby promoting mitochondrial fusion and maintaining the normal mitochondrial morphology (Wu et al. 2020a, b, c). This plays an important role in maintaining the normal function of mitochondria.
SIRT3 and cerebral ischemia–reperfusion injury
As mentioned above, cerebral ischemia can cause serious damage, and the degree of damage is closely related to the time of ischemia (Carden and Granger 2000; Kloner et al. 1974; Raedschelders et al. 2012). Therefore, timely blood perfusion to the ischemic site can ensure the survival of the ischemic tissue (Wu et al. 2021). However, the damage to ischemic tissues undergoing reperfusion therapy in some experiments was abnormally enhanced (Hearse et al. 1973; Reimer et al. 1977). This is a kind of OS damage caused by the entry of active oxygen into the dredged parts (Granger and Kvietys 2015). Therefore, the elimination of active oxygen has become an effective means of treating this kind of disease.
Increasing the content and activity of SIRT3 can inhibit cerebral I/R injury (Gao et al. 2020; Liu et al. 2020a, b, c; Su et al. 2017). The activation of MnSOD by SIRT3 can also alleviate the OS damage of the brain tissue caused by I/R (Liu et al. 2021a, b). SIRT3 can inhibit the phosphorylation level and activity of β-catenin, thereby inhibiting excessive mitochondrial fission and maintaining the stability of mitochondrial dynamics (Zhao et al. 2018a, b). SIRT3 can also promote the deacetylation level of COX-1, which can improve OS after I/R (Tu et al. 2019). In addition, the promotion of autophagy by SIRT3 mediated by the AMPK–mTOR pathway can also inhibit I/R injury, but this function is achieved by removing damaged nerve cells rather than by inhibiting OS (Chen et al. 2021a, b). However, an experiment conducted in 2016 showed that SIRT3 is activated by a currently unknown mechanism and increases the deacetylation level and activity of CerS1, CerS2, and CerS6 after cerebral ischemia–reperfusion. This leads to the accumulation of ceramides. High levels of ceramides can lead to OS damage (Novgorodov et al. 2016). This is completely contrary to the previous conclusion, which suggests that the protective effect of SIRT3 on the nervous system may depend on a state of balance. Once this balance is broken, SIRT3 exerts a negative effect. The specific reasons are expected to be verified in future experiments.
SIRT3 and CD caused by non-neurodegenerative diseases
Almost all neurodegenerative diseases can cause CD, especially PD and AD. However, some nonneurological diseases and clinical operations can also cause CD. There are some differences between the two. Therefore, it is necessary to explain this part separately.
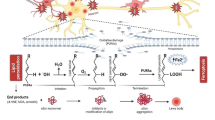
Postoperative delirium and cognitive dysfunction (POCD) is a common complication that occurs in elderly patients after surgery (Deiner and Silverstein 2009), which can lead to an increase in postoperative mortality. This phenomenon is related to the OS response of mitochondria (Netto et al. 2018). SIRT3 promotes the inhibition of OS by activating MnSOD, and the consequent inhibition of neuroinflammation can alleviate POCD (Bajwa et al. 2019; Liu et al. 2021a, b; Ye et al. 2019). In addition, the inhibitory effect of SIRT3 on neuroinflammation plays an important role in alleviating sleep-disordered breathing (SDB)-induced CD (Lin et al. 2020). Through acetylome analysis, SIRT3's deacetylation of brain mitochondrial proteins can also alleviate CD induced by metabolic syndrome (MetS) (Tyagi et al. 2018). Infection can also cause CD. Deacetylation of CypD by SIRT3 attenuates sepsis-associated encephalopathy (SAE)–induced CD through the inhibition of apoptosis and neuroinflammation (Sun et al. 2017). SIRT3 can inhibit the decrease in the expression of antioxidant enzymes MnSOD, CAT, and Gpx caused by human immunodeficiency virus (HIV) transactivator of transcription (TAT), thereby inhibiting OS-induced microglia senescence, and improving HIV-associated neurocognitive disorders (HAND) (Thangaraj et al. 2021). Thus, SIRT3 improves CD caused by different reasons (Fig. 3). The modulation of SIRT3 will help to improve the quality of life of related patients.
SIRT3 and CD caused by non-neurodegenerative diseases. SIRT3 can alleviate CD caused by non-neurodegenerative diseases. This is achieved through inhibition of neuroinflammation, OS and apoptosis. SIRT3 can inhibit the activation of microglia and thus inhibit neuroinflammation. However, the mechanism of this activation has not yet been elucidated. The green proteins represent the substrates of SIRT3. Brown circles represent acetyl group
SIRT3 and rehabilitation training
Neurological diseases are often incurable and irreversible. Therefore, it is critical to alleviate the progression of related diseases and complications through some treatment. This can greatly improve the patient's quality of life. Several types of rehabilitation training mediated by SIRT3 are briefly introduced here.
Effects of physical activity on SIRT3 and related diseases
A sedentary lifestyle is an important risk factor for various diseases, such as cardiovascular disease, diabetes, and neurological diseases (Korta et al. 2019). Reasonable exercise can improve the prognosis of most diseases and promote human health (Koltai et al. 2018; Vargas-Ortiz et al. 2015). A comparative study conducted in 2015 compared the cognitive function of athletes who exercised regularly with that of nonathletes who were sedentary and found that athletes had better memory and reaction times (Zhao et al. 2016). The results of another similar study were also reported in 2019. The results of the experiment showed that the average cognitive ability of rugby players is stronger than that of low-intensity groups of the same age. After comparing the blood samples of the above two groups of people, the expression levels of SIRT3, CAT, and SOD1 in the low-activity group were shown to be significantly lower than those in the rugby players. This suggests that the beneficial effects of exercise on neurological diseases may be related to the anti-OS effect of SIRT3 (Corpas et al. 2019).
Some researchers have used a treadmill to perform exercise training experiments on AD model mice. The results showed that compared with the non-exercise group, the symptoms of the mice after 20 weeks of standard training were relieved; additionally, the content of SIRT3 was increased and the acetylation level of MnSOD and OGG1 were decreased in the hippocampus. SIRT3 activates MnSOD and OGG1 to inhibit OS and repair DNA damage, which is an important mechanism by which physical exercise alleviates AD symptoms (Bo et al. 2014).
SIRT3 may also mediate the remission of physical exercise in neurological disorders caused by severe metabolic diseases. After performing aerobic interval training (AIT) on mice, we found that appropriate activity can effectively improve CD caused by obesity (Shi et al. 2018). This is not only related to the inhibition of OS by SIRT3 but also the inhibition of obesity by SIRT3. Aerobic training (AT) promotes the expression of SIRT3, which activates PGC-1α and key enzymes in the metabolism of substances. Therefore, the process of fatty acid oxidation and OXPHOS is accelerated, and the fat mass can be effectively reduced (Karvinen et al. 2016; Vargas-Ortiz et al. 2018).
Effects of caloric restriction (CR) on SIRT3 and related diseases
CR is a nutritional intervention that reduces energy intake by 25%–30% while maintaining normal energy requirements (Pignatti et al. 2020). Numerous studies have demonstrated that CR can suppress the clinical manifestations of aging-related diseases and prolong lifespan (Barger et al. 2015; Kobayashi et al. 2017; Qiu et al. 2010; Someya et al. 2010; Wegman et al. 2015; Yu et al. 2018). During CR, the low energy intake favors a catabolic state; a large amount of acetyl-CoA is produced from the fatty acid metabolism process; mitochondrial proteins are in a hyperacetylation state. Therefore, to balance the levels of acetylation and deacetylation in cells, the activities and expression levels of various deacetylases, including SIRT3, are increased (Silaghi et al. 2021).
Although this is a compensatory change, the body does not suffer the damage caused by energy deficiency because the regulation of downstream proteins by the compensatory activation of deacetylases can fully compensate for this part of energy loss (Liu et al. 2014; North and Sinclair 2007; Shimazu et al. 2010). Conversely, the compensatory activation of deacetylases additionally gives the body a variety of protective measures. Intermittent fasting (IF)–induced elevation of SIRT3 protects neurons from excitotoxic damage in animal models of EP and AD. The increased activity of MnSOD after being deacetylated by SIRT3 inhibits the accumulation of ROS in cells. In this case, GABAergic tone is enhanced, thereby protecting neurons (Liu et al. 2019). The inhibition of neuroinflammation by IF also effectively protects the normal function of the brain tissue after ICH (Dai et al. 2022). In addition, studies on SIRT3 knockout mice have shown that they are more susceptible to excitotoxicity. At this time, mitochondria in brain tissue are more prone to mitochondrial permeability transition (MPT), and CypD is in a hyperacetylated state (Cheng et al. 2016). Therefore, deacetylation of CypD by the compensatory activation of SIRT3 under CR conditions inhibits mitochondrial permeability, thereby promoting mitochondrial calcium retention, which in turn inhibits neurological diseases caused by excitotoxic substances and calcium overload (Amigo et al. 2017). In addition to this, a high-fat–based and low-carbohydrate–based ketogenic diet (KD) contributes to the treatment and prognosis of refractory EP through compensatory mechanisms (Hasan-Olive et al. 2019).
In addition, although the subjects in some studies did not have neurological diseases, exercise and CR were shown to inhibit OS (Andrianova et al. 2020; Donniacuo et al. 2019; Jang et al. 2012; Jiang et al. 2014; Liang et al. 2013; Shi et al. 2005; Tsukiyama et al. 2017), inhibit inflammation (Traba et al. 2017, 2015), and promote autophagy (Li et al. 2018) through SIRT3. This is expected to provide theoretical support for future research on the relationship between rehabilitation training, SIRT3, and neurological diseases. Moreover, the aforementioned exercise methods and diet management are more or less different in different studies. Therefore, determining an appropriate range and selecting appropriate rehabilitation methods for different neurological diseases may be the focus of future research.
Conclusion
To date, the morbidity and mortality of various neurological diseases are high, which is a global public health problem. However, because the pathogenesis of neurological diseases has not been fully clarified, various treatment measures have not had good therapeutic effects. Hence, a comprehensive understanding of neurological disease pathogeneses is necessary. In recent years, numerous experiments have shown that post-translational modifications are inseparable from the pathogenesis of neurological diseases. As a deacetylase, SIRT3 actively regulates the occurrence and development of neurological diseases through the regulation of downstream proteins. SIRT3 mainly inhibits various types of neurological diseases. This is achieved through the regulation of physiological activities such as OS, apoptosis, mitochondrial dynamics, and material metabolism by SIRT3. However, only one report has pointed to the deleterious effects of SIRT3 in nonneoplastic neurological diseases (Novgorodov et al. 2016). The accumulation of ceramides induced by SIRT3 is an important cause of cerebral ischemia–reperfusion injury, which is achieved through the activation of deacetylation of CerS1, CerS2, and CerS6 by SIRT3. In addition, the negative effects of SIRT3 on neurological diseases are mainly manifested in tumors because SIRT3 is ultimately responsible for the survival of nervous system cells through various forms of regulation. However, this kind of regulation acts on tumor cells to promote the proliferation and migration of cancer. For example, SIRT3 can promote the survival and invasion of glioma by inhibiting OS. Thus, for patients who have multiple diseases at the same time, establishing a clinical treatment method that correctly controls the dosage of SIRT3 activators and inhibitors to ensure an appropriate effect of SIRT3 remains challenging.
Data availability
All data generated or analysed during this study are included in this published article.
Code availability
Not applicable.
References
Ahn BH, Kim HS, Song S, Lee IH, Liu J, Vassilopoulos A, Deng CX, Finkel T (2008) A role for the mitochondrial deacetylase Sirt3 in regulating energy homeostasis. Proc Natl Acad Sci U S A 105(38):14447–14452. https://doi.org/10.1073/pnas.0803790105
Almalki WH, Alzahrani A, Mahmoud El-Daly ME, Fadel Ahmed AHF (2021) The emerging potential of SIRT-3 in oxidative stress-inflammatory axis associated increased neuroinflammatory component for metabolically impaired neural cell. Chem Biol Interact 333:109328. https://doi.org/10.1016/j.cbi.2020.109328
Alrob OA, Sankaralingam S, Ma C, Wagg CS, Fillmore N, Jaswal JS, Sack MN, Lehner R, Gupta MP, Michelakis ED, Padwal RS, Johnstone DE, Sharma AM, Lopaschuk GD (2014) Obesity-induced lysine acetylation increases cardiac fatty acid oxidation and impairs insulin signalling. Cardiovasc Res 103(4):485–497. https://doi.org/10.1093/cvr/cvu156
Amigo I, Menezes-Filho SL, Luévano-Martínez LA, Chausse B, Kowaltowski AJ (2017) Caloric restriction increases brain mitochondrial calcium retention capacity and protects against excitotoxicity. Aging Cell 16(1):73–81. https://doi.org/10.1111/acel.12527
Andrianova NV, Zorova LD, Pevzner IB, Popkov VA, Chernikov VP, Silachev DN, Plotnikov EY, Zorov DB (2020) Resemblance and differences in dietary restriction nephroprotective mechanisms in young and old rats. Aging (albany NY) 12(18):18693–18715. https://doi.org/10.18632/aging.103960
Bajwa E, Pointer CB, Klegeris A (2019) The Role of Mitochondrial Damage-Associated Molecular Patterns in Chronic Neuroinflammation. Mediators Inflamm 2019:4050796. https://doi.org/10.1155/2019/4050796
Bao Y, Qin L, Kim E, Bhosle S, Guo H, Febbraio M, Haskew-Layton RE, Ratan R, Cho S (2012) CD36 is involved in astrocyte activation and astroglial scar formation. J Cereb Blood Flow Metab 32(8):1567–1577. https://doi.org/10.1038/jcbfm.2012.52
Barger JL, Anderson RM, Newton MA, da Silva C, Vann JA, Pugh TD, Someya S, Prolla TA, Weindruch R (2015) A conserved transcriptional signature of delayed aging and reduced disease vulnerability is partially mediated by SIRT3. PLoS ONE 10(4):e0120738. https://doi.org/10.1371/journal.pone.0120738
Barres BA (2008) The mystery and magic of glia: a perspective on their roles in health and disease. Neuron 60(3):430–440. https://doi.org/10.1016/j.neuron.2008.10.013
Block ML, Zecca L, Hong JS (2007) Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci 8(1):57–69. https://doi.org/10.1038/nrn2038
Bo H, Kang W, Jiang N, Wang X, Zhang Y, Ji LL (2014) Exercise-induced neuroprotection of hippocampus in APP/PS1 transgenic mice via upregulation of mitochondrial 8-oxoguanine DNA glycosylase. Oxid Med Cell Longev 2014:834502. https://doi.org/10.1155/2014/834502
Broderick JP, Adeoye O, Elm J (2017) Evolution of the Modified Rankin Scale and Its Use in Future Stroke Trials. Stroke 48(7):2007–2012. https://doi.org/10.1161/strokeaha.117.017866
Brown RH, Al-Chalabi A (2017) Amyotrophic Lateral Sclerosis. N Engl J Med 377(2):162–172. https://doi.org/10.1056/NEJMra1603471
Cao R, Li S, Yin J, Guo L, Shi J (2019) Sirtuin 3 promotes microglia migration by upregulating CX3CR1. Cell Adh Migr 13(1):229–235. https://doi.org/10.1080/19336918.2019.1629224
Carden DL, Granger DN (2000) Pathophysiology of ischaemia-reperfusion injury. J Pathol 190(3):255–266. https://doi.org/10.1002/(sici)1096-9896(200002)190:3%3c255::Aid-path526%3e3.0.Co;2-6
Carrì MT, Polster BM, Beart PM (2018) Mitochondria in the nervous system: From health to disease, part II. Neurochem Int 117:1–4. https://doi.org/10.1016/j.neuint.2018.04.006
Chan DC (2020) Mitochondrial Dynamics and Its Involvement in Disease. Annu Rev Pathol 15:235–259. https://doi.org/10.1146/annurev-pathmechdis-012419-032711
Chang HC, Guarente L (2014) SIRT1 and other sirtuins in metabolism. Trends Endocrinol Metab 25(3):138–145. https://doi.org/10.1016/j.tem.2013.12.001
Chen D, Zheng K, Wu H, Zhang X, Ye W, Tan X, Xiong Y (2021a) Lin28a attenuates cerebral ischemia/reperfusion injury through regulating Sirt3-induced autophagy. Brain Res Bull 170:39–48. https://doi.org/10.1016/j.brainresbull.2021.01.022
Chen X, Lu W, Wu D (2021b) Sirtuin 2 (SIRT2): Confusing Roles in the Pathophysiology of Neurological Disorders. Front Neurosci 15:614107. https://doi.org/10.3389/fnins.2021.614107
Cheng A, Yang Y, Zhou Y, Maharana C, Lu D, Peng W, Liu Y, Wan R, Marosi K, Misiak M, Bohr VA, Mattson MP (2016) Mitochondrial SIRT3 Mediates Adaptive Responses of Neurons to Exercise and Metabolic and Excitatory Challenges. Cell Metab 23(1):128–142. https://doi.org/10.1016/j.cmet.2015.10.013
Cheng Y, Ren X, Gowda AS, Shan Y, Zhang L, Yuan YS, Patel R, Wu H, Huber-Keener K, Yang JW, Liu D, Spratt TE, Yang JM (2013) Interaction of Sirt3 with OGG1 contributes to repair of mitochondrial DNA and protects from apoptotic cell death under oxidative stress. Cell Death Dis 4(7):e731. https://doi.org/10.1038/cddis.2013.254
Cho I, Jeong KH, Zhu J, Choi YH, Cho KH, Heo K, Kim WJ (2019) Sirtuin3 Protected Against Neuronal Damage and Cycled into Nucleus in Status Epilepticus Model. Mol Neurobiol 56(7):4894–4903. https://doi.org/10.1007/s12035-018-1399-8
Choi J, Koh E, Lee YS, Lee HW, Kang HG, Yoon YE, Han WK, Choi KH, Kim KS (2016) Mitochondrial Sirt3 supports cell proliferation by regulating glutamine-dependent oxidation in renal cell carcinoma. Biochem Biophys Res Commun 474(3):547–553. https://doi.org/10.1016/j.bbrc.2016.04.117
Cimen H, Han MJ, Yang Y, Tong Q, Koc H, Koc EC (2010) Regulation of succinate dehydrogenase activity by SIRT3 in mammalian mitochondria. Biochemistry 49(2):304–311. https://doi.org/10.1021/bi901627u
Corpas R, Solana E, De la Rosa A, Sarroca S, Griñán-Ferré C, Oriol M, Corbella E, Rodríguez-Farré E, Vina J, Pallàs M, Bartrés-Faz D, Gomez-Cabrera MC, Sanfeliu C (2019) Peripheral Maintenance of the Axis SIRT1-SIRT3 at Youth Level May Contribute to Brain Resilience in Middle-Aged Amateur Rugby Players. Front Aging Neurosci 11:352. https://doi.org/10.3389/fnagi.2019.00352
Cui XX, Li X, Dong SY, Guo YJ, Liu T, Wu YC (2017) SIRT3 deacetylated and increased citrate synthase activity in PD model. Biochem Biophys Res Commun 484(4):767–773. https://doi.org/10.1016/j.bbrc.2017.01.163
Cui Y, Qin L, Wu J, Qu X, Hou C, Sun W, Li S, Vaughan AT, Li JJ, Liu J (2015) SIRT3 Enhances Glycolysis and Proliferation in SIRT3-Expressing Gastric Cancer Cells. PLoS One 10(6):e0129834. https://doi.org/10.1371/journal.pone.0129834
Dai S, Wei J, Zhang H, Luo P, Yang Y, Jiang X, Fei Z, Liang W, Jiang J, Li X (2022) Intermittent fasting reduces neuroinflammation in intracerebral hemorrhage through the Sirt3/Nrf2/HO-1 pathway. J Neuroinflammation 19(1):122. https://doi.org/10.1186/s12974-022-02474-2
Dai SH, Chen T, Li X, Yue KY, Luo P, Yang LK, Zhu J, Wang YH, Fei Z, Jiang XF (2017) Sirt3 confers protection against neuronal ischemia by inducing autophagy: Involvement of the AMPK-mTOR pathway. Free Radic Biol Med 108:345–353. https://doi.org/10.1016/j.freeradbiomed.2017.04.005
Dai SH, Chen T, Wang YH, Zhu J, Luo P, Rao W, Yang YF, Fei Z, Jiang XF (2014) Sirt3 attenuates hydrogen peroxide-induced oxidative stress through the preservation of mitochondrial function in HT22 cells. Int J Mol Med 34(4):1159–1168. https://doi.org/10.3892/ijmm.2014.1876
Deiner S, Silverstein JH (2009) Postoperative delirium and cognitive dysfunction. Br J Anaesth 103(Suppl 1):i41–46. https://doi.org/10.1093/bja/aep291
Dikalova AE, Pandey A, Xiao L, Arslanbaeva L, Sidorova T, Lopez MG, Billings FTt, Verdin E, Auwerx J, Harrison DG, Dikalov SI. (2020) Mitochondrial deacetylase Sirt3 reduces vascular dysfunction and hypertension while Sirt3 depletion in essential hypertension is linked to vascular inflammation and oxidative stress. Circ Res 126(4):439–452. https://doi.org/10.1161/circresaha.119.315767
Dong H, Zhou W, Xin J, Shi H, Yao X, He Z, Wang Z (2019) Salvinorin A moderates postischemic brain injury by preserving endothelial mitochondrial function via AMPK/Mfn2 activation. Exp Neurol 322:113045. https://doi.org/10.1016/j.expneurol.2019.113045
Donniacuo M, Urbanek K, Nebbioso A, Sodano L, Gallo L, Altucci L, Rinaldi B (2019) Cardioprotective effect of a moderate and prolonged exercise training involves sirtuin pathway. Life Sci 222:140–147. https://doi.org/10.1016/j.lfs.2019.03.001
Duan WJ, Li YF, Liu FL, Deng J, Wu YP, Yuan WL, Tsoi B, Chen JL, Wang Q, Cai SH, Kurihara H, He RR (2016) A SIRT3/AMPK/autophagy network orchestrates the protective effects of trans-resveratrol in stressed peritoneal macrophages and RAW 264.7 macrophages. Free Radic Biol Med 95:230–242. https://doi.org/10.1016/j.freeradbiomed.2016.03.022
Duan WJ, Liang L, Pan MH, Lu DH, Wang TM, Li SB, Zhong HB, Yang XJ, Cheng Y, Liu B, Li WX, Kurihara H, Bi W, Li YF, He RR (2020) Theacrine, a purine alkaloid from kucha, protects against Parkinson’s disease through SIRT3 activation. Phytomedicine 77:153281. https://doi.org/10.1016/j.phymed.2020.153281
Fan J, Shan C, Kang HB, Elf S, Xie J, Tucker M, Gu TL, Aguiar M, Lonning S, Chen H, Mohammadi M, Britton LM, Garcia BA, Alečković M, Kang Y, Kaluz S, Devi N, Van Meir EG, Hitosugi T, Seo JH, Lonial S, Gaddh M, Arellano M, Khoury HJ, Khuri FR, Boggon TJ, Kang S, Chen J (2014) Tyr phosphorylation of PDP1 toggles recruitment between ACAT1 and SIRT3 to regulate the pyruvate dehydrogenase complex. Mol Cell 53(4):534–548. https://doi.org/10.1016/j.molcel.2013.12.026
Fan JH, Song HM, Zhang X, Yan WJ, Han S, Yin YL (2021) Acute cerebral ischemia-induced down-regulation of Sirt3 protein expression contributes to neuronal injury via damaging mitochondrial function. Sheng Li Xue Bao 73(1):17–25
Fan L, Wang J, Ma C (2019) Pretreatment of bone mesenchymal stem cells with miR181-c facilitates craniofacial defect reconstruction via activating AMPK-Mfn1 signaling pathways. J Recept Signal Transduct Res 39(3):199–207. https://doi.org/10.1080/10799893.2019.1652649
Fernandes J, Weddle A, Kinter CS, Humphries KM, Mather T, Szweda LI, Kinter M (2015) Lysine Acetylation Activates Mitochondrial Aconitase in the Heart. Biochemistry 54(25):4008–4018. https://doi.org/10.1021/acs.biochem.5b00375
Forte M, Schirone L, Ameri P, Basso C, Catalucci D, Modica J, Chimenti C, Crotti L, Frati G, Rubattu S, Schiattarella GG, Torella D, Perrino C, Indolfi C, Sciarretta S (2021) The role of mitochondrial dynamics in cardiovascular diseases. Br J Pharmacol 178(10):2060–2076. https://doi.org/10.1111/bph.15068
Fu J, Jin J, Cichewicz RH, Hageman SA, Ellis TK, Xiang L, Peng Q, Jiang M, Arbez N, Hotaling K, Ross CA, Duan W (2012) trans-(-)-ε-Viniferin increases mitochondrial sirtuin 3 (SIRT3), activates AMP-activated protein kinase (AMPK), and protects cells in models of Huntington Disease. J Biol Chem 287(29):24460–24472. https://doi.org/10.1074/jbc.M112.382226
Fu R, Shen Q, Xu P, Luo JJ, Tang Y (2014) Phagocytosis of microglia in the central nervous system diseases. Mol Neurobiol 49(3):1422–1434. https://doi.org/10.1007/s12035-013-8620-6
Gano LB, Liang LP, Ryan K, Michel CR, Gomez J, Vassilopoulos A, Reisdorph N, Fritz KS, Patel M (2018) Altered mitochondrial acetylation profiles in a kainic acid model of temporal lobe epilepsy. Free Radic Biol Med 123:116–124. https://doi.org/10.1016/j.freeradbiomed.2018.05.063
Gao J, Chen N, Li N, Xu F, Wang W, Lei Y, Shi J, Gong Q (2020) Neuroprotective effects of Trilobatin, a novel naturally occurring Sirt3 agonist from Lithocarpus polystachyus Rehd, mitigate cerebral Ischemia/reperfusion injury: Involvement of TLR4/NF-κB and Nrf2/Keap-1 signaling. Antioxid Redox Signal 33(2):117–143. https://doi.org/10.1089/ars.2019.7825
Gao J, Liu S, Xu F, Liu Y, Lv C, Deng Y, Shi J, Gong Q (2018) Trilobatin protects against oxidative injury in neuronal PC12 cells through regulating mitochondrial ROS homeostasis mediated by AMPK/Nrf2/Sirt3 signaling pathway. Front Mol Neurosci 11:267. https://doi.org/10.3389/fnmol.2018.00267
Gaul DS, Weber J, van Tits LJ, Sluka S, Pasterk L, Reiner MF, Calatayud N, Lohmann C, Klingenberg R, Pahla J, Vdovenko D, Tanner FC, Camici GG, Eriksson U, Auwerx J, Mach F, Windecker S, Rodondi N, Lüscher TF, Winnik S, Matter CM (2018) Loss of Sirt3 accelerates arterial thrombosis by increasing formation of neutrophil extracellular traps and plasma tissue factor activity. Cardiovasc Res 114(8):1178–1188. https://doi.org/10.1093/cvr/cvy036
Geng L, Zhang T, Liu W, Chen Y (2018) miR-494-3p modulates the progression of in vitro and in vivo Parkinson’s disease models by targeting SIRT3. Neurosci Lett 675:23–30. https://doi.org/10.1016/j.neulet.2018.03.037
Giangregorio N, Tonazzi A, Console L, Indiveri C (2017) Post-translational modification by acetylation regulates the mitochondrial carnitine/acylcarnitine transport protein. Mol Cell Biochem 426(1–2):65–73. https://doi.org/10.1007/s11010-016-2881-0
Granger DN, Kvietys PR (2015) Reperfusion injury and reactive oxygen species: The evolution of a concept. Redox Biol 6:524–551. https://doi.org/10.1016/j.redox.2015.08.020
Guo J, Zhang XL, Bao ZR, Yang XK, Li LS, Zi Y, Li F, Wu CY, Li JJ, Yuan Y (2020) Gastrodin regulates the notch signaling pathway and Sirt3 in activated microglia in cerebral hypoxic-ischemia neonatal rats and in activated BV-2 microglia. Neuromolecular Med. https://doi.org/10.1007/s12017-020-08627-x
Hallows WC, Lee S, Denu JM (2006) Sirtuins deacetylate and activate mammalian acetyl-CoA synthetases. Proc Natl Acad Sci U S A 103(27):10230–10235. https://doi.org/10.1073/pnas.0604392103
Hallows WC, Yu W, Smith BC, Devries MK, Ellinger JJ, Someya S, Shortreed MR, Prolla T, Markley JL, Smith LM, Zhao S, Guan KL, Denu JM (2011) Sirt3 promotes the urea cycle and fatty acid oxidation during dietary restriction. Mol Cell 41(2):139–149. https://doi.org/10.1016/j.molcel.2011.01.002
Harlan BA, Killoy KM, Pehar M, Liu L, Auwerx J, Vargas MR (2020) Evaluation of the NAD(+) biosynthetic pathway in ALS patients and effect of modulating NAD(+) levels in hSOD1-linked ALS mouse models. Exp Neurol 327:113219. https://doi.org/10.1016/j.expneurol.2020.113219
Hasan-Olive MM, Lauritzen KH, Ali M, Rasmussen LJ, Storm-Mathisen J, Bergersen LH (2019) A ketogenic diet improves mitochondrial biogenesis and bioenergetics via the PGC1α-SIRT3-UCP2 axis. Neurochem Res 44(1):22–37. https://doi.org/10.1007/s11064-018-2588-6
Hearse DJ, Humphrey SM, Chain EB (1973) Abrupt reoxygenation of the anoxic potassium-arrested perfused rat heart: a study of myocardial enzyme release. J Mol Cell Cardiol 5(4):395–407. https://doi.org/10.1016/0022-2828(73)90030-8
Hirschey MD, Shimazu T, Goetzman E, Jing E, Schwer B, Lombard DB, Grueter CA, Harris C, Biddinger S, Ilkayeva OR, Stevens RD, Li Y, Saha AK, Ruderman NB, Bain JR, Newgard CB, Farese RV Jr, Alt FW, Kahn CR, Verdin E (2010) SIRT3 regulates mitochondrial fatty-acid oxidation by reversible enzyme deacetylation. Nature 464(7285):121–125. https://doi.org/10.1038/nature08778
Hor JH, Santosa MM, Lim VJW, Ho BX, Taylor A, Khong ZJ, Ravits J, Fan Y, Liou YC, Soh BS, Ng SY (2021) ALS motor neurons exhibit hallmark metabolic defects that are rescued by SIRT3 activation. Cell Death Differ 28(4):1379–1397. https://doi.org/10.1038/s41418-020-00664-0
Huang D, Liu M, Jiang Y (2019) Mitochonic acid-5 attenuates TNF-α-mediated neuronal inflammation via activating Parkin-related mitophagy and augmenting the AMPK-Sirt3 pathways. J Cell Physiol 234(12):22172–22182. https://doi.org/10.1002/jcp.28783
Huang W, Huang Y, Huang RQ, Huang CG, Wang WH, Gu JM, Dong Y (2016) SIRT3 expression decreases with reactive oxygen species generation in rat cortical neurons during early brain injury induced by experimental subarachnoid hemorrhage. Biomed Res Int 2016:8263926. https://doi.org/10.1155/2016/8263926
Huang Y, Liao J, Liu X, Zhong Y, Cai X, Long L (2021) Review: The role of intestinal dysbiosis in Parkinson’s disease. Front Cell Infect Microbiol 11:615075. https://doi.org/10.3389/fcimb.2021.615075
Imai S, Guarente L (2014) NAD+ and sirtuins in aging and disease. Trends Cell Biol 24(8):464–471. https://doi.org/10.1016/j.tcb.2014.04.002
Jamali-Raeufy N, Mojarrab Z, Baluchnejadmojarad T, Roghani M, Fahanik-Babaei J, Goudarzi M (2020) The effects simultaneous inhibition of dipeptidyl peptidase-4 and P2X7 purinoceptors in an in vivo Parkinson’s disease model. Metab Brain Dis 35(3):539–548. https://doi.org/10.1007/s11011-020-00538-x
Jang YC, Liu Y, Hayworth CR, Bhattacharya A, Lustgarten MS, Muller FL, Chaudhuri A, Qi W, Li Y, Huang JY, Verdin E, Richardson A, Van Remmen H (2012) Dietary restriction attenuates age-associated muscle atrophy by lowering oxidative stress in mice even in complete absence of CuZnSOD. Aging Cell 11(5):770–782. https://doi.org/10.1111/j.1474-9726.2012.00843.x
Jeong SR, Kwon MJ, Lee HG, Joe EH, Lee JH, Kim SS, Suh-Kim H, Kim BG (2012) Hepatocyte growth factor reduces astrocytic scar formation and promotes axonal growth beyond glial scars after spinal cord injury. Exp Neurol 233(1):312–322. https://doi.org/10.1016/j.expneurol.2011.10.021
Jiang DQ, Ma YJ, Wang Y, Lu HX, Mao SH, Zhao SH (2019) Microglia activation induces oxidative injury and decreases SIRT3 expression in dopaminergic neuronal cells. J Neural Transm (vienna) 126(5):559–568. https://doi.org/10.1007/s00702-019-02005-z
Jiang DQ, Wang Y, Li MX, Ma YJ, Wang Y (2017) SIRT3 in neural stem cells attenuates microglia activation-induced oxidative stress injury through mitochondrial pathway. Front Cell Neurosci 11:7. https://doi.org/10.3389/fncel.2017.00007
Jiang HK, Miao Y, Wang YH, Zhao M, Feng ZH, Yu XJ, Liu JK, Zang WJ (2014) Aerobic interval training protects against myocardial infarction-induced oxidative injury by enhancing antioxidase system and mitochondrial biosynthesis. Clin Exp Pharmacol Physiol 41(3):192–201. https://doi.org/10.1111/1440-1681.12211
Jimenez-Sanchez M, Licitra F, Underwood BR, Rubinsztein DC (2017) Huntington’s Disease: Mechanisms of Pathogenesis and Therapeutic Strategies. Cold Spring Harb Perspect Med 7(7):a024240. https://doi.org/10.1101/cshperspect.a024240
Jing E, O’Neill BT, Rardin MJ, Kleinridders A, Ilkeyeva OR, Ussar S, Bain JR, Lee KY, Verdin EM, Newgard CB, Gibson BW, Kahn CR (2013) Sirt3 regulates metabolic flexibility of skeletal muscle through reversible enzymatic deacetylation. Diabetes 62(10):3404–3417. https://doi.org/10.2337/db12-1650
Jung CH, Ro SH, Cao J, Otto NM, Kim DH (2010) mTOR regulation of autophagy. FEBS Lett 584(7):1287–1295. https://doi.org/10.1016/j.febslet.2010.01.017
Kane AE, Sinclair DA (2018) Sirtuins and NAD(+) in the development and treatment of metabolic and cardiovascular diseases. Circ Res 123(7):868–885. https://doi.org/10.1161/circresaha.118.312498
Karvinen S, Silvennoinen M, Vainio P, Sistonen L, Koch LG, Britton SL, Kainulainen H (2016) Effects of intrinsic aerobic capacity, aging and voluntary running on skeletal muscle sirtuins and heat shock proteins. Exp Gerontol 79:46–54. https://doi.org/10.1016/j.exger.2016.03.015
Kida Y, Goligorsky MS (2016) Sirtuins, cell senescence, and vascular aging. Can J Cardiol 32(5):634–641. https://doi.org/10.1016/j.cjca.2015.11.022
Kloner RA, Ganote CE, Whalen DA Jr, Jennings RB (1974) Effect of a transient period of ischemia on myocardial cells. II. Fine structure during the first few minutes of reflow. Am J Pathol 74(3):399–422
Kobayashi M, Takeda K, Narita T, Nagai K, Okita N, Sudo Y, Miura Y, Tsumoto H, Nakagawa Y, Shimano H, Higami Y (2017) Mitochondrial intermediate peptidase is a novel regulator of sirtuin-3 activation by caloric restriction. FEBS Lett 591(24):4067–4073. https://doi.org/10.1002/1873-3468.12914
Koltai E, Bori Z, Osvath P, Ihasz F, Peter S, Toth G, Degens H, Rittweger J, Boldogh I, Radak Z (2018) Master athletes have higher miR-7, SIRT3 and SOD2 expression in skeletal muscle than age-matched sedentary controls. Redox Biol 19:46–51. https://doi.org/10.1016/j.redox.2018.07.022
Korta P, Pocheć E, Mazur-Biały A (2019) Irisin as a Multifunctional Protein: Implications for Health and Certain Diseases. Medicina (Kaunas) 55(8):485. https://doi.org/10.3390/medicina55080485
Lee J, Kim Y, Liu T, Hwang YJ, Hyeon SJ, Im H, Lee K, Alvarez VE, McKee AC, Um SJ, Hur M, Mook-Jung I, Kowall NW, Ryu H (2018) SIRT3 deregulation is linked to mitochondrial dysfunction in Alzheimer’s disease. Aging Cell 17(1):e12679. https://doi.org/10.1111/acel.12679
Li FH, Li T, Ai JY, Sun L, Min Z, Duan R, Zhu L, Liu YY, Liu TC (2018) Beneficial autophagic activities, mitochondrial function, and metabolic phenotype adaptations promoted by high-intensity interval training in a rat model. Front Physiol 9:571. https://doi.org/10.3389/fphys.2018.00571
Li K, Li X, Shi G, Lei X, Huang Y, Bai L, Qin C (2021a) Effectiveness and mechanisms of adipose-derived stem cell therapy in animal models of Parkinson’s disease: a systematic review and meta-analysis. Transl Neurodegener 10(1):14. https://doi.org/10.1186/s40035-021-00238-1
Li L, Zhang P, Bao Z, Wang T, Liu S, Huang F (2016a) PGC-1α promotes ureagenesis in mouse periportal hepatocytes through SIRT3 and SIRT5 in response to glucagon. Sci Rep 6:24156. https://doi.org/10.1038/srep24156
Li PA, Hou X, Hao S (2017) Mitochondrial biogenesis in neurodegeneration. J Neurosci Res 95(10):2025–2029. https://doi.org/10.1002/jnr.24042
Li S, Yin J, Nielsen M, Beach TG, Guo L, Shi J (2019) Sirtuin 3 mediates tau deacetylation. J Alzheimers Dis 69(2):355–362. https://doi.org/10.3233/jad-190014
Li Y, Hu K, Liang M, Yan Q, Huang M, Jin L, Chen Y, Yang X, Li X (2021b) Stilbene glycoside upregulates SIRT3/AMPK to promotes neuronal mitochondrial autophagy and inhibit apoptosis in ischemic stroke. Adv Clin Exp Med 30(2):139–146. https://doi.org/10.17219/acem/130608
Li Y, Lu J, Cao X, Zhao H, Gao L, Xia P, Pei G (2020) A newly synthesized rhamnoside derivative alleviates alzheimer’s amyloid-β-induced oxidative stress, mitochondrial dysfunction, and cell senescence through upregulating SIRT3. Oxid Med Cell Longev 2020:7698560. https://doi.org/10.1155/2020/7698560
Li YH, Choi DH, Lee EH, Seo SR, Lee S, Cho EH (2016b) Sirtuin 3 (SIRT3) regulates α-smooth muscle actin (α-SMA) production through the succinate dehydrogenase-G protein-coupled receptor 91 (GPR91) pathway in hepatic stellate cells. J Biol Chem 291(19):10277–10292. https://doi.org/10.1074/jbc.M115.692244
Liang L, Li Q, Huang L, Li D, Li X (2015) Sirt3 binds to and deacetylates mitochondrial pyruvate carrier 1 to enhance its activity. Biochem Biophys Res Commun 468(4):807–812. https://doi.org/10.1016/j.bbrc.2015.11.036
Liang Q, Benavides GA, Vassilopoulos A, Gius D, Darley-Usmar V, Zhang J (2013) Bioenergetic and autophagic control by Sirt3 in response to nutrient deprivation in mouse embryonic fibroblasts. Biochem J 454(2):249–257. https://doi.org/10.1042/bj20130414
Liao LS, Zhang MW, Gu YJ, Sun XC (2020) Targeting CCL20 inhibits subarachnoid hemorrhage-related neuroinflammation in mice. Aging (albany NY) 12(14):14849–14862. https://doi.org/10.18632/aging.103548
Lin Y, Liu X, Tan D, Jiang Z (2020) Atractylon treatment prevents sleep-disordered breathing-induced cognitive dysfunction by suppression of chronic intermittent hypoxia-induced M1 microglial activation. Biosci Rep 40(6):BSR20192800. https://doi.org/10.1042/bsr20192800
Liu H, Li S, Liu X, Chen Y, Deng H (2018a) SIRT3 Overexpression Inhibits Growth of Kidney Tumor Cells and Enhances Mitochondrial Biogenesis. J Proteome Res 17(9):3143–3152. https://doi.org/10.1021/acs.jproteome.8b00260
Liu J, Wang Y, Wang Z, Hao Y, Bai W, Wang Z, Wang J (2020a) 5-Heptadecylresorcinol, a Biomarker for Whole Grain Rye Consumption, Ameliorates Cognitive Impairments and Neuroinflammation in APP/PS1 Transgenic Mice. Mol Nutr Food Res 64(11):e1901218. https://doi.org/10.1002/mnfr.201901218
Liu L, Cao Q, Gao W, Li B, Xia Z, Zhao B (2021a) Melatonin protects against focal cerebral ischemia-reperfusion injury in diabetic mice by ameliorating mitochondrial impairments: involvement of the Akt-SIRT3-SOD2 signaling pathway. Aging (Albany NY) 13(12):16105–16123. https://doi.org/10.18632/aging.203137
Liu L, Chen S, Yu M, Ge C, Ren M, Liu B, Yang X, Christian TW Jr, Hou YM, Zou J, Zhu WG, Luo J (2020b) Deacetylation of HSD17B10 by SIRT3 regulates cell growth and cell resistance under oxidative and starvation stresses. Cell Death Dis 11(7):563. https://doi.org/10.1038/s41419-020-02763-9
Liu L, Nam M, Fan W, Akie TE, Hoaglin DC, Gao G, Keaney JF Jr, Cooper MP (2014) Nutrient sensing by the mitochondrial transcription machinery dictates oxidative phosphorylation. J Clin Invest 124(2):768–784. https://doi.org/10.1172/jci69413
Liu L, Peritore C, Ginsberg J, Kayhan M, Donmez G (2015) SIRT3 attenuates MPTP-induced nigrostriatal degeneration via enhancing mitochondrial antioxidant capacity. Neurochem Res 40(3):600–608. https://doi.org/10.1007/s11064-014-1507-8
Liu M, Li X, Huang D (2020c) Mfn2 overexpression attenuates cardio-cerebrovascular ischemia-reperfusion injury through mitochondrial fusion and activation of the AMPK/Sirt3 signaling. Front Cell Dev Biol 8:598078. https://doi.org/10.3389/fcell.2020.598078
Liu P, Huang G, Wei T, Gao J, Huang C, Sun M, Zhu L, Shen W (2018b) Sirtuin 3-induced macrophage autophagy in regulating NLRP3 inflammasome activation. Biochim Biophys Acta Mol Basis Dis 1864(3):764–777. https://doi.org/10.1016/j.bbadis.2017.12.027
Liu Q, Sun YM, Huang H, Chen C, Wan J, Ma LH, Sun YY, Miao HH, Wu YQ (2021b) Sirtuin 3 protects against anesthesia/surgery-induced cognitive decline in aged mice by suppressing hippocampal neuroinflammation. J Neuroinflammation 18(1):41. https://doi.org/10.1186/s12974-021-02089-z
Liu X, Du H, Chai Q, Jia Q, Liu L, Zhao M, Li J, Tang H, Chen W, Zhao L, Fang L, Gao L, Zhao J (2018c) Blocking mitochondrial cyclophilin D ameliorates TSH-impaired defensive barrier of artery. Redox Biol 15:418–434. https://doi.org/10.1016/j.redox.2018.01.004
Liu Y, Cheng A, Li YJ, Yang Y, Kishimoto Y, Zhang S, Wang Y, Wan R, Raefsky SM, Lu D, Saito T, Saido T, Zhu J, Wu LJ, Mattson MP (2019) SIRT3 mediates hippocampal synaptic adaptations to intermittent fasting and ameliorates deficits in APP mutant mice. Nat Commun 10(1):1886. https://doi.org/10.1038/s41467-019-09897-1
Luo Y, Reis C, Chen S (2019) NLRP3 inflammasome in the pathophysiology of hemorrhagic stroke: A review. Curr Neuropharmacol 17(7):582–589. https://doi.org/10.2174/1570159x17666181227170053
Ma Q, Chen S, Hu Q, Feng H, Zhang JH, Tang J (2014) NLRP3 inflammasome contributes to inflammation after intracerebral hemorrhage. Ann Neurol 75(2):209–219. https://doi.org/10.1002/ana.24070
Ma S, Chen J, Feng J, Zhang R, Fan M, Han D, Li X, Li C, Ren J, Wang Y, Cao F (2018) Melatonin ameliorates the progression of atherosclerosis via mitophagy activation and NLRP3 inflammasome inhibition. Oxid Med Cell Longev 2018:9286458. https://doi.org/10.1155/2018/9286458
Magrané J, Hervias I, Henning MS, Damiano M, Kawamata H, Manfredi G (2009) Mutant SOD1 in neuronal mitochondria causes toxicity and mitochondrial dynamics abnormalities. Hum Mol Genet 18(23):4552–4564. https://doi.org/10.1093/hmg/ddp421
Mao RW, He SP, Lan JG, Zhu WZ (2022) Honokiol ameliorates cisplatin-induced acute kidney injury via inhibition of mitochondrial fission. Br J Pharmacol 179(14):3886–3904. https://doi.org/10.1111/bph.15837
Marchi N, Granata T, Janigro D (2014) Inflammatory pathways of seizure disorders. Trends Neurosci 37(2):55–65. https://doi.org/10.1016/j.tins.2013.11.002
Meyer JN, Leuthner TC, Luz AL (2017) Mitochondrial fusion, fission, and mitochondrial toxicity. Toxicology 391:42–53. https://doi.org/10.1016/j.tox.2017.07.019
Morigi M, Perico L, Benigni A (2018) Sirtuins in renal health and disease. J Am Soc Nephrol 29(7):1799–1809. https://doi.org/10.1681/asn.2017111218
Naia L, Carmo C, Campesan S, Fão L, Cotton VE, Valero J, Lopes C, Rosenstock TR, Giorgini F, Rego AC (2021) Mitochondrial SIRT3 confers neuroprotection in Huntington’s disease by regulation of oxidative challenges and mitochondrial dynamics. Free Radic Biol Med 163:163–179. https://doi.org/10.1016/j.freeradbiomed.2020.11.031
Nakajima K, Kohsaka S (2004) Microglia: neuroprotective and neurotrophic cells in the central nervous system. Curr Drug Targets Cardiovasc Haematol Disord 4(1):65–84. https://doi.org/10.2174/1568006043481284
Netto MB, de Oliveira Junior AN, Goldim M, Mathias K, Fileti ME, da Rosa N, Laurentino AO, de Farias BX, Costa AB, Rezin GT, Fortunato JJ, Giustina AD, Barichello T, Dal-Pizzol F, Petronilho F (2018) Oxidative stress and mitochondrial dysfunction contributes to postoperative cognitive dysfunction in elderly rats. Brain Behav Immun 73:661–669. https://doi.org/10.1016/j.bbi.2018.07.016
Nolfi-Donegan D, Braganza A, Shiva S (2020) Mitochondrial electron transport chain: Oxidative phosphorylation, oxidant production, and methods of measurement. Redox Biol 37:101674. https://doi.org/10.1016/j.redox.2020.101674
North BJ, Sinclair DA (2007) Sirtuins: a conserved key unlocking AceCS activity. Trends Biochem Sci 32(1):1–4. https://doi.org/10.1016/j.tibs.2006.11.002
Novgorodov SA, Riley CL, Keffler JA, Yu J, Kindy MS, Macklin WB, Lombard DB, Gudz TI (2016) SIRT3 deacetylates ceramide synthases: Implications for mitochondrial dysfunction and brain injury. J Biol Chem 291(4):1957–1973. https://doi.org/10.1074/jbc.M115.668228
Oliver D, Reddy PH (2019) Dynamics of dynamin-related protein 1 in alzheimer’s disease and other neurodegenerative diseases. Cells 8(9):961. https://doi.org/10.3390/cells8090961
Oppermann UC, Salim S, Tjernberg LO, Terenius L, Jörnvall H (1999) Binding of amyloid beta-peptide to mitochondrial hydroxyacyl-CoA dehydrogenase (ERAB): regulation of an SDR enzyme activity with implications for apoptosis in Alzheimer’s disease. FEBS Lett 451(3):238–242. https://doi.org/10.1016/s0014-5793(99)00586-4
Palomer X, Román-Azcona MS, Pizarro-Delgado J, Planavila A, Villarroya F, Valenzuela-Alcaraz B, Crispi F, Sepúlveda-Martínez Á, Miguel-Escalada I, Ferrer J, Nistal JF, García R, Davidson MM, Barroso E, Vázquez-Carrera M (2020) SIRT3-mediated inhibition of FOS through histone H3 deacetylation prevents cardiac fibrosis and inflammation. Signal Transduct Target Ther 5(1):14. https://doi.org/10.1038/s41392-020-0114-1
Park JH, Burgess JD, Faroqi AH, DeMeo NN, Fiesel FC, Springer W, Delenclos M, McLean PJ (2020) Alpha-synuclein-induced mitochondrial dysfunction is mediated via a sirtuin 3-dependent pathway. Mol Neurodegener 15(1):5. https://doi.org/10.1186/s13024-019-0349-x
Parzych KR, Klionsky DJ (2014) An overview of autophagy: morphology, mechanism, and regulation. Antioxid Redox Signal 20(3):460–473. https://doi.org/10.1089/ars.2013.5371
Pauletti A, Terrone G, Shekh-Ahmad T, Salamone A, Ravizza T, Rizzi M, Pastore A, Pascente R, Liang LP, Villa BR, Balosso S, Abramov AY, van Vliet EA, Del Giudice E, Aronica E, Patel M, Walker MC, Vezzani A (2019) Targeting oxidative stress improves disease outcomes in a rat model of acquired epilepsy. Brain 142(7):e39. https://doi.org/10.1093/brain/awz130
Pearson-Smith JN, Patel M (2017) Metabolic dysfunction and oxidative stress in epilepsy. Int J Mol Sci 18(11):2365. https://doi.org/10.3390/ijms18112365
Pignatti C, D’Adamo S, Stefanelli C, Flamigni F, Cetrullo S (2020) Nutrients and pathways that regulate health span and life span. Geriatrics (Basel) 5(4):95. https://doi.org/10.3390/geriatrics5040095
Pillai VB, Sundaresan NR, Kim G, Gupta M, Rajamohan SB, Pillai JB, Samant S, Ravindra PV, Isbatan A, Gupta MP (2010) Exogenous NAD blocks cardiac hypertrophic response via activation of the SIRT3-LKB1-AMP-activated kinase pathway. J Biol Chem 285(5):3133–3144. https://doi.org/10.1074/jbc.M109.077271
Qiu X, Brown K, Hirschey MD, Verdin E, Chen D (2010) Calorie restriction reduces oxidative stress by SIRT3-mediated SOD2 activation. Cell Metab 12(6):662–667. https://doi.org/10.1016/j.cmet.2010.11.015
Qureshi AI, Mendelow AD, Hanley DF (2009) Intracerebral haemorrhage. Lancet 373(9675):1632–1644. https://doi.org/10.1016/s0140-6736(09)60371-8
Raedschelders K, Ansley DM, Chen DD (2012) The cellular and molecular origin of reactive oxygen species generation during myocardial ischemia and reperfusion. Pharmacol Ther 133(2):230–255. https://doi.org/10.1016/j.pharmthera.2011.11.004
Rahman M, Nirala NK, Singh A, Zhu LJ, Taguchi K, Bamba T, Fukusaki E, Shaw LM, Lambright DG, Acharya JK, Acharya UR (2014) Drosophila Sirt2/mammalian SIRT3 deacetylates ATP synthase β and regulates complex V activity. J Cell Biol 206(2):289–305. https://doi.org/10.1083/jcb.201404118
Ramesh S, Govindarajulu M, Lynd T, Briggs G, Adamek D, Jones E, Heiner J, Majrashi M, Moore T, Amin R, Suppiramaniam V, Dhanasekaran M (2018) SIRT3 activator Honokiol attenuates β-Amyloid by modulating amyloidogenic pathway. PLoS ONE 13(1):e0190350. https://doi.org/10.1371/journal.pone.0190350
Rangarajan P, Karthikeyan A, Lu J, Ling EA, Dheen ST (2015) Sirtuin 3 regulates Foxo3a-mediated antioxidant pathway in microglia. Neuroscience 311:398–414. https://doi.org/10.1016/j.neuroscience.2015.10.048
Reimer KA, Lowe JE, Rasmussen MM, Jennings RB (1977) The wavefront phenomenon of ischemic cell death. 1. Myocardial infarct size vs duration of coronary occlusion in dogs. Circulation 56(5):786–794. https://doi.org/10.1161/01.cir.56.5.786
Ridet JL, Malhotra SK, Privat A, Gage FH (1997) Reactive astrocytes: cellular and molecular cues to biological function. Trends Neurosci 20(12):570–577. https://doi.org/10.1016/s0166-2236(97)01139-9
Risiglione P, Zinghirino F, Di Rosa MC, Magrì A, Messina A (2021) Alpha-synuclein and mitochondrial dysfunction in parkinson’s disease: The emerging role of VDAC. Biomolecules 11(5):718. https://doi.org/10.3390/biom11050718
Rosenblatt A (2007) Neuropsychiatry of Huntington’s disease. Dialogues Clin Neurosci 9(2):191–197. https://doi.org/10.31887/DCNS.2007.9.2/arosenblatt
Ross CA, Tabrizi SJ (2011) Huntington’s disease: from molecular pathogenesis to clinical treatment. Lancet Neurol 10(1):83–98. https://doi.org/10.1016/s1474-4422(10)70245-3
Samant SA, Zhang HJ, Hong Z, Pillai VB, Sundaresan NR, Wolfgeher D, Archer SL, Chan DC, Gupta MP (2014) SIRT3 deacetylates and activates OPA1 to regulate mitochondrial dynamics during stress. Mol Cell Biol 34(5):807–819. https://doi.org/10.1128/mcb.01483-13
Sasaki S, Iwata M (2007) Mitochondrial alterations in the spinal cord of patients with sporadic amyotrophic lateral sclerosis. J Neuropathol Exp Neurol 66(1):10–16. https://doi.org/10.1097/nen.0b013e31802c396b
Shi T, Wang F, Stieren E, Tong Q (2005) SIRT3, a mitochondrial sirtuin deacetylase, regulates mitochondrial function and thermogenesis in brown adipocytes. J Biol Chem 280(14):13560–13567. https://doi.org/10.1074/jbc.M414670200
Shi Z, Li C, Yin Y, Yang Z, Xue H, Mu N, Wang Y, Liu M, Ma H (2018) Aerobic interval training regulated SIRT3 attenuates high-fat-diet-associated cognitive dysfunction. Biomed Res Int 2018:2708491. https://doi.org/10.1155/2018/2708491
Shimazu T, Hirschey MD, Hua L, Dittenhafer-Reed KE, Schwer B, Lombard DB, Li Y, Bunkenborg J, Alt FW, Denu JM, Jacobson MP, Verdin E (2010) SIRT3 deacetylates mitochondrial 3-hydroxy-3-methylglutaryl CoA synthase 2 and regulates ketone body production. Cell Metab 12(6):654–661. https://doi.org/10.1016/j.cmet.2010.11.003
Shorvon SD (2011) The etiologic classification of epilepsy. Epilepsia 52(6):1052–1057. https://doi.org/10.1111/j.1528-1167.2011.03041.x
Shu M, Zhang W, Jin X, Zeng L, Xiang Y (2020) Sirt3 gene knockout protects mice from Alzheimer’s disease through activating autophagy. Zhejiang Da Xue Xue Bao Yi Xue Ban 49(6):750–757. https://doi.org/10.3785/j.issn.1008-9292.2020.12.10
Sidorova-Darmos E, Sommer R, Eubanks JH (2018) The role of SIRT3 in the brain under physiological and pathological conditions. Front Cell Neurosci 12:196. https://doi.org/10.3389/fncel.2018.00196
Silaghi CN, Farcaș M, Crăciun AM (2021) Sirtuin 3 (SIRT3) Pathways in age-related cardiovascular and neurodegenerative diseases. Biomedicines 9(11):1574. https://doi.org/10.3390/biomedicines9111574
Sloan JL, Johnston JJ, Manoli I, Chandler RJ, Krause C, Carrillo-Carrasco N, Chandrasekaran SD, Sysol JR, O’Brien K, Hauser NS, Sapp JC, Dorward HM, Huizing M, Barshop BA, Berry SA, James PM, Champaigne NL, de Lonlay P, Valayannopoulos V, Geschwind MD, Gavrilov DK, Nyhan WL, Biesecker LG, Venditti CP (2011) Exome sequencing identifies ACSF3 as a cause of combined malonic and methylmalonic aciduria. Nat Genet 43(9):883–886. https://doi.org/10.1038/ng.908
Smolková K, Špačková J, Gotvaldová K, Dvořák A, Křenková A, Hubálek M, Holendová B, Vítek L, Ježek P (2020) SIRT3 and GCN5L regulation of NADP+- and NADPH-driven reactions of mitochondrial isocitrate dehydrogenase IDH2. Sci Rep 10(1):8677. https://doi.org/10.1038/s41598-020-65351-z
Someya S, Yu W, Hallows WC, Xu J, Vann JM, Leeuwenburgh C, Tanokura M, Denu JM, Prolla TA (2010) Sirt3 mediates reduction of oxidative damage and prevention of age-related hearing loss under caloric restriction. Cell 143(5):802–812. https://doi.org/10.1016/j.cell.2010.10.002
Song CL, Tang H, Ran LK, Ko BC, Zhang ZZ, Chen X, Ren JH, Tao NN, Li WY, Huang AL, Chen J (2016) Sirtuin 3 inhibits hepatocellular carcinoma growth through the glycogen synthase kinase-3β/BCL2-associated X protein-dependent apoptotic pathway. Oncogene 35(5):631–641. https://doi.org/10.1038/onc.2015.121
Song J, Lu C, Zhao W, Shao X (2019) Melatonin attenuates TNF-α-mediated hepatocytes damage via inhibiting mitochondrial stress and activating the Akt-Sirt3 signaling pathway. J Cell Physiol 234(11):20969–20979. https://doi.org/10.1002/jcp.28701
Song S, Li B, Jia Z, Guo L (2020) Sirtuin 3 mRNA Expression is downregulated in the brain tissues of alzheimer’s disease patients: A bioinformatic and data mining approach. Med Sci Monit 26:e923547. https://doi.org/10.12659/msm.923547
Song W, Song Y, Kincaid B, Bossy B, Bossy-Wetzel E (2013) Mutant SOD1G93A triggers mitochondrial fragmentation in spinal cord motor neurons: neuroprotection by SIRT3 and PGC-1α. Neurobiol Dis 51:72–81. https://doi.org/10.1016/j.nbd.2012.07.004
Sporns PB, Psychogios MN, Boulouis G, Charidimou A, Li Q, Fainardi E, Dowlatshahi D, Goldstein JN, Morotti A (2021) Neuroimaging of acute intracerebral hemorrhage. J Clin Med 10(5):1086. https://doi.org/10.3390/jcm10051086
Su J, Liu J, Yan XY, Zhang Y, Zhang JJ, Zhang LC, Sun LK (2017) Cytoprotective effect of the UCP2-SIRT3 signaling pathway by decreasing mitochondrial oxidative stress on cerebral ischemia-reperfusion injury. Int J Mol Sci 18(7):1599. https://doi.org/10.3390/ijms18071599
Sun F, Si Y, Bao H, Xu Y, Pan X, Zeng L, Jing L (2017) Regulation of sirtuin 3-mediated deacetylation of cyclophilin D attenuated cognitive dysfunction induced by sepsis-associated encephalopathy in mice. Cell Mol Neurobiol 37(8):1457–1464. https://doi.org/10.1007/s10571-017-0476-2
Sun R, Kang X, Zhao Y, Wang Z, Wang R, Fu R, Li Y, Hu Y, Wang Z, Shan W, Zhou J, Tian X, Yao J (2020) Sirtuin 3-mediated deacetylation of acyl-CoA synthetase family member 3 by protocatechuic acid attenuates non-alcoholic fatty liver disease. Br J Pharmacol 177(18):4166–4180. https://doi.org/10.1111/bph.15159
Sun Y, Teng Z, Sun X, Zhang L, Chen J, Wang B, Lu F, Liu N, Yu M, Peng S, Wang Y, Zhao D, Zhao Y, Ren H, Cheng Z, Dong S, Lu F, Zhang W (2019) Exogenous H(2)S reduces the acetylation levels of mitochondrial respiratory enzymes via regulating the NAD(+)-SIRT3 pathway in cardiac tissues of db/db mice. Am J Physiol Endocrinol Metab 317(2):E284-e297. https://doi.org/10.1152/ajpendo.00326.2018
Sundaresan NR, Samant SA, Pillai VB, Rajamohan SB, Gupta MP (2008) SIRT3 is a stress-responsive deacetylase in cardiomyocytes that protects cells from stress-mediated cell death by deacetylation of Ku70. Mol Cell Biol 28(20):6384–6401. https://doi.org/10.1128/mcb.00426-08
Taylor JP, Brown RH Jr, Cleveland DW (2016) Decoding ALS: from genes to mechanism. Nature 539(7628):197–206. https://doi.org/10.1038/nature20413
Thangaraj A, Chivero ET, Tripathi A, Singh S, Niu F, Guo ML, Pillai P, Periyasamy P, Buch S (2021) HIV TAT-mediated microglial senescence: Role of SIRT3-dependent mitochondrial oxidative stress. Redox Biol 40:101843. https://doi.org/10.1016/j.redox.2020.101843
Todorova V, Blokland A (2017) Mitochondria and synaptic plasticity in the mature and aging nervous system. Curr Neuropharmacol 15(1):166–173. https://doi.org/10.2174/1570159x14666160414111821
Tolosa E, Wenning G, Poewe W (2006) The diagnosis of Parkinson’s disease. Lancet Neurol 5(1):75–86. https://doi.org/10.1016/s1474-4422(05)70285-4
Traba J, Geiger SS, Kwarteng-Siaw M, Han K, Ra OH, Siegel RM, Gius D, Sack MN (2017) Prolonged fasting suppresses mitochondrial NLRP3 inflammasome assembly and activation via SIRT3-mediated activation of superoxide dismutase 2. J Biol Chem 292(29):12153–12164. https://doi.org/10.1074/jbc.M117.791715
Traba J, Kwarteng-Siaw M, Okoli TC, Li J, Huffstutler RD, Bray A, Waclawiw MA, Han K, Pelletier M, Sauve AA, Siegel RM, Sack MN (2015) Fasting and refeeding differentially regulate NLRP3 inflammasome activation in human subjects. J Clin Invest 125(12):4592–4600. https://doi.org/10.1172/jci83260
Tseng AH, Shieh SS, Wang DL (2013) SIRT3 deacetylates FOXO3 to protect mitochondria against oxidative damage. Free Radic Biol Med 63:222–234. https://doi.org/10.1016/j.freeradbiomed.2013.05.002
Tseng AH, Wu LH, Shieh SS, Wang DL (2014) SIRT3 interactions with FOXO3 acetylation, phosphorylation and ubiquitinylation mediate endothelial cell responses to hypoxia. Biochem J 464(1):157–168. https://doi.org/10.1042/bj20140213
Tsukiyama Y, Ito T, Nagaoka K, Eguchi E, Ogino K (2017) Effects of exercise training on nitric oxide, blood pressure and antioxidant enzymes. J Clin Biochem Nutr 60(3):180–186. https://doi.org/10.3164/jcbn.16-108
Tu LF, Cao LF, Zhang YH, Guo YL, Zhou YF, Lu WQ, Zhang TZ, Zhang T, Zhang GX, Kurihara H, Li YF, He RR (2019) Sirt3-dependent deacetylation of COX-1 counteracts oxidative stress-induced cell apoptosis. Faseb j 33(12):14118–14128. https://doi.org/10.1096/fj.201900708R
Tyagi A, Nguyen CU, Chong T, Michel CR, Fritz KS, Reisdorph N, Knaub L, Reusch JEB, Pugazhenthi S (2018) SIRT3 deficiency-induced mitochondrial dysfunction and inflammasome formation in the brain. Sci Rep 8(1):17547. https://doi.org/10.1038/s41598-018-35890-7
Tysnes OB, Storstein A (2017) Epidemiology of Parkinson’s disease. J Neural Transm (vienna) 124(8):901–905. https://doi.org/10.1007/s00702-017-1686-y
van Asch CJ, Luitse MJ, Rinkel GJ, van der Tweel I, Algra A, Klijn CJ (2010) Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. Lancet Neurol 9(2):167–176. https://doi.org/10.1016/s1474-4422(09)70340-0
van der Bliek AM, Shen Q, Kawajiri S (2013) Mechanisms of mitochondrial fission and fusion. Cold Spring Harb Perspect Biol 5(6):a011072. https://doi.org/10.1101/cshperspect.a011072
Vargas-Ortiz K, Perez-Vazquez V, Diaz-Cisneros FJ, Figueroa A, Jiménez-Flores LM, Rodriguez-DelaRosa G, Macias MH (2015) Aerobic training increases expression levels of SIRT3 and PGC-1α in skeletal muscle of overweight adolescents without change in caloric intake. Pediatr Exerc Sci 27(2):177–184. https://doi.org/10.1123/pes.2014-0112
Vargas-Ortiz K, Pérez-Vázquez V, Figueroa A, Díaz FJ, Montaño-Ascencio PG, Macías-Cervantes MH (2018) Aerobic training but no resistance training increases SIRT3 in skeletal muscle of sedentary obese male adolescents. Eur J Sport Sci 18(2):226–234. https://doi.org/10.1080/17461391.2017.1406007
Vezzani A, French J, Bartfai T, Baram TZ (2011) The role of inflammation in epilepsy. Nat Rev Neurol 7(1):31–40. https://doi.org/10.1038/nrneurol.2010.178
Villemagne VL, Fodero-Tavoletti MT, Masters CL, Rowe CC (2015) Tau imaging: early progress and future directions. Lancet Neurol 14(1):114–124. https://doi.org/10.1016/s1474-4422(14)70252-2
Wan X, Wang C, Huang Z, Zhou D, Xiang S, Qi Q, Chen X, Arbely E, Liu CY, Du P, Yu W (2020) Cisplatin inhibits SIRT3-deacetylation MTHFD2 to disturb cellular redox balance in colorectal cancer cell. Cell Death Dis 11(8):649. https://doi.org/10.1038/s41419-020-02825-y
Wang BH, Hou Q, Lu YQ, Jia MM, Qiu T, Wang XH, Zhang ZX, Jiang Y (2018) Ketogenic diet attenuates neuronal injury via autophagy and mitochondrial pathways in pentylenetetrazol-kindled seizures. Brain Res 1678:106–115. https://doi.org/10.1016/j.brainres.2017.10.009
Wang N, Xu F, Lu S, Zhang N, Sun Y (2020a) Septin4 as an autophagy modulator regulates Angiotensin-II mediated VSMCs proliferation and migration. Biochem Biophys Res Commun 525(2):272–279. https://doi.org/10.1016/j.bbrc.2020.02.064
Wang Q, Li L, Li CY, Pei Z, Zhou M, Li N (2015) SIRT3 protects cells from hypoxia via PGC-1α- and MnSOD-dependent pathways. Neuroscience 286:109–121. https://doi.org/10.1016/j.neuroscience.2014.11.045
Wang XQ, Shao Y, Ma CY, Chen W, Sun L, Liu W, Zhang DY, Fu BC, Liu KY, Jia ZB, Xie BD, Jiang SL, Li RK, Tian H (2014) Decreased SIRT3 in aged human mesenchymal stromal/stem cells increases cellular susceptibility to oxidative stress. J Cell Mol Med 18(11):2298–2310. https://doi.org/10.1111/jcmm.12395
Wang Z, Sun R, Wang G, Chen Z, Li Y, Zhao Y, Liu D, Zhao H, Zhang F, Yao J, Tian X (2020b) SIRT3-mediated deacetylation of PRDX3 alleviates mitochondrial oxidative damage and apoptosis induced by intestinal ischemia/reperfusion injury. Redox Biol 28:101343. https://doi.org/10.1016/j.redox.2019.101343
Wegman MP, Guo MH, Bennion DM, Shankar MN, Chrzanowski SM, Goldberg LA, Xu J, Williams TA, Lu X, Hsu SI, Anton SD, Leeuwenburgh C, Brantly ML (2015) Practicality of intermittent fasting in humans and its effect on oxidative stress and genes related to aging and metabolism. Rejuvenation Res 18(2):162–172. https://doi.org/10.1089/rej.2014.1624
Wei L, Zhou Y, Dai Q, Qiao C, Zhao L, Hui H, Lu N, Guo QL (2013a) Oroxylin A induces dissociation of hexokinase II from the mitochondria and inhibits glycolysis by SIRT3-mediated deacetylation of cyclophilin D in breast carcinoma. Cell Death Dis 4(4):e601. https://doi.org/10.1038/cddis.2013.131
Wei SJ, Xing JH, Wang BL, Xue L, Wang JL, Li R, Qin WD, Wang J, Wang XP, Zhang MX, Chen YG (2013b) Poly(ADP-ribose) polymerase inhibition prevents reactive oxygen species induced inhibition of aldehyde dehydrogenase2 activity. Biochim Biophys Acta 1833(3):479–486. https://doi.org/10.1016/j.bbamcr.2012.11.007
Wild EJ, Tabrizi SJ (2017) Therapies targeting DNA and RNA in Huntington’s disease. Lancet Neurol 16(10):837–847. https://doi.org/10.1016/s1474-4422(17)30280-6
Woods A, Johnstone SR, Dickerson K, Leiper FC, Fryer LG, Neumann D, Schlattner U, Wallimann T, Carlson M, Carling D (2003) LKB1 is the upstream kinase in the AMP-activated protein kinase cascade. Curr Biol 13(22):2004–2008. https://doi.org/10.1016/j.cub.2003.10.031
Wu B, You S, Qian H, Wu S, Lu S, Zhang Y, Sun Y, Zhang N (2021) The role of SIRT2 in vascular-related and heart-related diseases: A review. J Cell Mol Med. https://doi.org/10.1111/jcmm.16618
Wu G, Liu J, Li S, Gao W, Qiu M, Yang C, Ma Y, Song X (2020a) Glycyrrhizic acid protects juvenile epileptic rats against hippocampal damage through activation of Sirtuin3. Brain Res Bull 164:98–106. https://doi.org/10.1016/j.brainresbull.2020.08.008
Wu H, Yu N, Wang X, Yang Y, Liang H (2020b) Tauroursodeoxycholic acid attenuates neuronal apoptosis via the TGR5/ SIRT3 pathway after subarachnoid hemorrhage in rats. Biol Res 53(1):56. https://doi.org/10.1186/s40659-020-00323-1
Wu X, Luo J, Liu H, Cui W, Feng D, Qu Y (2020c) SIRT3 protects against early brain injury following subarachnoid hemorrhage via promoting mitochondrial fusion in an AMPK dependent manner. Chin Neurosurg J 6:1. https://doi.org/10.1186/s41016-019-0182-7
Xu LX, Hao LJ, Ma JQ, Liu JK, Hasim A (2020) SIRT3 promotes the invasion and metastasis of cervical cancer cells by regulating fatty acid synthase. Mol Cell Biochem 464(1–2):11–20. https://doi.org/10.1007/s11010-019-03644-2
Xue RQ, Yu XJ, Zhao M, Xu M, Wu Q, Cui YL, Yang S, Li DL, Zang WJ (2019) Pyridostigmine alleviates cardiac dysfunction via improving mitochondrial cristae shape in a mouse model of metabolic syndrome. Free Radic Biol Med 134:119–132. https://doi.org/10.1016/j.freeradbiomed.2019.01.011
Yan SD, Fu J, Soto C, Chen X, Zhu H, Al-Mohanna F, Collison K, Zhu A, Stern E, Saido T, Tohyama M, Ogawa S, Roher A, Stern D (1997) An intracellular protein that binds amyloid-beta peptide and mediates neurotoxicity in Alzheimer’s disease. Nature 389(6652):689–695. https://doi.org/10.1038/39522
Yang H, Zhou L, Shi Q, Zhao Y, Lin H, Zhang M, Zhao S, Yang Y, Ling ZQ, Guan KL, Xiong Y, Ye D (2015a) SIRT3-dependent GOT2 acetylation status affects the malate-aspartate NADH shuttle activity and pancreatic tumor growth. Embo j 34(8):1110–1125. https://doi.org/10.15252/embj.201591041
Yang S, Chen X, Li S, Sun B, Hang C (2018a) Melatonin Treatment Regulates SIRT3 Expression in Early Brain Injury (EBI) Due to Reactive Oxygen Species (ROS) in a Mouse Model of Subarachnoid Hemorrhage (SAH). Med Sci Monit 24:3804–3814. https://doi.org/10.12659/msm.907734
Yang W, Nagasawa K, Münch C, Xu Y, Satterstrom K, Jeong S, Hayes SD, Jedrychowski MP, Vyas FS, Zaganjor E, Guarani V, Ringel AE, Gygi SP, Harper JW, Haigis MC (2016) Mitochondrial sirtuin network reveals dynamic SIRT3-dependent deacetylation in response to membrane depolarization. Cell 167(4):985-1000.e1021. https://doi.org/10.1016/j.cell.2016.10.016
Yang W, Zou Y, Zhang M, Zhao N, Tian Q, Gu M, Liu W, Shi R, Lü Y, Yu W (2015b) Mitochondrial sirt3 expression is decreased in APP/PS1 double transgenic mouse model of alzheimer’s disease. Neurochem Res 40(8):1576–1582. https://doi.org/10.1007/s11064-015-1630-1
Yang X, Geng K, Zhang J, Zhang Y, Shao J, Xia W (2017) Sirt3 mediates the inhibitory effect of adjudin on astrocyte activation and glial scar formation following ischemic stroke. Front Pharmacol 8:943. https://doi.org/10.3389/fphar.2017.00943
Yang X, Geng KY, Zhang YS, Zhang JF, Yang K, Shao JX, Xia WL (2018b) Sirt3 deficiency impairs neurovascular recovery in ischemic stroke. CNS Neurosci Ther 24(9):775–783. https://doi.org/10.1111/cns.12853
Yang Z, Klionsky DJ (2009) An overview of the molecular mechanism of autophagy. Curr Top Microbiol Immunol 335:1–32. https://doi.org/10.1007/978-3-642-00302-8_1
Ye JS, Chen L, Lu YY, Lei SQ, Peng M, Xia ZY (2019) SIRT3 activator honokiol ameliorates surgery/anesthesia-induced cognitive decline in mice through anti-oxidative stress and anti-inflammatory in hippocampus. CNS Neurosci Ther 25(3):355–366. https://doi.org/10.1111/cns.13053
Yin J, Han P, Song M, Nielsen M, Beach TG, Serrano GE, Liang WS, Caselli RJ, Shi J (2018) Amyloid-β increases tau by mediating sirtuin 3 in alzheimer’s disease. Mol Neurobiol 55(11):8592–8601. https://doi.org/10.1007/s12035-018-0977-0
Yin J, Han P, Tang Z, Liu Q, Shi J (2015) Sirtuin 3 mediates neuroprotection of ketones against ischemic stroke. J Cereb Blood Flow Metab 35(11):1783–1789. https://doi.org/10.1038/jcbfm.2015.123
Yoon SP, Kim J (2016) Poly(ADP-ribose) polymerase 1 contributes to oxidative stress through downregulation of sirtuin 3 during cisplatin nephrotoxicity. Anat Cell Biol 49(3):165–176. https://doi.org/10.5115/acb.2016.49.3.165
Yu W, Gao B, Li N, Wang J, Qiu C, Zhang G, Liu M, Zhang R, Li C, Ji G, Zhang Y (2017) Sirt3 deficiency exacerbates diabetic cardiac dysfunction: Role of Foxo3A-Parkin-mediated mitophagy. Biochim Biophys Acta Mol Basis Dis 1863(8):1973–1983. https://doi.org/10.1016/j.bbadis.2016.10.021
Yu W, Qin J, Chen C, Fu Y, Wang W (2018) Moderate calorie restriction attenuates age-associated alterations and improves cardiac function by increasing SIRT1 and SIRT3 expression. Mol Med Rep 18(4):4087–4094. https://doi.org/10.3892/mmr.2018.9390
Zhang B, Cui S, Bai X, Zhuo L, Sun X, Hong Q, Fu B, Wang J, Chen X, Cai G (2013) SIRT3 overexpression antagonizes high glucose accelerated cellular senescence in human diploid fibroblasts via the SIRT3-FOXO1 signaling pathway. Age (dordr) 35(6):2237–2253. https://doi.org/10.1007/s11357-013-9520-4
Zhang DY, Zhang CF, Fu BC, Sun L, Wang XQ, Chen W, Liu W, Liu KY, Du GQ, Ma CY, Jiang SL, Li RK, Tian H (2018a) Sirtuin3 protects aged human mesenchymal stem cells against oxidative stress and enhances efficacy of cell therapy for ischaemic heart diseases. J Cell Mol Med 22(11):5504–5517. https://doi.org/10.1111/jcmm.13821
Zhang J, Xiang H, Liu J, Chen Y, He RR, Liu B (2020a) Mitochondrial Sirtuin 3: New emerging biological function and therapeutic target. Theranostics 10(18):8315–8342. https://doi.org/10.7150/thno.45922
Zhang K, Cheng H, Song L, Wei W (2020b) Inhibition of the peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α)/sirtuin 3 (SIRT3) pathway aggravates oxidative stress after experimental subarachnoid hemorrhage. Med Sci Monit 26:e923688. https://doi.org/10.12659/msm.923688
Zhang M, Deng YN, Zhang JY, Liu J, Li YB, Su H, Qu QM (2018b) SIRT3 protects rotenone-induced injury in SH-SY5Y cells by promoting autophagy through the LKB1-AMPK-mTOR pathway. Aging Dis 9(2):273–286. https://doi.org/10.14336/ad.2017.0517
Zhang S, Ma Y, Feng J (2020c) Neuroprotective mechanisms of ε-viniferin in a rotenone-induced cell model of Parkinson’s disease: significance of SIRT3-mediated FOXO3 deacetylation. Neural Regen Res 15(11):2143–2153. https://doi.org/10.4103/1673-5374.282264
Zhang X, Ren X, Zhang Q, Li Z, Ma S, Bao J, Li Z, Bai X, Zheng L, Zhang Z, Shang S, Zhang C, Wang C, Cao L, Wang Q, Ji J (2016) PGC-1α/ERRα-Sirt3 pathway regulates DAergic neuronal death by directly deacetylating SOD2 and ATP synthase β. Antioxid Redox Signal 24(6):312–328. https://doi.org/10.1089/ars.2015.6403
Zhang Y, Bharathi SS, Rardin MJ, Uppala R, Verdin E, Gibson BW, Goetzman ES (2015) SIRT3 and SIRT5 regulate the enzyme activity and cardiolipin binding of very long-chain acyl-CoA dehydrogenase. PLoS ONE 10(3):e0122297. https://doi.org/10.1371/journal.pone.0122297
Zhang Y, Yang X, Ge X, Zhang F (2019) Puerarin attenuates neurological deficits via Bcl-2/Bax/cleaved caspase-3 and Sirt3/SOD2 apoptotic pathways in subarachnoid hemorrhage mice. Biomed Pharmacother 109:726–733. https://doi.org/10.1016/j.biopha.2018.10.161
Zhang YK, Qu YY, Lin Y, Wu XH, Chen HZ, Wang X, Zhou KQ, Wei Y, Guo F, Yao CF, He XD, Liu LX, Yang C, Guan ZY, Wang SD, Zhao J, Liu DP, Zhao SM, Xu W (2017) Enoyl-CoA hydratase-1 regulates mTOR signaling and apoptosis by sensing nutrients. Nat Commun 8(1):464. https://doi.org/10.1038/s41467-017-00489-5
Zhang Z, Han K, Wang C, Sun C, Jia N (2020d) Dioscin Protects against Aβ1-42 Oligomers-Induced Neurotoxicity via the Function of SIRT3 and Autophagy. Chem Pharm Bull (tokyo) 68(8):717–725. https://doi.org/10.1248/cpb.c20-00046
Zhao E, Tranovich MJ, DeAngelo R, Kontos AP, Wright VJ (2016) Chronic exercise preserves brain function in masters athletes when compared to sedentary counterparts. Phys Sportsmed 44(1):8–13. https://doi.org/10.1080/00913847.2016.1103641
Zhao H, Chen Y, Feng H (2018a) P2X7 Receptor-Associated Programmed Cell Death in the Pathophysiology of Hemorrhagic Stroke. Curr Neuropharmacol 16(9):1282–1295. https://doi.org/10.2174/1570159x16666180516094500
Zhao H, Luo Y, Chen L, Zhang Z, Shen C, Li Y, Xu R (2018b) Sirt3 inhibits cerebral ischemia-reperfusion injury through normalizing Wnt/β-catenin pathway and blocking mitochondrial fission. Cell Stress Chaperones 23(5):1079–1092. https://doi.org/10.1007/s12192-018-0917-y
Zheng J, Shi L, Liang F, Xu W, Li T, Gao L, Sun Z, Yu J, Zhang J (2018) Sirt3 ameliorates oxidative stress and mitochondrial dysfunction after intracerebral hemorrhage in diabetic rats. Front Neurosci 12:414. https://doi.org/10.3389/fnins.2018.00414
Zhou R, Tardivel A, Thorens B, Choi I, Tschopp J (2010) Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat Immunol 11(2):136–140. https://doi.org/10.1038/ni.1831
Acknowledgements
We thank Yanhui He for his support of this review. We also thank LetPub (www.letpub.com) for its linguistic assistance during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Writing – Original Draft Preparation: Yanlin Li; Data Curation: Jing Li, Guangbin Wu, Hua Yang, Xiaosong Yang, Dongyu Wang; Writing – Review & Editing: Yanhui He.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, Y., Li, J., Wu, G. et al. Role of SIRT3 in neurological diseases and rehabilitation training. Metab Brain Dis 38, 69–89 (2023). https://doi.org/10.1007/s11011-022-01111-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-01111-4