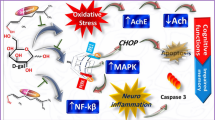
Abstract
Aging is widely thought to be associated with oxidative stress. Momordica charantia (MC) is a classic vegetable and traditional herbal medicine widely consumed in Asia, and M. charantia polysaccharide (MCP) is the main bioactive ingredient of MC. We previously reported an antioxidative and neuroprotective effect of MCP in models of cerebral ischemia/reperfusion and hemorrhage injury. However, the role played by MCP in neurodegenerative diseases, especially during aging, remains unknown. In this study, we investigated the protective effect of MCP against oxidative stress and brain damage in a D-galactose-induced aging model (DGAM). The Morris water maze test was performed to evaluate the spatial memory function of model rats. The levels of malondialdehyde (MDA), glutathione (GSH), and superoxide dismutase (SOD) were measured and telomerase activity was determined. The results showed that MCP treatment attenuated spatial memory dysfunction induced by D-galactose. In addition, MCP increased antioxidant capacity by decreasing MDA and increasing SOD and GSH levels. MCP treatment also improved telomerase activity in aging rats. Mechanistically, MCP promoted the entry of both Nrf2 and β-Catenin into the nucleus, which is the hallmark of antioxidation signaling pathway activation. This study highlights a role played by MCP in ameliorating aging-induced oxidative stress injury and reversing the decline in learning and memory capacity. Our work provides evidence that MCP administration might be a potential antiaging strategy.






Similar content being viewed by others
Data availability
I hereby certify that all material has been included in the text and is freely accessible without limitation and that we do not have any more information.
References
Adnan M, Rasul A, Hussain G, Shah MA, Zahoor MK, Anwar H, Sarfraz I, Riaz A, Manzoor M, Adem Ş, Selamoglu Z (2020) Ginkgetin: A natural biflavone with versatile pharmacological activities. Food Chem Toxicol 145:111642. https://doi.org/10.1016/j.fct.2020.111642
Al Omairi NE, Al-Brakati AY, Kassab RB, Lokman MS, Elmahallawy EK, Amin HK, Abdel Moneim AE (2019) Soursop fruit extract mitigates scopolamine-induced amnesia and oxidative stress via activating cholinergic and Nrf2/HO-1 pathways. Metab Brain Dis 34(3):853–864. https://doi.org/10.1007/s11011-019-00407-2
Ali T, Rehman SU, Shah FA, Kim MO (2018) Acute dose of melatonin via Nrf2 dependently prevents acute ethanol-induced neurotoxicity in the developing rodent brain. J Neuroinflammation 15(1):119. https://doi.org/10.1186/s12974-018-1157-x
Azman KF, Zakaria R (2019) D-Galactose-induced accelerated aging model: an overview. Biogerontology 20(6):763–782. https://doi.org/10.1007/s10522-019-09837-y
Bellezza I, Giambanco I, Minelli A, Donato R (2018) Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim Biophys Acta Mol Cell Res 1865(5):721–733. https://doi.org/10.1016/j.bbamcr.2018.02.010
Chen F, Huang G, Yang Z, Hou Y (2019) Antioxidant activity of Momordica charantia polysaccharide and its derivatives. Int J Biol Macromol 138:673–680. https://doi.org/10.1016/j.ijbiomac.2019.07.129
Clevers H, Nusse R (2012) Wnt/β-catenin signaling and disease. Cell 149(6):1192–1205. https://doi.org/10.1016/j.cell.2012.05.012
Deng YY, Yi Y, Zhang LF, Zhang RF, Zhang Y, Wei ZC, Tang XJ, Zhang MW (2014) Immunomodulatory activity and partial characterisation of polysaccharides from Momordica charantia. Molecules 19(9):13432–13447. https://doi.org/10.3390/molecules190913432
Duan ZZ, Zhou XL, Li YH, Zhang F, Li FY, Su-Hua Q (2015) Protection of Momordica charantia polysaccharide against intracerebral hemorrhage-induced brain injury through JNK3 signaling pathway. J Recept Signal Transduct Res 35(6):523–529. https://doi.org/10.3109/10799893.2014.963871
Franceschi C, Garagnani P, Parini P, Giuliani C, Santoro A (2018) Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat Rev Endocrinol 14(10):576–590. https://doi.org/10.1038/s41574-018-0059-4
Gao M, Kang Y, Zhang L, Li H, Qu C, Luan X, Liu L, Zhang S (2020) Troxerutin attenuates cognitive decline in the hippocampus of male diabetic rats by inhibiting NADPH oxidase and activating the Nrf2/ARE signaling pathway. Int J Mol Med 46(3):1239–1248. https://doi.org/10.3892/ijmm.2020.4653
Ge M, Yao W, Yuan D, Zhou S, Chen X, Zhang Y, Li H, Xia Z, Hei Z (2017) Brg1-mediated Nrf2/HO-1 pathway activation alleviates hepatic ischemia-reperfusion injury. Cell Death Dis 8(6):e2841. https://doi.org/10.1038/cddis.2017.236
Godoy JA, Rios JA, Picón-Pagès P, Herrera-Fernández V, Swaby B, Crepin G, Vicente R, Fernández-Fernández JM, Muñoz FJ (2021) Mitostasis, calcium and free radicals in health, aging and neurodegeneration. Biomolecules 11(7). https://doi.org/10.3390/biom11071012
Gong J, Sun F, Li Y, Zhou X, Duan Z, Duan F, Zhao L, Chen H, Qi S, Shen J (2015) Momordica charantia polysaccharides could protect against cerebral ischemia/reperfusion injury through inhibiting oxidative stress mediated c-Jun N-terminal kinase 3 signaling pathway. Neuropharmacology 91:123–134. https://doi.org/10.1016/j.neuropharm.2014.11.020
Gorbunova V, Rezazadeh S, Seluanov A (2016) Dangerous Entrapment for NRF2. Cell 165(6):1312–1313. https://doi.org/10.1016/j.cell.2016.05.061
Guan L (2012) Synthesis and anti-tumour activities of sulphated polysaccharide obtained from Momordica charantia. Nat Prod Res 26(14):1303–1309. https://doi.org/10.1080/14786419.2011.571214
Haider K, Haider MR, Neha K, Yar MS (2020) Free radical scavengers: An overview on heterocyclic advances and medicinal prospects. Eur J Med Chem 204:112607. https://doi.org/10.1016/j.ejmech.2020.112607
Harvey AL, Edrada-Ebel R, Quinn RJ (2015) The re-emergence of natural products for drug discovery in the genomics era. Nat Rev Drug Discov 14(2):111–129. https://doi.org/10.1038/nrd4510
Holt SE, Shay JW, Wright WE (1996) Refining the telomere-telomerase hypothesis of aging and cancer. Nat Biotechnol 14(7):836–839. https://doi.org/10.1038/nbt0796-836
Hu Z, Li F, Zhou X, Zhang F, Huang L, Gu B, Shen J, Qi S (2020) Momordica charantia polysaccharides modulate the differentiation of neural stem cells via SIRT1/Β-catenin axis in cerebral ischemia/reperfusion. Stem Cell Res Ther 11(1):485. https://doi.org/10.1186/s13287-020-02000-2
Liochev SI (2013) Reactive oxygen species and the free radical theory of aging. Free Radic Biol Med 60:1–4. https://doi.org/10.1016/j.freeradbiomed.2013.02.011
Lv H, Zhu C, Wei W, Lv X, Yu Q, Deng X, Ci X (2020) Enhanced Keap1-Nrf2/Trx-1 axis by daphnetin protects against oxidative stress-driven hepatotoxicity via inhibiting ASK1/JNK and Txnip/NLRP3 inflammasome activation. Phytomedicine 71:153241. https://doi.org/10.1016/j.phymed.2020.153241
Ma J, Fan H, Cai H, Hu Z, Zhou X, Li F, Chen H, Shen J, Qi S (2021) Promotion of Momordica Charantia polysaccharides on neural stem cell proliferation by increasing SIRT1 activity after cerebral ischemia/reperfusion in rats. Brain Res Bull 170:254–263. https://doi.org/10.1016/j.brainresbull.2021.02.016
MacDonald BT, Tamai K, He X (2009) Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 17(1):9–26. https://doi.org/10.1016/j.devcel.2009.06.016
Malik I, Shah FA, Ali T, Tan Z, Alattar A, Ullah N, Khan AU, Alshaman R, Li S (2020) Potent natural antioxidant carveol attenuates MCAO-stress induced oxidative, neurodegeneration by regulating the Nrf-2 pathway. Front Neurosci 14:659. https://doi.org/10.3389/fnins.2020.00659
Marchetti B, L’Episcopo F, Morale MC, Tirolo C, Testa N, Caniglia S, Serapide MF, Pluchino S (2013) Uncovering novel actors in astrocyte-neuron crosstalk in Parkinson’s disease: the Wnt/β-catenin signaling cascade as the common final pathway for neuroprotection and self-repair. Eur J Neurosci 37(10):1550–1563. https://doi.org/10.1111/ejn.12166
Marchetti B, Tirolo C, L’Episcopo F, Caniglia S, Testa N, Smith JA, Pluchino S, Serapide MF (2020) Parkinson’s disease, aging and adult neurogenesis: Wnt/β-catenin signalling as the key to unlock the mystery of endogenous brain repair. Aging Cell 19(3):e13101. https://doi.org/10.1111/acel.13101
Melzer D, Pilling LC, Ferrucci L (2020) The genetics of human ageing. Nat Rev Genet 21(2):88–101. https://doi.org/10.1038/s41576-019-0183-6
Méndez-Armenta M, Nava-Ruíz C, Juárez-Rebollar D, Rodríguez-Martínez E, Gómez PY (2014) Oxidative stress associated with neuronal apoptosis in experimental models of epilepsy. Oxid Med Cell Longev 2014:293689. https://doi.org/10.1155/2014/293689
Naeem K, Tariq Al Kury L, Nasar F, Alattar A, Alshaman R, Shah FA, Khan AU, Li S (2021) Natural dietary supplement, carvacrol, alleviates LPS-induced oxidative stress, neurodegeneration, and depressive-like behaviors via the Nrf2/HO-1 pathway. J Inflamm Res 14:1313–1329. https://doi.org/10.2147/jir.S294413
Panahzadeh F, Mirnasuri R, Rahmati M (2022) Exercise and Syzygium aromaticum reverse memory deficits, apoptosis and mitochondrial dysfunction of the hippocampus in Alzheimer’s disease. J Ethnopharmacol 286:114871. https://doi.org/10.1016/j.jep.2021.114871
Pomatto LCD, Davies KJA (2018) Adaptive homeostasis and the free radical theory of ageing. Free Radic Biol Med 124:420–430. https://doi.org/10.1016/j.freeradbiomed.2018.06.016
Reuter S, Gupta SC, Chaturvedi MM, Aggarwal BB (2010) Oxidative stress, inflammation, and cancer: how are they linked? Free Radic Biol Med 49(11):1603–1616. https://doi.org/10.1016/j.freeradbiomed.2010.09.006
Ru Y, Liu K, Kong X, Li X, Shi X, Chen H (2020) Synthesis of selenylated polysaccharides from Momordica charantia L. and its hypoglycemic activity in streptozotocin-induced diabetic mice. Int J Biol Macromol 152:295–304. https://doi.org/10.1016/j.ijbiomac.2020.02.288
Safdar A, Zakaria R, Aziz CBA, Rashid U, Azman KF (2020) Goat milk attenuates mimetic aging related memory impairment via suppressing brain oxidative stress, neurodegeneration and modulating neurotrophic factors in D-galactose-induced aging model. Biogerontology 21(2):203–216. https://doi.org/10.1007/s10522-019-09854-x
Sarfraz A, Javeed M, Shah MA, Hussain G, Shafiq N, Sarfraz I, Riaz A, Sadiqa A, Zara R, Zafar S, Kanwal L, Sarker SD, Rasul A (2020) Biochanin A: A novel bioactive multifunctional compound from nature. Sci Total Environ 722:137907. https://doi.org/10.1016/j.scitotenv.2020.137907
Savall M, Senni N, Lagoutte I, Sohier P, Dentin R, Romagnolo B, Perret C, Bossard P (2021) Cooperation between the NRF2 pathway and oncogenic β-catenin during HCC tumorigenesis. Hepatol Commun 5(9):1490–1506. https://doi.org/10.1002/hep4.1746
Schunk SJ, Floege J, Fliser D, Speer T (2021) WNT-β-catenin signalling - a versatile player in kidney injury and repair. Nat Rev Nephrol 17(3):172–184. https://doi.org/10.1038/s41581-020-00343-w
Shu XS, Zhu H, Huang X, Yang Y, Wang D, Zhang Y, Zhang W, Ying Y (2020) Loss of β-catenin via activated GSK3β causes diabetic retinal neurodegeneration by instigating a vicious cycle of oxidative stress-driven mitochondrial impairment. Aging 12(13):13437–13462. https://doi.org/10.18632/aging.103446
Torres-Cuevas I, Corral-Debrinski M, Gressens P (2019) Brain oxidative damage in murine models of neonatal hypoxia/ischemia and reoxygenation. Free Radic Biol Med 142:3–15. https://doi.org/10.1016/j.freeradbiomed.2019.06.011
Uddin MS, Hossain MF, Mamun AA, Shah MA, Hasana S, Bulbul IJ, Sarwar MS, Mansouri RA, Ashraf GM, Rauf A, Abdel-Daim MM, Bin-Jumah MN (2020) Exploring the multimodal role of phytochemicals in the modulation of cellular signaling pathways to combat age-related neurodegeneration. Sci Total Environ 725:138313. https://doi.org/10.1016/j.scitotenv.2020.138313
Wang Y, Wang Q, Li J, Lu G, Liu Z (2019) Glutamine improves oxidative stress through the Wnt3a/β-catenin signaling pathway in Alzheimer’s disease in vitro and in vivo. Biomed Res Int 2019:4690280. https://doi.org/10.1155/2019/4690280
Wheeler HE, Kim SK (2011) Genetics and genomics of human ageing. Philos Trans R Soc Lond B Biol Sci 366(1561):43–50. https://doi.org/10.1098/rstb.2010.0259
Yu C, Xiao JH (2021) The Keap1-Nrf2 System: a mediator between oxidative stress and aging. Oxid Med Cell Longev 2021:6635460. https://doi.org/10.1155/2021/6635460
Yu L, Liu Y, Jin Y, Cao X, Chen J, Jin J, Gu Y, Bao X, Ren Z, Xu Y, Zhu X (2018) Lentivirus-mediated HDAC3 inhibition attenuates oxidative stress in APPswe/PS1dE9 mice. J Alzheimers Dis 61(4):1411–1424. https://doi.org/10.3233/jad-170844
Zafar S, Sarfraz I, Rasul A, Shah MA, Hussain G, Zahoor MK, Shafiq N, Riaz A, Selamoglu Z, Sarker SD (2021) Osthole: a multifunctional natural compound with potential anticancer, antioxidant and anti-inflammatory activities. Mini Rev Med Chem 21(18):2747–2763. https://doi.org/10.2174/1389557520666200709175948
Zhi SM, Fang GX, Xie XM, Liu LH, Yan J, Liu DB, Yu HY (2020) Melatonin reduces OGD/R-induced neuron injury by regulating redox/inflammation/apoptosis signaling. Eur Rev Med Pharmacol Sci 24(3):1524–1536. https://doi.org/10.26355/eurrev_202002_20211
Zhu D, Zhang X, Wang F, Ye Q, Yang C, Liu D (2022) Irisin rescues diabetic cardiac microvascular injury via ERK1/2/Nrf2/HO-1 mediated inhibition of oxidative stress. Diabetes Res Clin Pract 183:109170. https://doi.org/10.1016/j.diabres.2021.109170
Zhu J, Mu X, Zeng J, Xu C, Liu J, Zhang M, Li C, Chen J, Li T, Wang Y (2014) Ginsenoside Rg1 prevents cognitive impairment and hippocampus senescence in a rat model of D-galactose-induced aging. PLoS ONE 9(6):e101291. https://doi.org/10.1371/journal.pone.0101291
Zole E, Zadinane K, Pliss L, Ranka R (2018) Linkage between mitochondrial genome alterations, telomere length and aging population. Mitochondrial DNA A DNA Mapp Seq Anal 29(3):431–438. https://doi.org/10.1080/24701394.2017.1303490
Funding
This work was supported by Grants of the National Natural Science Foundation of China (grant no. 81671164) and Natural Science Foundation of Jiangsu Province (No. BK20211348).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Yue Jun, Peng Guo, Ming Li and Xiaotong Hu. The first draft of the manuscript was written by Yuexinzi Jin, Wan Wang and Xuewen Wei. Supervision and proofreading of this article were performed by Suhua Qi, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Laboratory Animal Ethics Committee of Xuzhou Medical University (ID: 202102W001, Date: February 01, 2021).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors have no relevant financial or nonfinancial interests to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yue, J., Guo, P., Jin, Y. et al. Momordica charantia polysaccharide ameliorates D-galactose-induced aging through the Nrf2/β-Catenin signaling pathway. Metab Brain Dis 38, 1067–1077 (2023). https://doi.org/10.1007/s11011-022-01103-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-01103-4