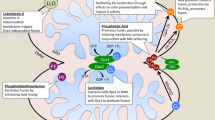
Abstract
Cardiac pathology including hypertrophy has been associated with an imbalance between mitochondrial fission and fusion. Generally, well-balanced mitochondrial fission and fusion are essential for proper functions of mitochondria. Leptin is a 16-kDa appetite-suppressing protein which has been shown to induce cardiomyocyte hypertrophy. In the present study, we determined whether leptin can influence mitochondrial fission or fusion and whether this can be related to its hypertrophic effect. Cardiomyocytes treated for 24 h with 3.1 nM leptin (50 ng/ml), a concentration representing plasma levels in obese individuals, demonstrated an increase in surface area and a significant 1.6-fold increase in the expression of the β-myosin heavy chain. Mitochondrial staining with MitoTracker Green dye showed elongated structures in control cells with an average length of 4.5 µm. Leptin produced a time-dependent increase in mitochondrial fragmentation with decreasing mitochondrial length. The hypertrophic response to leptin was also associated with increased protein levels of the mitochondrial fission protein dynamin-related protein1 (Drp1) although gene expression of Drp1 was unaffected possibly suggesting post-translational modifications of Drp1. Indeed, leptin treatment was associated with decreased levels of phosphorylated Drp1 and increased translocation of Drp1 to the mitochondria thereby demonstrating a pro-fission effect of leptin. As calcineurin may dephosphorylate Drp1, we determined the effect of a calcineurin inhibitor, FK506, which prevented leptin-induced hypertrophy as well as mitochondrial fission and mitochondrial dysfunction. In conclusion, our data show that leptin-induced cardiomyocyte hypertrophy is associated with enhanced mitochondrial fission via a calcineurin-mediated pathway. The ability of leptin to stimulate mitochondrial fission may be important in understanding the role of this protein in cardiac pathology especially that related to mitochondrial dysfunction.







Similar content being viewed by others
References
Cammisotto PG, Bukowiecki LJ, Deshaies Y, Bendayan M (2006) Leptin biosynthetic pathway in white adipocytes. Biochem Cell Biol 84:207–214
Xie L, O’Reilly CP, Chape SK, Mora S (2008) Adiponectin and leptin are secreted through distinct trafficking pathways in adipocytes. Biochim Biophys Acta 1782:99–108
Purdham DM, Zou MX, Rajapurohitam V, Karmazyn M (2004) Rat heart is a site of leptin production and action. Am J Physiol Heart Circ Physiol 287:H441–H446
Rajapurohitam V, Gan XT, Kirshenbaum LA, Karmazyn M (2003) The obesity-associated peptide leptin induces hypertrophy in neonatal rat ventricular myocytes. Circ Res 93:277–279
Considine RV, Sinha MK, Heiman ML, Kriauciunas A, Stephens TW, Nyce MR, Ohannesian JP, Marco CC, McKee LJ, Bauer TL, Caro JF (1996) Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N Engl J Med 334:292–295
Perego L, Pizzocri P, Corradi D, Maisano F, Paganelli M, Fiorina P, Barbieri M, Morabito A, Paolisso G, Folli F, Pontiroli AE (2005) Circulating leptin correlates with left ventricular mass in morbid (Grade III) obesity before and after weight loss induced by bariatric surgery: a potential role for leptin in mediating human left ventricular hypertrophy. J Clin Endocrinol Metab 90:4087–4093
Schulze PC, Kratzsch J, Linke A, Schoene N, Adams V, Gielen S, Erbs S, Moebius-Winkler S, Schuler G (2003) Elevated serum levels of leptin and soluble leptin receptor in patients with advanced chronic heart failure. Eur J Heart Fail 5:33–40
Toth MJ, Gottlieb SS, Fisher ML, Ryan AS, Nicklas BJ, Poehlman ET (1997) Plasma leptin concentrations and energy expenditure in heart failure patients. Metabolism 46:450–453
Fruhbeck G (2006) Intracellular signaling pathways activated by leptin. Biochem J 393:7–20
Yamashita T, Murakami T, Otani S, Kuwajima M, Shima K (1998) Leptin receptor signal transduction:OBRa and OBRb of fa type. Biochem Biophys Res Commun 246:752–759
Gan XT, Zhao G, Huang CX, Rowe AC, Purdham DM, Karmazyn M (2013) Identification of fat mass and obesity associated (FTO) protein expression in cardiomyocytes:regulation by leptin and its contribution to leptin-induced hypertrophy. PLoS ONE 8:e74235
Rajapurohitam V, Izaddoustdar F, Martinez-Abundis E, Karmazyn M (2012) Leptin-induced cardiomyocyte hypertrophy reveals both calcium-dependent and calcium-independent/RhoA-dependent calcineurin activation and NFAT nuclear translocation. Cell Signal 24:2283–2290
Xu FP, Chen MS, Wang YZ, Yi Q, Lin SB, Chen AF, Luo JD (2004) Leptin induces hypertrophy via endothelin-1-reactive oxygen species pathway in cultured neonatal rat cardiomyocytes. Circulation 110:1269–1275
Zeidan A, Javadov S, Chakrabarti S, Karmazyn M (2008) Leptin-induced cardiomyocyte hypertrophy involves selective caveolae and RhoA/ROCK-dependent p38 MAPK translocation to nuclei. Cardiovasc Res 77:64–72
Ashrafian H, Docherty L, Leo V, Towlson C, Neilan M, Steeples V, Lygate CA, Hough T, Townsend S, Williams D, Wells S, Norris D, Glyn-Jones S, Land J, Barbaric I, Lalanne Z, Denny P, Szumska D, Bhattacharya S, Griffin JL, Hargreaves I, Fernandez-Fuentes N, Cheeseman M, Watkins H, Dear TN (2010) A mutation in the mitochondrial fission gene Dnm1l leads to cardiomyopathy. PLoS Genet 6:e1001000
Watanabe T, Saotome M, Nobuhara M, Sakamoto A, Urushida T, Katoh H, Satoh H, Funaki M, Hayashi H (2014) Roles of mitochondrial fragmentation and reactive oxygen species in mitochondrial dysfunction and myocardial insulin resistance. Exp Cell Res 323:314–325
Dezfulian C, Shiva S, Alekseyenko A, Pendyal A, Beiser DG, Munasinghe JP, Anderson SA, Chesley CF, Hoek TLV, Gladwin MT (2009) Nitrite therapy after cardiac arrest reduces reactive oxygen species generation improves cardiac and neurological function and enhances survival via reversible inhibition of mitochondrial complex I. Circulation 120:897–905
Graham D, Huynh NN, Hamilton CA, Beattie E, Smith RA, Cocheme HM, Murphy MP, Dominiczak AF (2009) Mitochondria-targeted antioxidant MitoQ10 improves endothelial function and attenuates cardiac hypertrophy. Hypertension 54:322–328
Griffiths EJ, Halestrap AP (1993) Protection by cyclosporin A of ischemia/reperfusion-induced damage in isolated rat hearts. J Mol Cell Cardiol 25:1461–1469
Matsushima S, Ide T, Yamato M, Matsusaka H, Hattori R, Ikeuchi M, Kubota T, Sunagawa K, Hasegawa Y, Kurihara T, Oikawa S, Kinugawa S, Tsutsui H (2006) Overexpression of mitochondria peroxiredoxin-3 prevents left ventricular remodeling and failure after myocardial infarction in mice. Circulation 113:1779–1786
Javadov S, Karmazyn M (2007) Mitochondrial permeability transition pore opening as an endpoint to initiate cell death and as a putative target for cardioprotection. Cell Physiol Biochem 20:1–22
Detmer SA, Chan DC (2007) Functions and dysfunctions of mitochondrial dynamics. Nat Rev Mol Cell Biol 8:870–879
Twig G, Elorza A, Molina AJA, Mohamed H, Wikstrom JK, Walzer G, Stiles L, Haigh SE, Katz S, Las G, Alroy J, Wu M, Py BF, Yuan J, Deeney JT, Corkey BE, Shirihai OS (2008) Fission and selective fusion govern mitochondrial segregation and elimination by autophagy. Embo J 27:433–446
Jheng HF, Tsai PJ, Guo SM, Kuo LH, Chang CS, Su IJ, Chang CR, Tsai YS (2012) Mitochondrial fission contributes to mitochondrial dysfunction and insulin resistance in skeletal muscle. Mol Cell Biol 32:309–319
Pennanen C, Parra V, Lopez-Crisosto C, Morales PE, del Campo A, Gutierrez T, Rivera-Mejias P, Kuzmicic J, Chiong M, Zorzano A, Rothermel BA, Lavandero S (2014) Mitochondrial fission is required for cardiomyocyte hypertrophy mediated by a Ca2+-calcineurin signaling pathway. J Cell Sci 127:2659–2671
Molina AJA, Wikstrom JD, Stiles L, Las G, Mohamed H, Elorza A, Walzer G, Twig G, Katz S, Corkey BE, Shirihai OS (2009) Mitochondrial networking protects b-cells from nutrient-induced apoptosis. Diabetes 58:2303–2315
Sharp WW, Fang YH, Han M, Zhang HJ, Hong Z, Banathy A, Morrow E, Ryan JJ, Archer SL (2014) Dynamin-related protein (Drp1)-mediated diastolic dysfunction in myocardial ischemia-reperfusion injury: therapeutic benefits of Drp1 inhibition to reduce mitochondrial fission. FASEB J 28:316–326
Marsboom G, Toth PT, Ryan JJ, Hong Z, Wu X, Fang YH, Thenappan T, Piao L, Zhang HJ, Pogoriler J, Chen Y, Morrow E, Weir EK, Rehman J, Archer SL (2012) Dynamin-related protein 1-mediated mitochondrial mitotic fission permits hyperproliferation of vascular smooth muscle cells and offers a novel therapeutic target in pulmonary hypertension. Circ Res 110:1484–1497
Cereghetti GM, Stangherlin A, de Brito OM, Chang CR, Blakstone C, Bernardi P, Scorrano L (2008) Dephosphorylation by calcineurin regulates translocation of Drp1 to mitochondria. Proc Natl Acad Sci USA 105:15803–15808
Wai T, Garcia-Prieto J, Baker MJ, Merkwirth C, Benit P, Rustin P, Ruperez FJ, Barbas C, Ibanez B, Langer T (2015) Imbalanced OPA1 processing and mitochondrial fragmentation cause heart failure in mice. Science 350:aad0116
Song M, Mihara K, Chen Y, Scorrano L, Dorn GW (2015) Mitochondrial fission and fusion factors reciprocally orchestrate mitophagic culling in mouse hearts and cultured fibroblasts. Cell Metab 21:273–285
Martinez-Abundis E, Rajapurohitam V, Haist JV, Gan XT, Karmazyn M (2012) The obesity-related peptide leptin sensitizes cardiac mitochondria to calcium-induced permeability transition pore opening and apoptosis. PLoS ONE 7:e41612
Javadov S, Rajapurohitam V, Kilic A, Zeidan A, Choi A, Karmazyn M (2009) Anti-hypertrophic effect of NHE-1 inhibition involves GSK-3β-dependent attenuation of mitochondrial dysfunction. J Mol Cell Cardiol 46:998–1007
Mikusova A, Kralova E, Tylkova L, Novotova M, Stankovicoya T (2009) Myocardial remodeling induced by repeated low doses of isoproterenol. Can J Physiol Pharmacol 87:641–651
Banerjee P, Chander V, Bandyopadhyay A (2015) Balancing functions of annexin A6 maintain equilibrium between hypertrophy and apoptosis in cardiomyocytes. Cell Death Dis 6:e1873
Yin W, Li R, Feng X, Kang YJ (2018) The involvement of cytochrome c oxidase in mitochondrial fusion in primary cultures of neonatal cardiomyocytes. Cardiovasc Toxicol 8:365–373
De Windt LJ, Lim HW, Bueno OF, Liang Q, Delling U, Braz JC, Glascock BJ, Kimball TF, del Monte F, Hajjar RJ, Molkentin JD (2001) Targeted inhibition of calcineurin attenuates cardiac hypertrophy in vivo. Proc Natl Acad Sci USA 98:3322–3327
Zou Y, Hiroi Y, Uozumi H, Takimoto E, Toko H, Zhu W, Kudoh S, Mizukami M, Shimoyama M, Shibasaki F, Nagai R, Yazaki Y, Komuro I (2001) Calcineurin plays a clinical role in the development of pressure overload-induced cardiac hypertrophy. Circulation 104:97–101
Zeidan A, Purdham DM, Rajapurohitam V, Javadov S, Chakrabarti S, Karmazyn M (2005) Leptin induces vascular smooth cell hypertrophy through angiotensin II- and endothelin-1-dependent mechanisms and mediates stretch-induced hypertrophy. J Pharmacol Exp Therap 315:1075–1084
Lee MPS, Orlov D, Sweeney G (2005) Leptin induces rat glomerular mesangial cell hypertrophy, but does not regulate hyperplasia or apoptosis. Int J Obes 29:1395–1401
Hou N, Luo MS, Liu SM, Zhang HN, Xiao Q, Sun P, Zhang GS, Luo JD, Chen MS (2010) Leptin induces hypertrophy through activating the peroxisome proliferator-activated receptor α pathway in cultured neonatal rat cardiomyocytes. Clin Exp Pharmacol Physiol 37:1087–1095
Paolisso G, Tagliamonte MR, Galderisi M, Zito GA, D’Errico A, Marfella R, Carella C, de Divitiis O, Varricchio M (2001) Plasma leptin concentration, insulin sensitivity, and 24-hour ambulatory blood pressure and left ventricular geometry. Am J Hypertens 14:114–120
Romero-Corral A, Sierra-Johnson J, Lopez-Jimenez F, Thomas RJ, Singh P, Hoffmann M, Okcay A, Korinek J, Wolk R, Somes WK (2008) Relationships between leptin and C-reactive protein with cardiovascular disease in the adult general population. Nat Clin Pract Cardiovasc Med 5:418–425
Aizawa-Abe M, Ogawa Y, Masuzaki H, Ebihara K, Satoh N, Iwai H, Matsuoka N, Hayashi T, Hosoda K, Inoue G, Yoshimasa Y, Nakao K (2000) Pathophysiological role of leptin in obesity-related hypertension. J Clin Invest 105:1243–1252
Leyva F, Godsland IF, Ghatei M, Proudler AJ, Aldis S, Walton C, Bloom S, Stevenson JC (1998) Hyperleptinemia as a component of a metabolic syndrome of cardiovascular risk. Arterioscler Thromb Vasc Biol 18:928–933
Chen L, Gong Q, Stice JP, Knowlton AA (2009) Mitochondrial OPA1, apoptosis and heart failure. Cardiovasc Res 84:91–99
Alaimo A, Gorojoh RM, Beauquis J, Munoz MJ, Saravia F, Kotler ML (2014) Deregulation of mitochondria-shaping proteins Opa-1 and Drp-1 in manganese-induced apoptosis. PLos ONE 9:e91848
Longo DL (2013) Mitochondrial dynamics-mitochondrial fission and fusion in human diseases. N Engl J Med 369:2236–2251
Dorn GWII (2013) Mitochondrial dynamics in heart disease. Biochim Biophys Acta 1833:233–241
Baandrup U, Florio RA, Roters F, Olsen EGJ (1981) Electron microscopic investigation of endomyocardial biopsy samples in hypertrophy and cardiomyocytes: a semiquantitative study in 48 patients. Circulation 63:1289–1298
Harder Z, Zunino R, McBride H (2004) Sumo1 conjugates mitochondrial substrates and participates in mitochondrial fission. Curr Biol 14:340–345
Karbowski M, Neutzner A, Youle RJ (2007) The mitochondrial E3 ubiquitin ligase March5 is required for Drp1 dependent mitochondrial division. J Cell Biol 178:71–84
Nakamura T, Cieplak P, Cho DH, Godzik A, Lipton SA (2010) S-nitrosylation of Drp1 links excessive mitochondrial fission to neuronal injury in neurodegeneration. Mitochondrion 10:573–578
Manukyan I, Galatioto J, Mascareno E, Bhaduri S, Siddiqui MA (2010) Cross-talk between calcineurin/NFAT and Jak/STAT signalling induces cardioprotective αB-crystallin gene expression in response to hypertrophic stimuli. J Cell Mol Med 14:1707–1716
Javadov S, Rajapurohitam V, Kilić A, Hunter JC, Zeidan A, Said Faruq N, Escobales N, Karmazyn M (2011) Expression of mitochondrial fusion-fission proteins during post-infarction remodeling: the effect of NHE-1 inhibition. Basic Res Cardiol 106:99–109
Ventura-Clapier R, Garnier A, Veksler V (2008) Transcriptional control of mitochondrial biogenesis: the central role of PGC-1α. Cardiovasc Res 79:208–217
Rimbaud S, Garnier A, Ventura-Clapier R (2009) Mitochondrial biogenesis in cardiac pathophysiology. Pharmacol Rep 61:131–138
Pfluger PT, Kabra DG, Aichler M, Schriever SC, Pfuhlmann K, García VC, Lehti M, Weber J, Kutschke M, Rozman J, Elrod JW, Hevener AL, Feuchtinger A, Hrabě de Angelis M, Walch A, Rollmann SM, Aronow BJ, Müller TD, Perez-Tilve D, Jastroch M, De Luca M, Molkentin JD, Tschöp MH (2015) Calcineurin links mitochondrial elongation with energy metabolism. Cell Metab 22:838–850
Martinez-Abundis E, Rajapurohitam V, Gertler A, Karmazyn M (2015) Identification of functional leptin receptors expressed in ventricular mitochondria. Mol Cell Biochem 408:155–162
Acknowledgements
This work was supported by a Grant from the Canadian Institutes of Health Research (MOP 62764). M Karmazyn held a Tier 1 Canada Research Chair in Experimental Cardiology (2004–2016).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jong, C.J., Yeung, J., Tseung, E. et al. Leptin-induced cardiomyocyte hypertrophy is associated with enhanced mitochondrial fission. Mol Cell Biochem 454, 33–44 (2019). https://doi.org/10.1007/s11010-018-3450-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-018-3450-5