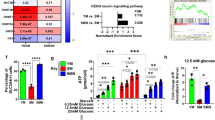
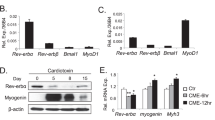
Abstract
Skeletal muscle tissue has a remarkable high regenerative capacity. The underlying cellular events are governed by complex signaling processes, and the proliferation of skeletal myoblasts is a key initial event. The role of nitric oxide (NO) in cell cycle regulation is well-appreciated. Nitrite, an NO oxidation product, is a stable source for NO-like bioactivity particularly in cases when oxygen shortage compromises NO-synthases activity. Although numerous studies suggest that nitrite effects are largely related to NO-dependent signaling, emerging evidence also implicates that nitrite itself can activate protein pathways albeit under physiological, normoxic conditions. This includes a recently demonstrated cyclic guanosine monophosphate-(cGMP)-independent enhancement of endothelial cell proliferation. Whether nitrite itself has the potential to affect myoblast proliferation and metabolism with or without activation of the canonical NO/cGMP pathway to subsequently support muscle cell regeneration is not known. Here we show that nitrite increases proliferation and metabolic activity of murine cultured myoblasts dose-dependently. This effect is not abolished by the NO scavenger 2-(4-carboxy-phenyl)-4,4,5,5-tetramethylimida-zoline-1-oxyl-3 oxide and does not affect intracellular cGMP levels, implicating a cGMP-independent mechanism. Nitrite circumvents the rapamycin induced attenuation of myoblast proliferation and enhances mTOR activity. Our results provide evidence for a novel potential physiological and therapeutic approach of nitrite in skeletal muscle regeneration processes under normoxia independent of NO and cGMP.





Similar content being viewed by others
References
Le Grand F, Rudnicki MA (2007) Skeletal muscle satellite cells and adult myogenesis. Curr Opin Cell Biol 19:628–633
Dhawan J, Rando TA (2005) Stem cells in postnatal myogenesis: molecular mechanisms of satellite cell quiescence, activation and replenishment. Trends Cell Biol 15:666–673
Mauro A (1961) Satellite cell of skeletal muscle fibers. J Biophys Biochem Cytol 9:493–495
Zammit PS, Relaix F, Nagata Y et al (2006) Pax7 and myogenic progression in skeletal muscle satellite cells. J Cell Sci 119:1824–1832
Charge SB, Rudnicki MA (2004) Cellular and molecular regulation of muscle regeneration. Physiol Rev 84:209–238
Schultz E, Jaryszak DL (1985) Effects of skeletal muscle regeneration on the proliferation potential of satellite cells. Mech Ageing Dev 30:63–72
Moss FP, Leblond CP (1971) Satellite cells as the source of nuclei in muscles of growing rats. Anat Rec 170:421–435
Germani A, Di Carlo A, Mangoni A et al (2003) Vascular endothelial growth factor modulates skeletal myoblast function. Am J Pathol 163:1417–1428
Tatsumi R, Hattori A, Ikeuchi Y et al (2002) Release of hepatocyte growth factor from mechanically stretched skeletal muscle satellite cells and role of pH and nitric oxide. Mol Biol Cell 13:2909–2918
Chakravarthy MV, Booth FW, Spangenburg EE (2001) The molecular responses of skeletal muscle satellite cells to continuous expression of IGF-1: implications for the rescue of induced muscular atrophy in aged rats. Int J Sport Nutr Exerc Metab 11:S44–S48
Zalin RJ (1987) The role of hormones and prostanoids in the in vitro proliferation and differentiation of human myoblasts. Exp Cell Res 172:265–281
Ohanna M, Sobering AK, Lapointe T et al (2005) Atrophy of S6K1(−/−) skeletal muscle cells reveals distinct mTOR effectors for cell cycle and size control. Nat Cell Biol 7:286–294
Halevy O, Cantley LC (2004) Differential regulation of the phosphoinositide 3-kinase and MAP kinase pathways by hepatocyte growth factor vs. insulin-like growth factor-I in myogenic cells. Exp Cell Res 297:224–234
Jones NC, Fedorov YV, Rosenthal RS et al (2001) ERK1/2 is required for myoblast proliferation but is dispensable for muscle gene expression and cell fusion. J Cell Physiol 186:104–115
Conejo R, Lorenzo M (2001) Insulin signaling leading to proliferation, survival, and membrane ruffling in C2C12 myoblasts. J Cell Physiol 187:96–108
Bennett AM, Tonks NK (1997) Regulation of distinct stages of skeletal muscle differentiation by mitogen-activated protein kinases. Science 278:1288–1291
Sarbassov DD, Ali SM, Kim DH et al (2004) Rictor, a novel binding partner of mTOR, defines a rapamycin-insensitive and raptor-independent pathway that regulates the cytoskeleton. Curr Biol 14:1296–1302
Hara K, Maruki Y, Long X et al (2002) Raptor, a binding partner of target of rapamycin (TOR), mediates TOR action. Cell 110:177–189
Sengupta S, Peterson TR, Sabatini DM (2010) Regulation of the mTOR complex 1 pathway by nutrients, growth factors, and stress. Mol Cell 40:310–322
Laplante M, Sabatini DM (2009) mTOR signaling at a glance. J Cell Sci 122:3589–3594
Madhusoodanan KS, Murad F (2007) NO-cGMP signaling and regenerative medicine involving stem cells. Neurochem Res 32:681–694
Lincoln TM, Wu X, Sellak H et al (2006) Regulation of vascular smooth muscle cell phenotype by cyclic GMP and cyclic GMP-dependent protein kinase. Front Biosci 11:356–367
Pilz RB, Broderick KE (2005) Role of cyclic GMP in gene regulation. Front Biosci 10:1239–1268
Soltow QA, Lira VA, Betters JL et al (2010) Nitric oxide regulates stretch-induced proliferation in C2C12 myoblasts. J Muscle Res Cell Motil 31:215–225
Wang L, Frizzell SA, Zhao X et al (2012) Normoxic cyclic GMP-independent oxidative signaling by nitrite enhances airway epithelial cell proliferation and wound healing. Nitric Oxide 26:203–210
Rammos C, Hendgen-Cotta UB, Sobierajski J et al (2014) Dietary nitrate reverses vascular dysfunction in old adults with moderately increased cardiovascular risk. J Am Coll Cardiol 63:1584–1585
Heiss C, Meyer C, Totzeck M et al (2012) Dietary inorganic nitrate mobilizes circulating angiogenic cells. Free Radic Biol Med 52:1767–1772
Lundberg JO, Gladwin MT, Ahluwalia A et al (2009) Nitrate and nitrite in biology, nutrition and therapeutics. Nat Chem Biol 5:865–869
Hendgen-Cotta U, Grau M, Rassaf T et al (2008) Reductive gas-phase chemiluminescence and flow injection analysis for measurement of the nitric oxide pool in biological matrices. Methods Enzymol 441:295–315
Rassaf T, Totzeck M, Hendgen-Cotta UB et al (2014) Circulating nitrite contributes to cardioprotection by remote ischemic preconditioning. Circ Res 114:1601–1610
Totzeck M, Hendgen-Cotta UB, Luedike P et al (2012) Nitrite regulates hypoxic vasodilation via myoglobin-dependent nitric oxide generation. Circulation 126:325–334
Hendgen-Cotta UB, Flogel U, Kelm M et al (2010) Unmasking the Janus face of myoglobin in health and disease. J Exp Biol 213:2734–2740
Hendgen-Cotta UB, Kelm M, Rassaf T (2010) A highlight of myoglobin diversity: the nitrite reductase activity during myocardial ischemia-reperfusion. Nitric Oxide 22:75–82
Hendgen-Cotta UB, Merx MW, Shiva S et al (2008) Nitrite reductase activity of myoglobin regulates respiration and cellular viability in myocardial ischemia-reperfusion injury. Proc Natl Acad Sci USA 105:10256–10261
Rassaf T, Flogel U, Drexhage C et al (2007) Nitrite reductase function of deoxymyoglobin: oxygen sensor and regulator of cardiac energetics and function. Circ Res 100:1749–1754
Totzeck M, Hendgen-Cotta UB, Kelm M et al (2014) Crosstalk between nitrite, myoglobin and reactive oxygen species to regulate vasodilation under hypoxia. PLoS One 9:e105951
Totzeck M, Hendgen-Cotta UB, Rammos C et al (2012) Assessment of the functional diversity of human myoglobin. Nitric Oxide 26:211–216
Hendgen-Cotta UB, Luedike P, Totzeck M et al (2012) Dietary nitrate supplementation improves revascularization in chronic ischemia. Circulation 126:1983–1992
Repetto G, del Peso A, Zurita JL (2008) Neutral red uptake assay for the estimation of cell viability/cytotoxicity. Nat Protoc 3:1125–1131
Leonhardt H, Rahn HP, Weinzierl P et al (2000) Dynamics of DNA replication factories in living cells. J Cell Biol 149:271–280
Ishiyama M, Miyazono Y, Sasamoto K et al (1997) A highly water-soluble disulfonated tetrazolium salt as a chromogenic indicator for NADH as well as cell viability. Talanta 44:1299–1305
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Ulibarri JA, Mozdziak PE, Schultz E et al (1999) Nitric oxide donors, sodium nitroprusside and S-nitroso-N-acetylpencillamine, stimulate myoblast proliferation in vitro. In Vitro Cell Dev Biol Anim 35:215–218
Rassaf T, Bryan NS, Maloney RE et al (2003) NO adducts in mammalian red blood cells: too much or too little? Nat Med 9:481–482
Rassaf T, Preik M, Kleinbongard P et al (2002) Evidence for in vivo transport of bioactive nitric oxide in human plasma. J Clin Investig 109:1241–1248
Rassaf T, Kleinbongard P, Preik M et al (2002) Plasma nitrosothiols contribute to the systemic vasodilator effects of intravenously applied NO: experimental and clinical study on the fate of NO in human blood. Circ Res 91:470–477
Stamler JS, Simon DI, Jaraki O et al (1992) S-nitrosylation of tissue-type plasminogen activator confers vasodilatory and antiplatelet properties on the enzyme. Proc Natl Acad Sci USA 89:8087–8091
Weerapana E, Wang C, Simon GM et al (2010) Quantitative reactivity profiling predicts functional cysteines in proteomes. Nature 468:790–795
Sarbassov DD, Ali SM, Sengupta S et al (2006) Prolonged rapamycin treatment inhibits mTORC2 assembly and Akt/PKB. Mol Cell 22:159–168
Luedike P, Hendgen-Cotta UB, Sobierajski J et al (2012) Cardioprotection through S-nitros(yl)ation of macrophage migration inhibitory factor. Circulation 125:1880–1889
Hamid SA, Totzeck M, Drexhage C et al (2010) Nitric oxide/cGMP signalling mediates the cardioprotective action of adrenomedullin in reperfused myocardium. Basic Res Cardiol 105:257–266
Ferri N (2012) AMP-activated protein kinase and the control of smooth muscle cell hyperproliferation in vascular disease. Vasc Pharmacol 56:9–13
Acknowledgments
MT was supported by a scholarship from the German Heart Foundation (Deutsche Herzstiftung) and by a Grant from the Forschungskommission of the Medical Faculty of the University of Duesseldorf. AS is a scholar of the Studienstiftung des Deutschen Volkes. TR is a Heisenberg professor funded by the DFG (RA969/7-2). TR was supported by a grant from the DFG (RA969/4-2).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Totzeck, M., Schicho, A., Stock, P. et al. Nitrite circumvents canonical cGMP signaling to enhance proliferation of myocyte precursor cells. Mol Cell Biochem 401, 175–183 (2015). https://doi.org/10.1007/s11010-014-2305-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-014-2305-y