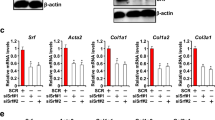
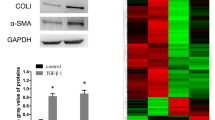
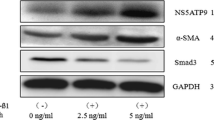
Abstract
Smad1 is an important signaling molecule for members of transforming growth factor-β (TGF-β) superfamily. Increased expression of Smad1 in activated hepatic stellate cells (HSCs) indicates a role of Smad1 in liver fibrosis. Therefore, understanding of Smad1 gene expression could be important to control the activation of HSCs. Current study reports the cloning and characterizing rat Smad1 5′-flanking region in liver cells. Rat Smad1 5′-flanking region was cloned by PCR method. Promoter deletional analysis and electrophoretic mobility shift assay (EMSA) were examined in hepatocyte and HSCs cell line (CFSC-8B cells), respectively. Results indicated that rat Smad1 used GC-box as its promoter and there was a transcriptional regulatory element located at the region of −163 to −56bp. EMSA demonstrated two bands on Smad1 promoter region. Smad1 promoter activity was higher in CFSC-8B cells cultured on uncoated plastic dish than that of CFSC-8B cells cultured on Matrigel-coated plastic dish. In conclusion, rat Smad1 promoter was cloned and characterized in hepatocyte and HSC cell line (CFSC-8B cells) at different culture conditions.





Similar content being viewed by others
References
Massague J (1998) TGF-beta signal transduction. Annu Rev Biochem 67:753–791
Itoh S, Itoh F, Goumans MJ, Ten Dijke P (2000) Signaling of transforming growth factor-beta family members through Smad proteins. Eur J Biochem 267:6954–6967
Liu F, Hata A, Baker JC, Doody J, Carcamo J, Harland RM, Massague J (1996) A human Mad protein acting as a BMP-regulated transcriptional activator. Nature 381:620–623
Fan J, Shen H, Sun Y, Li P, Burczynski F, Namaka M, Gong Y (2006) Bone morphogenetic protein 4 mediates bile duct ligation induced liver fibrosis through activation of Smad1 and ERK1/2 in rat hepatic stellate cells. J Cell Physiol 207:499–505
Huang S, Flanders KC, Roberts AB (2000) Characterization of the mouse Smad1 gene and its expression pattern in adult mouse tissues. Gene 258:43–53
Shen H, Huang G, Hadi M, Choy P, Zhang M, Minuk GY, Chen Y, Gong Y (2003) Transforming growth factor-beta1 downregulation of Smad1 gene expression in rat hepatic stellate cells. Am J Physiol Gastrointest Liver Physiol 285:G539–G546
Friedman SL, Roll FJ, Boyles J, Bissell DM (1985) Hepatic lipocytes: the principal collagen-producing cells of normal rat liver. Proc Natl Acad Sci USA 82:8681–8685
Hendriks HF, Blaner WS, Wennekers HM, Piantedosi R, Brouwer A, de Leeuw AM, Goodman DS, Knook DL (1988) Distributions of retinoids, retinoid-binding proteins and related parameters in different types of liver cells isolated from young and old rats. Eur J Biochem 171:237–244
Wu J, Zern MA (2000) Hepatic stellate cells: a target for the treatment of liver fibrosis. J Gastroenterol 35:665–672
Casini A, Pinzani M, Milani S, Grappone C, Galli G, Jezequel AM, Schuppan D, Rotella CM, Surrenti C (1993) Regulation of extracellular matrix synthesis by transforming growth factor beta 1 in human fat-storing cells. Gastroenterology 105:245–253
Albanis E, Friedman SL (2001) Hepatic fibrosis. Pathogenesis and principles of therapy. Clin Liver Dis 5:315–334
Friedman SL (2003) Liver fibrosis—from bench to bedside. J Hepatol 38(Suppl1):S38–S53
Mann DA, Smart DE (2002) Transcriptional regulation of hepatic stellate cell activation. Gut 50:891–896
Lum A, Le Marchand L (1998) A simple mouthwash method for obtaining genomic DNA in molecular epidemiological studies. Cancer Epidemiol Biomarkers Prev 7:719–724
Greenwel P, Rubin J, Schwartz M, Hertzberg EL, Rojkind M (1993) Liver fat-storing cell clones obtained from a CCl4-cirrhotic rat are heterogeneous with regard to proliferation, expression of extracellular matrix components, interleukin-6, and connexin 43. Lab Invest 69:210–216
Moustakas A, Souchelnytskyi S, Heldin CH (2001) Smad regulation in TGF-beta signal transduction. J Cell Sci 114:4359–4369
Benoist C, Chambon P (1981) In vivo sequence requirements of the SV40 early promotor region. Nature 290:304–310
Kadonaga JT, Carner KR, Masiarz FR, Tjian R (1987) Isolation of cDNA encoding transcription factor Sp1 and functional analysis of the DNA binding domain. Cell 51:1079–1090
Tamura T, Mikoshiba K (1991) Role of a GC-rich motif in transcription regulation of the adenovirus type 2 IVa2 promoter which lacks typical TATA-box element. FEBS Lett 282:87–90
Chow CW, Clark M, Rinaldo J, Chalkley R (1994) Identification of the rat xanthine dehydrogenase/oxidase promoter. Nucleic Acids Res 22:1846–1854
Denney RM, Sharma A, Dave SK, Waguespack A (1994) A new look at the promoter of the human monoamine oxidase A gene: mapping transcription initiation sites and capacity to drive luciferase expression. J Neurochem 63:843–856
Acknowledgement
This work was supported by the grant (MOP-62923) from Canadian Institute of Health Research to Dr. Yuewen Gong.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, Y., Fan, J., Shen, H. et al. Cloning and promoter activity of rat Smad1 5′-flanking region in rat hepatic stellate cells. Mol Cell Biochem 304, 227–234 (2007). https://doi.org/10.1007/s11010-007-9504-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-007-9504-8