Abstract
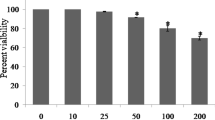
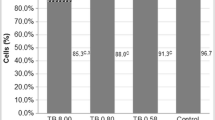
The role of oxidative stress in immune cell toxicity caused by the pesticides lindane, malathion and permethrin was investigated in thymic cells from C57BL/6 mice. Thymocytes treated with any of these pesticides (concentrations ranging between 50–150 μM) were found to generate both superoxide (•O2 −) and H2O2. The production of •O2 − was detected with hydroethidine-ethidium bromide assay. H2O2 production was monitored with a flow cytometric fluorescent (DCFH-DA) assay. All three pesticides stimulated •O2 − release after 5 min exposure. Lindane and permethrin, but not malathion, continued to have significant (p ≤ 0.05) effects on •O2 − generation following 15 min of exposure. The lindane + malathion mixture was found to cause more-than-additive increase in •O2 − production compared to individual pesticide treatments (at both 5 and 15 min). However, the effect of the lindane + permethrin mixture was not significantly different than individual components of this mixture. The effects of these pesticides on levels of antioxidant enzymes were also investigated, and only mixtures were found to have significant (p ≤ 0.05) effects. Thus, lindane + malathion and lindane + permethrin mixtures increased total superoxide dismutase (SOD) specific activity, had no effect on catalase levels and inhibited GSH-peroxidase and GSH-reductase specific activities. Although the results of these studies do not explain the mechanism of action of these pesticides on the generation of •O2 − and H2O2, it is worthy of note that mixtures of these chemicals have oxidative responses greater than those of single chemicals.
Similar content being viewed by others
References
Olgun S, Gogal Jr RM, Adeshina F, Choudhury H, Misra HP: Pesticide mixtures potentiate the toxicity in murine thymocytes. Toxicology 196(3): 181–195, 2004
Banerjee BD, Seth V, Bhattacharya A, Pasha ST, Chakraborty AK: Biochemical effects of some pesticides on lipid peroxidation and free radical scavengers. Toxicol Lett 107(1–3): 33–47, 1999
Schweich MD, Lison D, Lauwerys R: Assesment of lipid peroxidation associated with lung damage induced by oxidative stress: In vivo and in vitro study. Biochem Pharmacol 47(8): 1395–1400, 1994
Videla LA, Barros SBM, Junqueira VBC: Lindane induced liver oxidative stress. Free Radic Biol Med 9(2): 169–179, 1990
Marks DB, Marks AD Smith CM (eds): Oxygen metabolism and oxygen toxicity. In: Basic Medical Biochemistry, Wiliams and Wilkins, MD, pp. 327–340, 1996
Banerjee BD, Seth V, Ahmed RS: Pesticide-induced oxidative stress: Perspectives and Trends. Rev Env Health 16(1): 1–40, 2001
Bachowski S, Xu Y, Stevenson DE, Walborg EF Jr, Klaunig JE: Role of oxidative stress in the selective toxicity of dieldrin in the mouse liver. Toxicol Appl Pharmacol 150(2): 301–309, 1998
Hsu CH, Quistad GB, Casida JE: Phosphine-induced oxidative stress in Hepa 1c1c7 cells. Toxicol Sci 46(1): 204–210, 1998
Gauthier M, Roberge CJ, Pelletier M, Tessier PA, Girard D: Activation of human neutrophils by technical toxaphene. Clin Immunol 98(1): 46–53, 2001
Bagchi D, Balmoori J, Bagchi M, Ye X, Williams CB, Stohs SJ: Role of p53 tumor suppressor gene in the toxicity of TCDD, endrin, naphthalene, and chromium (VI) in liver and brain tissues of mice. Free Radic Biol Med 28(6): 895–903, 2000
El-Gohary M, Awara WM, Nassar S, Hawas S: Deltamethrin-induced testicular apoptosis in rats: the protective effect of nitric oxide synthase inhibitor. Toxicology 132(1): 1–8, 1999
Kale M, Rathore N, John S, Bhatnagar D: Lipid peroxidative damage on pyrethroid exposure and alterations in antioxidant status in rat erythrocytes: a possible involvement of reactive oxygen species. Toxicol Lett 105(3): 197–205, 1999
Abdollahi M, Ranjbar A, Shadnia S, Nikfar S, Rezaiee A: Pesticides and oxidative stress: a review. Med Sci Monit 10(6): 141–147, 2004
Sharma RP, Reddy RV: Toxic effects of chemicals on the immune system. In: TJ Haley and WO Berndt (eds). Handbook of Toxicology, Hemisphere Publishing Corp, Philadelphia, pp. 555–591, 1997
Gleichmann E, Kimber I, Purchase IFH: Immunotoxicology: suppressive and stimulatory effects of drugs and environmental chemicals on the immune system. Arch Toxicol 63(4): 257–273, 1989
Repetto R, Baliga SS: Pesticides and the immune system: the public health risks. Executive summary. Cent Eur J Public Health 4(4): 263–265, 1996
Junqueira VB, Koch OR, Arisi AC, Fuzaro AP, Azzalis LA, Barros SB, Cravero A, Farre S, Videla LA: Regression of morphological alterations and oxidative stress-related parameters after acute lindane-induced hepatotoxicity in rats. Toxicology 117(2–3): 199–205, 1997
Barros SB, Simizu K, Junqueira VB: Liver lipid peroxidation-related parameters after short-term administration of hexachlorocyclohexane isomers to rats. Toxicol Lett 56(1–2): 137–144, 1991
Raszyk J, Toman M, Gajduskova V, Nezveda K, Ulrich R, Jarosova A, Docekalova H, Salava J, Palac J: Effects of environmental pollutants on the porcine and bovine immune systems. Vet Med (Praha) 42 (11): 313–317, 1997
Meera P, Rao PR, Shanker R, Tripathi O: Immunomodulatory effects of γ-HCH (lindane) in mice. Immunopharmacol Immunotoxicol 14(1–2): 261–282, 1992
Rose RL, Hodgson E, Roe RM: Pesticides. In: H Marquardt, SG Schafer, RO McClellan and F Welsch (eds). Toxicology, Academic Press, San Diego, CA, Chapter 17, pp. 663–698, 1999
Johnson VJ, Rosenberg AM, Lee K, Blakley BR: Increased T-lymphocyte dependent antibody production in female SJL/J mice following exposure to commercial grade malathion. Toxicology 170(1–2): 119–129, 2002
Beaman JR, Finch R, Gardner H, Hoffmann F, Rosencrance A, Zelikoff JT: Mammalian immunoassays for predicting the toxicity of malathion in a laboratory fish model. J Toxicol Environ Health A 56(8): 523–542, 1999
Rodgers K, Xiong S: Effect of acute administration of malathion by oral and dermal routes on serum histamine levels. Int J Immunopharmacol 19(8): 437–441 (1997)
Blaylock BL, Abdel-Nasser M, McCarty SM, Knesel JA, Tolson KM, Ferguson PW, Mehendale HM: Suppression of cellular immune responses in BALB/c mice following oral exposure to permethrin. Bull Environ Contam Toxicol 54(5): 768–774, 1995
Kitazawa M, Anantharam V, Kanthasamy AG: Dieldrin-induced oxidative stress and neurochemical changes contribute to apoptopic cell death in dopaminergic cells. Free Radic Biol Med 31(11): 1473–1485, 2001
Wang H, Joseph JA: Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic Biol Med 27(5–6): 612–616, 1999
Royall JA, Ischiropoulos H: Evaluation of 2′,7′-dichlorofluorescin and dihydrorhodamine 123 as fluorescent probes for intracellular H2O2 in cultured endothelial cells. Arch Biochem Biophys 302(2): 348–356, 1993
Misra HP: Adrenochrome Assay for Superoxide Dismutase. In: RA Greenwald (ed). CRC Handbook of Methods for Oxygen Radical Research, CRC Press, FL. pp. 237–241, 1985
Misra HP, Fridovich I: The Role of Superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem, 247(10): 3170–3175, 1972
Beers RF, Sizer IW: A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. J Biol Chem 195(1): 133–140, 1952
Worthington Enzyme Manual: Catalase (Beef Liver). Worthington Biochemical Corporation, Freehold, NJ, 1972
Paglia DE, Valentine WN: Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Meth 70(1): 158–169, 1967
Tappel AL: Glutathione peroxidase and hydroperoxides. Methods Enzymol 52: 506–513, 1978
Carlberg I, Mannervik B: Glutathione reductase. Methods Enzymol 113: 484–490, 1985
Hassoun EA, Stohs SJ: TCDD, endrin and lindane induced oxidative stress in fetal and placental tissues of C57BL/6J and DBA/2J mice. Comp Biochem Physiol C Pharmacol Toxicol Endocrinol 115(1): 11–18, 1996
Junqueira VB, Simizu K, Van Halsema L, Koch OR, Barros SB, Videla LA: Lindane-induced oxidative stress. I. Time course of changes in hepatic microsomal parameters, antioxidant enzymes, lipid peroxidative indices and morphological characteristics. Xenobiotica 18(11): 1297–1304, 1988
Bagchi M, Stohs SJ: In vitro induction of reactive oxygen species by 2,3,7,8-tetrachloro-dibenzo-p-dioxin, endrin and lindane in rat peritoneal macrophages and hepatic mitochondria and microsomes. Free Radical Biol Med 14(1): 11–18, 1993
Keaney JF Jr, Vita JA: Atherosclerosis, oxidative stress, and antioxidant protection in endothelium-derived relaxing factor action. Prog Cardiovasc Dis 38(2): 129–154, 1995
Albers DS, Beal MF: Mitochondrial dysfunction and oxidative stress in aging and neurodegenerative disease. J Neural Transm Suppl, 59:133–154, 2000
Junqueira VBC, Simizu K, Videla LA, Barros SBM: Dose-dependent study of the effect of acute lindane administration on rat liver superoxide anion production, antioxidant enzyme activities and lipid peroxidation. Toxicology 41(2): 193–204, 1986
John S, Kale M, Rathore N, Bhatnagar D: Protective effect of vitamin E in dimethoate and malathion induced oxidative stress in rat erythrocytes. J Nutr Biochem 12(9): 500–504, 2001
Ahmed RS, Seth V, Pasha ST, Banerjee BD: Influence of dietary ginger (Zingiber officinales Rosc) on oxidative stress induced by malathion in rats. Food Chem Toxicol 38(5): 443–450, 2000
Kumar A, Dwivedi PP: Relative induction of molecular forms of cytochrome P-450 in gamma-hexachlorocyclohexane exposed rat liver microsomes. Arch Toxicol 62(6): 479–481, 1988
Pazirandeh A, Xue Y, Rafter I, Sjovall J, Jondal M, Okret S: Paracrine glucocorticoid activity produced by mouse thymic epithelial cells. FASEB J 13(8): 893–901, 1999
von Moltke LL, Greenblatt DJ, Schmider J, Wright CE, Harmatz JS, Shader RI: In vitro approaches to predicting drug interactions in vivo. Biochem Pharmacol 55(2): 113–122, 1998
Kremers P: In vitro tests for predicting drug-drug interactions: the need for validated procedures. Pharmacol Toxicol 91(5): 209–217, 2002
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s11010-006-9311-7
Rights and permissions
About this article
Cite this article
Olgun, S., Misra, H.P. Pesticides induced oxidative stress in thymocytes. Mol Cell Biochem 290, 137–144 (2006). https://doi.org/10.1007/s11010-006-9178-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-006-9178-7