Abstract
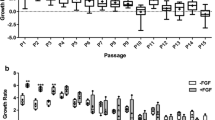
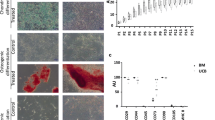
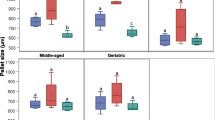
Stem cell therapy is a new approach in veterinary medicine for the treatment of complicated disorders such as articular cartilage injuries in horses. Although the equine bone marrow-derived mesenchymal stem cells (eBMSCs) have a unique capacity in chondrogenic differentiation, it needs to be optimized. In the current study, we evaluated the enhancement of chondrogenic differentiation in eBMSCs using transforming growth factor beta-3 (TGF-β3) and bone morphogenic protein 6 (BMP-6) in vitro. The characterized eBMSCs at passage 3 (P3) were cultivated as micro-pellets for 21 days under different conditions: (1) basic culture medium (BCM), (2) chondrogenic differentiation medium (CDM), (3) CDM with BMP-6, (4) CDM with TGF-β3, and (5) CDM with BMP-6 and TGF-β3 to evaluate the chondrogenic differentiation by assessment of pellet size, histological characteristics, and gene expression profile. Pellet size in groups that contained growth factors had a significant difference in comparison with the control group or chondrogenic group without growth factors. The histological assessment showed that the cartilage progression score in groups containing TGF-β3, alone or combined with BMP-6, was significantly higher in comparison with that of the other groups (P < 0.05). Additionally, the aggrecan gene was expressed in all groups except the control group, while collagen type II was just expressed in groups containing TGF-β3, alone or combined with BMP-6. TGF-β3 may have more promising effects on the enhancement of chondrogenic differentiation of eBMSCs in comparison with BMP-6. However, the combination of TGF-β3 and BMP-6 promoted chondrogenic differentiation of eBMSCs in a synergistic manner.




Similar content being viewed by others
References
Afizah H, Yang Z, Hui JHP, Ouyang H-W, Lee E-H (2007) A comparison between the chondrogenic potential of human bone marrow stem cells (BMSCs) and adipose-derived stem cells (ADSCs) taken from the same donors. Tissue Eng 13:659–666. https://doi.org/10.1089/ten.2006.0118
Bosnakovski D et al (2004) Chondrogenic differentiation of bovine bone marrow mesenchymal stem cells in pellet cultural system. Exp Hematol 32:502–509
De Schauwer C, Meyer E, Van de Walle GR, Van Soom A (2011) Markers of stemness in equine mesenchymal stem cells: a plea for uniformity. Theriogenology 75:1431–1443
Del Bue M, Riccò S, Ramoni R, Conti V, Gnudi G, Grolli S (2008) Equine adipose-tissue derived mesenchymal stem cells and platelet concentrates: their association in vitro and in vivo. Vet Res Commun 32(Suppl 1):S51–S55. https://doi.org/10.1007/s11259-008-9093-3
Docheva D, Haasters F, Schieker M (2008) Mesenchymal stem cells and their cell surface receptors. Curr Rheumatol Rev 4:155–160
Dominici M et al (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. Int Soc Cell Ther Pos statement Cytother 8:315–317
Fan J, Varshney RR, Ren L, Cai D, Wang D-A (2009) Synovium-derived mesenchymal stem cells: a new cell source for musculoskeletal regeneration. Tissue Eng B 15:75–86
Fortier L, Nixon AJ, Williams J, Cable C (1998) Isolation and chondrocytic differentiation of equine bone marrow-derived mesenchymal stem cells. Am J Vet Res 59:1182–1187
Friedenstein A, Chailakhjan R, Lalykina K (1970) The development of fibroblast colonies in monolayer cultures of guinea-pig bone marrow and spleen cells. Cell Prolif 3:393–403
Giovannini S, Brehm W, Mainil-Varlet P, Nesic D (2008) Multilineage differentiation potential of equine blood-derived fibroblast-like cells. Differentiation 76:118–129
Hegewald AA et al (2004) Hyaluronic acid and autologous synovial fluid induce chondrogenic differentiation of equine mesenchymal stem cells: a preliminary study. Tissue Cell 36:431–438. https://doi.org/10.1016/j.tice.2004.07.003
Hennig T, Lorenz H, Thiel A, Goetzke K, Dickhut A, Geiger F, Richter W (2007) Reduced chondrogenic potential of adipose tissue derived stromal cells correlates with an altered TGFβ receptor and BMP profile and is overcome by BMP-6. J Cell Physiol 211:682–691
Huang JI, Kazmi N, Durbhakula MM, Hering TM, Yoo JU, Johnstone BJJoOR (2005) Chondrogenic potential of progenitor cells derived from human bone marrow and adipose tissue: a patient-matched comparison. J Orthop Res 23:1383–1389
Im G II, Shin Y-W, Lee K-B (2005) Do adipose tissue-derived mesenchymal stem cells have the same osteogenic and chondrogenic potential as bone marrow-derived cells? Osteoarthritis Cartil 13:845–853. https://doi.org/10.1016/j.joca.2005.05.005
Indrawattana N et al (2004) Growth factor combination for chondrogenic induction from human mesenchymal stem cell. Biochem Biophys Res Commun 320:914–919
Jiang Y, Vaessen B, Lenvik T, Blackstad M, Reyes M, Verfaillie CM (2002) Multipotent progenitor cells can be isolated from postnatal murine bone marrow, muscle, and brain. Exp Hematol 30:896–904
Kern S, Eichler H, Stoeve J, Kluter H, Bieback K (2006) Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood or adipose tissue. Stem Cells 24:1294–1301
Kinner B, Zaleskas JM, Spector M (2002) Regulation of smooth muscle actin expression and contraction in adult human mesenchymal stem cells. Exp Cell Res 278:72–83
Koch TG, Berg LC, Betts DH (2009) Current and future regenerative medicine—principles, concepts, and therapeutic use of stem cell therapy and tissue engineering in equine medicine. Can Vet J 50:155–165
Kolf CM, Cho E, Tuan RS (2007) Mesenchymal stromal cells: biology of adult mesenchymal stem cells: regulation of niche, self-renewal and differentiation. Arthritis Res Ther 9(1):204
Malda J, Martens DE, Tramper J, van Blitterswijk CA, Riesle J (2003) Cartilage tissue engineering: controversy in the effect of oxygen. Crit Rev Biotechnol 23:175–194
Mescher AL (2010) Junqueira’s basic histology: Text & atlas. Mcgraw-Hill medical, New York
Murphy JM, Fink DJ, Hunziker EB, Barry FP (2003) Stem cell therapy in a caprine model of osteoarthritis. Arthritis Rheum 48:3464–3474. https://doi.org/10.1002/art.11365
O’Driscoll SW, Saris DB, Ito Y, Fitzimmons JS (2001) The chondrogenic potential of periosteum decreases with age. J Orthop Res 19:95–103
Payne KA, Didiano DM, Chu CR (2010) Donor sex and age influence the chondrogenic potential of human femoral bone marrow stem cells. Osteoarthritis Cartil 18:705–713
Pogue R, Lyons K (2006) BMP signaling in the cartilage growth plate. Curr Top Dev Biol 76:1–48
Prockop DJ, Phinney DG, Bunnell BA (2008) Mesenchymal stem cells: methods and protocols. Humana Press Totowa, NJ
Radcliffe CH, Flaminio MJ, Fortier LA (2010) Temporal analysis of equine bone marrow aspirate during establishment of putative mesenchymal progenitor cell populations. Stem Cells Dev 19:269–282
Richardson LE, Dudhia J, Clegg PD, Smith R (2007) Stem cells in veterinary medicine–attempts at regenerating equine tendon after injury. Trends Biotechnol 25:409–416. https://doi.org/10.1016/j.tibtech.2007.07.009
Sacchetti B et al (2007) Self-renewing osteoprogenitors in bone marrow sinusoids can organize a hematopoietic microenvironment. Cell 131:324–336
Sakaguchi Y, Sekiya I, Yagishita K, Muneta T (2005) Comparison of human stem cells derived from various mesenchymal tissues: superiority of synovium as a cell source. Arthritis Rheum 52:2521–2529
Sekiya I, Colter DC, Prockop DJ (2001) BMP-6 enhances chondrogenesis in a subpopulation of human marrow stromal cells. Biochem Biophys Res Commun 284:411–418
Shademan M, Parham A, Dehghani H (2016) Enhancement of chondrogenic differentiation potential of equine adiposetissue-derived mesenchymal stem cells using TGF-ß3 and BMP-6. Turk J Biol 40:360–368
Smith R, Korda M, Blunn G, Goodship AJEVJ (2003) Isolation and implantation of autologous equine mesenchymal stem cells from bone marrow into the superficial digital flexor tendon as a potential novel treatment. Equine Vet J 35:99–102
Somoza RA, Welter JF, Correa D, Caplan AI (2014) Chondrogenic differentiation of mesenchymal stem cells: challenges and unfulfilled expectations. Tissue Eng B 20:596–608
Stewart AA, Byron CR, Pondenis H, Stewart MC (2007) Effect of fibroblast growth factor-2 on equine mesenchymal stem cell monolayer expansion and chondrogenesis. Am J Vet Res 68:941–945
Tew SR, Kwan A, Hann A, Thomson BM, Archier C (2000) The reactions of articular cartilage to experimental wounding. Arthritis Rheum 43:25
Vidal MA et al (2008) Comparison of chondrogenic potential in equine mesenchymal stromal cells derived from adipose tissue and bone marrow. Vet Surg 37:713–724
Villapol S, Logan TT, Symes AJ (2013) Role of TGF-β signaling in neurogenic regions after brain injury. Trends Cell Signal Pathw Neuronal Fate Decis. https://doi.org/10.5772/53941
Volk SW, Wang Y, Hankenson KD (2012) Effects of donor characteristics and ex vivo expansion on canine mesenchymal stem cell properties: implications for MSC-based therapies. Cell Transplant 21:2189–2200
Winter A et al (2003) Cartilage-like gene expression in differentiated human stem cell spheroids: a comparison of bone marrow–derived and adipose tissue–derived stromal cells. Arthritis Rheum 48:418–429
Worster AA, Nixon AJ, Brower-Toland BD, Williams J (2000) Effect of transforming growth factor β1 on chondrogenic differentiation of cultured equine mesenchymal stem cells. Am J Vet Res 61:1003–1010
Yang IH, Kim SH, Kim YH, Sun HJ, Kim SJ, Lee JW (2004) Comparison of phenotypic characterization between “alginate bead” and “pellet” culture systems as chondrogenic differentiation models for human mesenchymal stem cells. Yonsei Med J 45:891–900
Zahedi M, Parham A, Dehghani H, Mehrjerdi H (2017) Stemness signature of equine marrow-derived mesenchymal stem cells. Int J Stem Cells 10:93
Zuk PA et al (2002) Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 13:4279–4295
Acknowledgements
This study was financially supported by Ferdowsi University of Mashhad, Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has any potential financial conflict of interest related to this paper. No financial or other relationships could lead to a conflict of interests.
Ethics Approval
All animal procedures were approved by The Ethics and Animal Welfare Committee of the Ferdowsi University of Mashhad, Mashhad, Iran.
Research Involving Human Participants
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Taghavi, M., Parham, A. & Raji, A. The Combination of TGF-β3 and BMP-6 Synergistically Promotes the Chondrogenic Differentiation of Equine Bone Marrow-Derived Mesenchymal Stem Cells. Int J Pept Res Ther 26, 727–735 (2020). https://doi.org/10.1007/s10989-019-09880-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-019-09880-w