Abstract
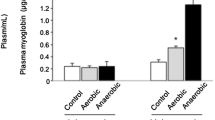
Striated muscles exhibit a wide range of metabolic activity levels. Heart and diaphragm are muscles with continuous contractile performance, which requires life-long function. In contrast, skeletal muscles like longissimus muscle can adapt metabolism from resting to different stages of exercise. The aim of this study was to compare the morphological features of these three muscles and the expression of genes that are important for energy metabolism. Therefore, histochemical studies were performed for determination of muscle fibre type composition. Oxidative and glycolytic capacity was assessed by measuring isocitrate dehydrogenase (ICDH) and lactate dehydrogenase (LDH) activities. The mRNA expression of glucose transporter 4 (GLUT 4), growth hormone receptor (GHR) and AMP-activated kinase (AMPK) α1 and α2 subunits was studied by semiquantitative Northern blotting. Heart, and to a slightly lesser extent diaphragm were highly oxidative muscles characterised by high expression of oxidative muscle fibres and ICDH activity. Longissimus muscle exhibited the highest percentage of glycolytic fibres and LDH activity. GLUT 4 mRNA was lowest in heart reflecting the dependency of heart muscle on fatty acids as major energy source. Higher expression of GLUT 4 in diaphragm indicated that glucose is an important energy substrate in this oxidative muscle. Highest GLUT 4 expression in longissimus should be essential for the refilling of glycogen stores after exercise. AMPK subunits, which are important stimulators of GLUT 4 protein insertion into the sarcolemma, are also highest expressed in longissimus muscle indicating the strong capacity to adapt energy metabolism to large changes in energy demand. Interestingly, AMPK α1 subunit expression on protein level is strongly restricted to muscle fibres containing type I myosin in this muscle. GHR mRNA expression was also highest in longissimus muscle indicating that an enhanced effect of growth hormone, which is described to be diabetogenic, could be involved in the lower insulin sensitivity of glycolytic muscles.







Similar content being viewed by others
References
Ai H, Ihlemann J, Hellsten Y, Lauritzen HP, Hardie DG, Galbo H, Ploug T (2002) Effect of fibre type and nutritional state on AICAR- and contraction-stimulated glucose transport in rat muscle. Am J Physiol Endocrinol Metab 282:E1291–E1300
An D, Pulinilkunnil T, Qi D, Ghosh S, Abrahani A, Rodrigues B (2005) The metabolic “switch” AMPK regulates cardiac heparin-releasable lipoprotein lipase. Am J Physiol Endocrinol Metab 288:E246–E253
Casse AH, Desplanches D, Mayet-Sornay MH, Raccurt M, Jegou S, Morel G (2003) Growth hormone receptor expression in atrophying muscle fibers of rats. Endocrinology 144:3692–3697
Chin LL, Tsai YL, Lee WC, Cho YM, Ho HY, Chen SM, Chen MT, Kuo CH (2005) Acute effect of exercise-hypoxia challenge on GLUT 4 protein expression in rat cardiac muscle. High Alt Med Biol 6:256–262
Davey KA, Garlick PB, Warley A, Southworth R (2007) An immunogold labelling study of the distribution of GLUT 1 & GLUT 4 in cardiac tissue following stimulation by insulin or ischemia. Am J Physiol Heart Circ Physiol 22: doi 00663.2006v2
Essen-Gustavsson B, Fjelkner-Modig S (1985) Skeletal muscle characteristics in different breeds of pigs in relation to sensory properties of meat. Meat Sci 13:33–47
Fiedler I, Wegner J, Feige KD (1991) Ein Wachstumsmodell für den Faserdurchmesser in zwei Muskeln des Schweines. Arch Tierz Berlin 34:57–62
Fiedler I, Ender K Wicke M, Maak G, v Lengerken G, Meyer W (1999) Structural and functional characteristics of muscle fibres in pigs with different malignant hyperthermia susceptibility (MHS) and different meat quality. Meat Sci 53:9–15
Fiedler I, Kuhn G, Hartung M, Nürnberg K, Rehfeldt C, Huber K (2001) Auswirkungen des Malignen Hyperthermie Syndroms (MHS) auf Fleischqualität, Muskelfasereigenschaften und Stoffwechselkriterien des M. longissimus von Pietrain-Schweinen. Arch Tierz Dummerstorf 44:203–217
Guth L, Samaha FJ (1970) Procedure for the histochemical demonstration of actomyosin ATPase. Exp Neurol 28:365–367
Hardie DG (2003) Minireview: the AMP-activated protein kinase cascade: the key sensor of cellular energy status. Endocrinology 144:5179–5183
He A, Liu X, Liu L, Chang Y, Fang F (2007) How many signals impinge on GLUT4 activation by insulin. Cell Signal 19:1–7
Henckel P, Oksbjerg N, Erlandsen E, Barton-Gade P, Bejerholm C (1997) Histo- and biochemical characteristics of the Longissimus dorsi muscle in pigs and their relationship to performance and meat quality. Meat Sci 47:311–321
Horak V (1983) Successive histochemical staining for succinate dehydrogenase and “reversed”-ATPase in a single selection for the skeletal muscle fibre typing. Histochemistry 78:545–553
Hou CW, Chou SW, Ho HY, Lee WC, Lin CH, Kuo CH (2003) Interactive effect of exercise training and growth hormone administration on glucose tolerance and muscle GLUT 4 protein expression in rats. J Biomed Sci 10:689–696
Huber K, Roesler U, Muscher A, Hansen K, Widiyono I, Pfeffer E, Breves G (2003) Ontogenesis of epithelial phosphate transport systems in goats. Am J Physiol Regul Intergr Comp Physiol 284:R413–421
Jorgensen SB, Viollet B, Andreelli C, Frosig C, Birk JB, Schierling P, Vaulont S, Richter EA, Wojtaszewski JF (2004) Knockout of the α2 but not α1 5´AMP-activated protein kinase isoform abolishes 5-Aminoimidazole-4-carboxamide-1-β-4-ribofuranoside- but not contraction-induced glucose uptake in skeletal muscle. J Biol Chem 279:1070–1079
Jorgensen JO, Vestergaard ET, Krag M, Nielsen C, Moller L, Gormsen L, Jessen N (2006a) Skeletal muscle as a metabolic target for growth hormone. Horm Res 66(Suppl 1):22–25
Jorgensen SB, Richter EA, Wojtaszewski JF (2006b) Role of AMPK in skeletal muscle metabolic regulation and adaptation in relation to exercise. J Physiol 574(1):17–31
Katsumata M, Burton KA, Li J, Dauncey MJ (1999) Suboptimal energy balance selectively up-regulates muscle GLUT gene expression but reduces insulin-dependent glucose uptake during postnatal development. FASEB J 13:1405–1413
Katsumata M, Cattaneo D, White P, Burton KA, Dauncey MJ (2000) Growth hormone receptor gene expression in porcine skeletal and cardiac muscles is selectively regulated by postnatal undernutrition. J Nutr 130:2482–2488
Kraniou GN, Cameron-Smith D, Hargreaves M (2006) Acute exercise and GLUT 4 expression in human skeletal muscle: influence of exercise intensity. J Appl Physiol 101:934–937
Lefaucheur L, Milan D, Ecolan P, Le Callenec C (2004) Myosin heavy chain composition of different skeletal muscles in Large White and Meishan pigs. J Anim Sci 82:1931–1941
Le Roith D, Bondy C, Yakar S, Liu JL, Butler A (2001) The somatomedin hypothesis: 2001. Endocr Rev 22:53–74
Novikoff AB, Shin Vy, Drucker J (1961) Mitochondrial localization of oxidation enzymes: staining results with two tetrazolium salts. J Biophys Biochem Cytol 9:47–61
Onay-Besikci A (2006) Regulation of cardiac energy metabolism in newborn. Mol Cell Biochem 287:1–11
Petersen HA, Fueger PT, Bracy DP, Wasserman DH, Halseth AE (2003) Fiber type-specific determinants of Vmax for insulin-stimulated muscle glucose uptake in vivo. Am J Physiol Endocrinol Metab 284:541–548
Petzold J (2006) Parameter des Muskelenergiestoffwechsels in genetisch differenten Schweinen. Dissertation, School of Veterinary Medicine, Hannover, Germany
Toniolo L, Patruno M, Maccatrozzo L, Pellegrino MA, Canepari M, Rossi R, D’Antona G, Botticelli R, Reggiani C, Mascarello F (2004) Fast fibres in a large animal: fibre types, contractile properties and myosin expression in pig skeletal muscles. J Exp Biol 207:1875–1886
Toyoda T, Hayashi T, Miyamoto L, Yonemitsu S, Nakano M, Tanaka S, Ebihara K, Masuzaki H, Hosoda K, Inoue G, Otaka A, Sato K, Fushiki T, Nakao K (2004) Possible involvement of the alpha 1 isoform of 5´AMP-activated protein kinase in oxidative stress-stimulated glucose transport in skeletal muscle. Am J Physiol Endocrinol Metab 287:E166–E173
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huber, K., Petzold, J., Rehfeldt, C. et al. Muscle energy metabolism: structural and functional features in different types of porcine striated muscles. J Muscle Res Cell Motil 28, 249–258 (2007). https://doi.org/10.1007/s10974-007-9123-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-007-9123-8