Abstract
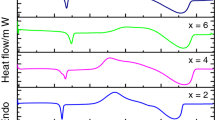
Multicomponent glassy alloys Se78−xTe18Sn2Sbx (x = 0, 2, 4 and 6) have been synthesized using melt quench technique. The prepared samples have been characterized by X-ray diffraction technique and differential scanning calorimetry (DSC). Glass transition kinetics of Se78−xTe18Sn2Sbx (x = 0, 2, 4 and 6 at.%) glassy alloys has been examined using DSC. DSC runs have been recorded at different heating rates (5, 10, 15 and 20 K min−1) for each sample under investigation. Heating rate dependence of glass transition temperature (Tg) has been studied using Lasocka empirical relation. The activation energy of glass transition has been evaluated using Kissinger and Moynihan’s relation. The effect of antimony concentration on glass transition temperature and activation energy has been investigated in the prepared samples. Glass-forming ability and thermal stability of Se78−xTe18Sn2Sbx (x = 0, 2, 4 and 6) glassy alloys have been monitored through the evaluation of thermal stability using Dietzal relation, Hurby parameter, and Saad and Poulin parameter. The above-mentioned parameters are found to be compositionally dependent, which indicates that among the studied glass samples the stability is maximum for Sb at 2% content.








Similar content being viewed by others
References
Ammar AH, Faridb AM, Farag AAM. Non-isothermal kinetic analysis of crystallization of vacuum prepared Se90In9.9Cu0.1 alloy thin films. J Non-Cryst Solids. 2016;434:85–91.
Madan A, Shaw MP. The physics and applications of amorphous semiconductors. New York: Academic Press; 1988.
Singh AK. A short over view on advantage of chalcogenide glassy alloys. J Non-Oxide Glasses. 2012;3(1):1–4.
Stronski A, Paiuk O, Gudymenko A, Kladko V, Oleksenko P, Vuichyk N, Vlček M, Lishchynskyy I, Lahderanta E, Lashkul A, Gubanova A, Kryskov T. Effect of doping by transitional elements on properties of chalcogenide glasses. Ceram Int. 2015;41:7543–8.
Tanaka K, Shimakawa K. Amorphous chalcogenide semiconductors and related materials. New York: Springer; 2011.
Mouawad O, Vitry P, Strutynski C, Picot-Clémente J, Désévédavy F, Gadret G, Jules JC, Lesniewska E, Smektala F. Atmospheric aging and surface degradation in As2S3 fibers in relation with suspended-core profile. Opt Mater. 2015;44:25–32.
Srivastava A, Tiwari SN, Upadhaya AN, Zulfequar M, Khan SA. First-order phase transformation and structural studies in Se85In15−xZnx chalcogenide glasses. J Therm Anal Calorim. 2017;129:1435–44.
Mohamed M, Abd-el Salam MN, Abdel-Rahim MA, Abdel-Latief AY, Shaaban ER. Effect of Ag addition on crystallization kinetics and thermal stability of As–Se chalcogenide glasses. J Therm Anal Calorim. 2018;132:91–101.
Lafi OA, Imran MMA. Microscopic origin of demixing in Ge20SexTe80−x alloys. J Alloys Compd. 2011;509:5090–4.
Patial BS, Thakur N, Tripathi SK. Crystallization study of Sn additive Se–Te chalcogenide alloys. J Therm Anal Calorim. 2011;106:845–52.
Terao M, Morikawa T, Ohta T. Electrical phase-change memory: fundamentals and State of the Art. Jpn J Appl Phys. 2009;48:080001–14.
Lafi OA. Correlation of some opto-electrical properties of Se–Te–Sn glassy semiconductors with the average single bond energy and the average elctronegativity. J Alloys Compd. 2016;660:503–8.
Heera P, Kumar A, Sharma R. Physical and dielectric properties of Sn doped Se–Te glassy system. J Ovonic Res. 2012;8(2):29–40.
Kumar H, Mehta N, Singh K. Calorimetric studies of glass transition phenomenon in glassy Se80−xTe20Snx alloys. Phys Scr. 2009;80:065602.
Kumar H, Mehta N. Kinetic parameters of glass transition and crystallization for glassy Se and glassy Se98M2 (M = In, Sb and Sn) alloys. Mater Chem Phys. 2012;134:834–8.
Lafi OA. ImranMMA, AbdullahMK, Al-SakhelSA, Thermal characterization of Se100 − xSnx(x = 4, 6 and 8) chalcogenide glasses using differential scanning calorimeter. Thermochim Acta. 2013;560:71–5.
Srivastava A, Mehta N. Investigation of some thermo-mechanical and dielectric properties in multi-component chalcogenide glasses of Se–Te–Sn–Ag quaternary system. J Alloys Compd. 2016;658:533–42.
Kumar H, Mehta N. Thermal characterization of Se78−xTe20Sn2Pbx (0 ≤ x ≤ 6) glassies for phase change optical recording technique. Glass Phys Chem. 2013;39:490–8.
Kumar H, Mehta N. Kinematical Studies of Thermal Crystallization in Glassy Se78−xTe20Sn2Bix (0 ≤ x ≤ 6) Alloys. J Adv Phys. 2013;2:163–9.
Saraswat S, Sharma SD. Investigation of silver as chemical modifier for tailoring of some physico-chemical properties in inorganic glassy Se80Te20 alloy. Glass Phys Chem. 2015;41(4):402–9.
Rao V, Dwivedi DK. Glass transition kinetics and thermal stability of Se82−xTe18Sbx (x = 0, 4, 8 and 12 at.%) glassy alloys. J Mater Sci Mater Electron. 2017;28(8):6208–16.
Rao V, Dwivedi DK. Crystallization kinetics of Se82−xTe18Sbx (x = 0, 4, 8, 12) glassy alloys by iso-conversional approach. Mater Res Innov. 2017. https://doi.org/10.1080/14328917.2017.1401389.
Dimitrov D, Tzocheva D, Kovacheva D. Calorimetric study of amorphous Sb–Se thin films. Thin Solid Films. 1998;323:79–84.
Thornburg DD. Substrate effects on the crystallization kinetics of amorphous selenium thin films. Thin Solid Films. 1979;37:215–9.
Rabinal MK, Sangunni KS, Gopal ESR. Chemical ordering in Ge20Se80−xInx glasses. J Non-Cryst Solids. 1995;188:98–106.
Lasocka M. The effect of scanning rate on glass transition temperature of splat-cooled Te85Ge15. Mater Sci Eng. 1976;23:173–7.
Yin H, Li L, Liu Y, Hu L, Zeng H, Chen G. Impact of tellurium on glass transition and crystallization in the Ge–Se–Te–Bi system. Ceram Inter. 2016. https://doi.org/10.1016/j.ceramint.2016.11.167.
Imran MMA, Bhandari D, Saxena NS. Kinetic studies of bulk Ge22Se78−xBix (x = 0, 4 and 8) semiconducting glasses. J Therm Anal Calorim. 2001;65:257–74.
Moynihan CT, Easteal AJ, Wilder J, Tucke J. Dependence of the glass transition temperature on heating and cooling rate. J Phys Chem. 1974;78:2673–7.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem. 1957;29:1702–6.
Fayek SA. Study of non-isothermal kinetics, electrical and optical properties of (GaSeTe) films. Vacuum. 2004;72:11–20.
Pascual MI, Lara C, Duran A. Non-isothermal crystallization kinetics of devitrifying RO–BaO–SiO2 (R=Mg, Zn) glasses. Phys Chem Glasses Eur J Glass Sci Technol B. 2006;47:572–81.
Mehta N, Singh K, Kumar A. On the glass transition phenomenon in Se–Te and Se–Ge based ternary chalcogenide glasses. Phys B. 2009;404:1835–9.
Mehta N, Kumar A. Applicability of Kissinger’s relation in the determination of activation energy of glass transition process. J Optoelectron Adv Mater. 2009;7(3):1473–8.
Moynihan CT, Cantor S. Viscosity and its temperature dependence in molten BeF2. J Chem Phys. 1968;48:115–21.
Angell CA. Structural instability and relaxation in liquid and glassy phases near the fragile liquid limit. J Non-Cryst Solids. 1988;102(1–3):205–21.
Bohmer R, Ngai KL, Angell CA, Plazek DJ. Non-exponential relaxations in strong and fragile glass formers. J Chem Phys. 1993;99:4201–9.
Sharma A, Mehta N. Observation of switching behavior in some multi-component glasses of Se–Te–Sn–Pb system. Mater Lett. 2016;178:178–80.
Zanotto ED. Isothermal and adiabatic nucleation in glass. J Non-Cryst Solids. 1987;89:10–6.
Zanotto ED. Experimental tests of the classical nucleation theory for glasses. J Non-Cryst Solids. 1985;74:373–94.
Nascimento MLF, Souza LA, Ferreira EB, Zanotto ED. Can glass stability parameters infer glass forming ability? J Non-Cryst Solids. 2005;351:3296–308.
Turnbll D. Under what conditions can a glass be formed? Contemp Phys. 1969;10:473–88.
Dietzel A. Glass structure and glass properties. Glasstech Ber. 1968;22:41.
Hruby A. Evaluation of glass-forming tendency by means of DTA C. Zech J Phys B. 1972;22:1187–93.
Hruby A. Glass-forming tendency in the GeSx system. Czech J Phys B. 1973;23:1263–72.
Rahim MAA, Hafiz MM, Mahmoud AZ. Crystallization kinetics and thermal stability inSe85−xTe15Sbx chalcogenide glasses. Phase Transit. 2016. https://doi.org/10.1080/01411594.2015.1136912.
Saad M, Pouling M. Glass forming ability criterion. Mater Sci Foum. 1987;19:11–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rao, V., Chandel, N., Mehta, N. et al. Effect of antimony on glass transition and thermal stability of Se78−xTe18Sn2Sbx (x = 0, 2, 4 and 6 at.%) multicomponent glassy alloys. J Therm Anal Calorim 134, 915–922 (2018). https://doi.org/10.1007/s10973-018-7309-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-018-7309-5