Abstract
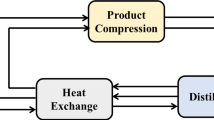
Chemical looping air separation is a new method to separate oxygen from air. In the process, oxygen carrier releases oxygen in the reduction reactor and absorbs oxygen in the oxidation reactor. The reduction is endothermic, and the oxidation is exothermic. The heat transfer from oxidation reactor to reduction reactor is essential to design the CLAS process with low energy consumption. In this paper, the optimization CLAS process of CuO/ZrO2 oxygen carrier was established based on thermal analysis and pressure analysis. In the system, the heat can achieve self-balance with little heat supplied from outside. The effects of the technological parameters on the system energy consumption were investigated using Aspen Plus software. The results show that the energy consumption decreases with reduction temperature increasing and oxidation temperature decreasing. Moreover, the smaller the difference of temperature between the two reactors is, the lower energy consumption is. Under negative pressure, with the decreasing reduction pressure, the energy consumption decreases. Under positive pressure, with the increasing oxidation pressure, the energy consumption decreases firstly and then increases. The optimization operating parameters are obtained. The lowest energy consumption of the system is 0.132 kWh m−3 at the conditions of 990 °C reduction and 1000 °C oxidation temperatures and 0.5 atm. reduction and 1.7 atm. oxidation pressures.








Similar content being viewed by others
References
Hashim SS, Mohamed AR, Bhatia S. Oxygen separation from air using ceramic-based membrane technology for sustainable fuel production and power generation. Renew Sust Energy Rev. 2011;15(2):1284–93.
Smith AR, Klosek J. A review of air separation technologies and their integration with energy conversion processes. Fuel Process Technol. 2001;70(2):115–34.
Moghtaderi B. Application of chemical looping concept for air separation at high temperatures. Energy Fuels. 2010;24(1):190–8.
Adanez-Rubio I, Gayan P, Abad A, de Diego LF, Garcia-Labiano F, Adanez J. Evaluation of a spray-dried CuO/MgAl2O4 oxygen carrier for the chemical looping with oxygen uncoupling process. Energy Fuels. 2012;26:3069–81.
Shah K, Moghtaderi B, Wall T. Selection of suitable oxygen carriers for chemical looping air separation: a thermodynamic approach. Energy Fuels. 2012;26(4):2038–45.
Shulman A, Cleverstam E, Mattisson T, Lyngfelt A. Manganese/iron, manganese/nickel, and manganese/silicon oxides used in chemical-looping with oxygen uncoupling (CLOU) for combustion of methane. Energy Fuels. 2009;23:5269–75.
Wang K, Yu QB, Qin Q, Duan WJ. Feasibility of a Co oxygen carrier for chemical looping air separation: thermodynamics and Kinetics. Chem Eng Technol. 2014;37(9):1500–6.
Song H, Shah K, Doroodchi E, Wall T, Moghtaderi B. Reactivity of Al2O3- or SiO2-supported Cu-, Mn-, and Co-oxygen carriers for chemical looping air separation. Energy Fuels. 2014;28:1284–94.
Song H, Shah K, Doroodchi E, Moghtaderi B. Development of a Cu-Mg-based oxygen carrier with SiO2 as a support for chemical looping air separation. Energy Fuels. 2014;28(1):163–72.
Wang K, Yu QB, Qin Q. The thermodynamic method for selecting oxygen carriers used for chemical looping air separation. J Therm Anal Calorim. 2013;112(2):747–53.
Wang K, Yu QB, Duan WJ, Qin Q. The adaptability of Cu/Zr oxides as oxygen carrier used for chemical looping air separation (CLAS). J Therm Anal Calorim. 2014;115(5):1163–72.
Wang K, Yu QB, Xie HQ, Qin Q, Li JC. Properties of Cu-based oxygen carrier used for chemical looping oxygen production. J Inorg Mater. 2013;28(10):1115–20.
Wang K, Yu QB, Qin Q, Duan WJ, Wu TW. Study of the sorption property of copper oxygen carrier used for chemical looping air separation. J Therm Anal Calorim. 2015;120(3):1627–33.
Wang K, Yu QB, Qin Q, Zuo ZL. Analysis of oxygen releasing rate of Cu-based oxygen carrier in N2–O2 atmosphere. J Therm Anal Calorim. 2015;119(3):2221–7.
Wang K, Yu QB, Qin Q. Reduction kinetics of Cu-based oxygen carriers for chemical looping air separation. Energy Fuels. 2013;27(9):5466–74.
Wang K, Yu QB, Qin Q, Li JC, Wang ZM. Kinetics analysis of Cu–Zr oxygen carrier for chemical looping oxygen production. J Inorg Mater. 2014;39(3):301–8.
Zhang T. Experimental study and analyses of O2–CO2 production based on chemical looping method. Beijing: Qinghua University; 2010.
Moldenhauer P, Ryden M, Mattisson T, Lyngfelt A. Chemical-looping combustion and chemical-looping with oxygen uncoupling of kerosene with Mn- and Cu-based oxygen carriers in a circulating fluidized-bed 300 W laboratory reactor. Fuel Process Technol. 2012;104:378–89.
Arjmand M, Azad A, Leion H, Lyngfelt A, Mattisson T. Prospects of Al2O3 and MgAl2O4-supported CuO oxygen carriers in chemical-looping combustion (CLC) and chemical-looping with oxygen uncoupling (CLOU). Energy Fuels. 2011;25(11):5493–502.
Karimi E, Forutan HR, Saidi M, Rahimpour MR, Shariati A. Experimental study of chemical-looping reforming in a fixed-bed reactor: performance investigation of different oxygen carriers on Al2O3 and TiO2 support. Energy Fuels. 2014;28:2811–20.
Corbella BM, De Diego LF, Garcia-Labiano F, Adanez J, Palacios JM. Characterization study and five-cycle tests in a fixed-bed reactor of titania-supported nickel oxide as oxygen carriers for the chemical-looping combustion of methane. Environ Sci Technol. 2005;39(15):5796–803.
Gayan P, Adanez-Rubio I, Abad A, de Diego LF, Garcia-Labiano F, Adanez J. Development of Cu-based oxygen carriers for chemical-looping with oxygen uncoupling (CLOU) process. Fuel. 2012;96(1):226–38.
Acknowledgements
The present work was supported by National Natural Science Foundation of China (51576035) and National Science & Technology Pillar Program of China (2013BAA03B03).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, K., Yu, Q., Hou, L. et al. Simulation and energy consumption analysis of chemical looping air separation system on Aspen Plus. J Therm Anal Calorim 124, 1555–1560 (2016). https://doi.org/10.1007/s10973-016-5237-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5237-9