Abstract
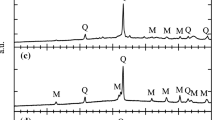
The objective of the present study was to use the zero-order reaction rate (ZORR) assumption and transition state theory (TST) to examine the kinetics of the thermal changes in solids. The method was firstly applied to the thermal transformation of spinel–mullite in a kaolinitic clay. The selected clay contains kaolinite (K), illite (I), and quartz (Q) minerals. The cylindrical compacts of the clay powder with a diameter of 40.22 mm were prepared under 150 MPa by uniaxial pressing using water (7 % by mass) as binder. The compacts after dried at 100 °C for 4 h were fired at various temperatures in the interval of 900–1050 °C by changing the time between 20 and 120 min. Besides the bulk density measurements, phase transformation in the compacts depending on the firing temperature was examined by X-ray diffraction and scanning electron microscopy techniques. Time derivative of the bulk densities for each firing temperature was taken as the transformation rate constant (k) according to the ZORR assumption. Arrhenius equation for the transformation was found as k = A exp(−E #/RT) = (136 kg m−3 s−1) exp(−12,022 J mol−1/RT) where A is the proexponential factor, E # is the activation energy, R is the universal gas constant and T is the absolute temperature of firing. The most general thermodynamic relation of the activation for the transformation was obtained from the TST with the SI units as ΔG # = −RT ln K # = ΔH # − TΔS # = 89,367 + 224T where ΔG #, K #, ΔH #, and ΔS # are the Gibbs energy, equilibrium constant, enthalpy, and entropy for the activation of the thermal transformation, respectively. The positive value of the ΔG # between 900 and 1050 °C indicated that the activation complex for the transformation does not form spontaneously.








Similar content being viewed by others
References
Kingery WD, Bowen HK, Uhlmann DR. Introduction to ceramics. 2nd ed. New York: Wiley-Interscience; 1976.
Kang SJL. Sintering densification grain growth and microstructure. Butterworth: Elsevier; 2005.
German RM. Sintering theory and practice. New York: Wiley; 1996.
Bartholomew CH. Sintering kinetics of supported metals: new perspectives from a unifying GPLE treatment. Appl Catal A Gen. 1993;107:1–57.
Su H, Johnson DL. Practical approach to sintering. Am Ceram Soc Bull. 1997;76:72–6.
Perez-Maqueda LA, Criado JM, Real C. Kinetics of the initial stage of sintering from shrinkage data: simultaneous determination of activation energy and kinetic model from a single nonisothermal experiment. J Am Ceram Soc. 2002;85:763–8.
Roy SN, Saha SR, Guha SK. Sintering kinetics of pure and doped chromium oxide. J Mater Sci. 1986;21:3673–6.
Thompson AM, Harmer MP. Influence of atmosphere on the final-stage sintering kinetics of ultra-high-purity alumina. J Am Ceram Soc. 1993;76:2248–56.
Ting CJ, Lu HY. Defect reactions and the controlling mechanism in the sintering of magnesium aluminate spinel. J Am Ceram Soc. 1999;82:841–8.
Wang H, Liu X, Chen F, Meng G, Sorensen OT. Kinetics and mechanism of a sintering process for macroporous alumina ceramics by extrusion. J Am Ceram Soc. 1998;81:781–4.
German RM, Munir ZA. Surface area reduction during isothermal sintering. J Am Ceram Soc. 1976;59:379–83.
Sarıkaya Y, Ada K, Önal M. Applications of the zero-order reaction rate model and transition state theory on the intra-particle sintering of an alumina powder by using surface area measurements. J Alloy Compd. 2007;432:194–9.
Wang H, Li C, Peng Z, Zhang S. Characterization and thermal behavior of kaolin. J Therm Anal Calorim. 2011;105:157–60.
Wang Z, Marin G, Naterer GF, Gabriel KS. Thermodynamics and kinetics of the thermal decomposition of cupric chloride in its hydrolysis reaction. J Therm Anal Calorim. 2015;119:815–23.
Dion P, Alcover JF, Bergaya F, Ortega A, Llevellyn PL, Rouquerol F. Kinetic study by controlled-transformation rate thermal analysis of the dehydroxylation of kaolinite. Clay Miner. 1998;33:269–76.
Edomwonyi-Otu LC, Aderemi BO, Ahmed AS, Coville NJ, Maaza M. Influence of thermal treatment on Kankara kaolinite. Opticon. 1826;2013(15):1–5.
Chakraborty AK. Phase transformation of kaolinite clay. Berlin: Springer; 2014.
Kahraman S, Önal M, Sarıkaya Y, Bozdoğan İ. Characterization of silica polymorphs in kaolins by X-ray diffraction before and after phosphoric acid digestion and thermal treatment. Anal Chim Acta. 2005;552:201–6.
Önal M, Sarıkaya Y. The effect of heat treatment on the paracrystallinity of an opal-CT found in a bentonite. J Non-Cryst Solids. 2007;353:4195–8.
Önal M, Kahraman S, Sarıkaya Y. Differentiation of α-cristobalite from opals in bentonites from Turkey. Appl Clay Sci. 2007;35:25–30.
Choudhary VR, Vaidya SH. Simple specific gravity bottle methods for measuring porosity of solid catalysts and adsorbents. Res Ind. 1981;26:1–3.
Hugo P, Koch H. Production of porous alumina with defined bimodal pore structure. Ger Chem Eng. 1979;2:24–30.
Baran B, Sarıkaya Y, Alemdaroğlu T, Önal M. The effect of boron containing frits on the anorthite formation temperature in kaolin-wollastonite mixtures. J Euro Ceram Soc. 2003;21:2061–6.
Ada K, Önal M, Sarıkaya Y. Investigation of the intra-particle sintering kinetics of a mainly agglomerated alumina powder by using surface area reduction. Powder Technol. 2006;168:37–41.
Sarıkaya Y, Önal M. High temperature carburizing of a stainless steel with uranium carbide. J Alloys Compd. 2012;542:253–6.
Acknowledgements
The authors thank to Ankara University Research Fund (Project No.: 12B4240016) for financial support to this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pekdemir, A.D., Sarıkaya, Y. & Önal, M. Thermal transformation kinetics of a kaolinitic clay. J Therm Anal Calorim 123, 767–772 (2016). https://doi.org/10.1007/s10973-015-4866-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-015-4866-8