Abstract
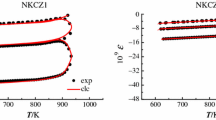
The structural relaxation of three compositional series of PbO–WO3–P2O5 glasses with composition (0.5 − x/2)PbO·xWO3·(0.5 − x/2)P2O5, x = 0, 0.1, 0.2, 0.3, 0.4, and 0.5; 0.5PbO·xWO3·(0.5 − x)P2O5, x = 0, 0.1, 0.2, and 0.3; and (0.5 − x)PbO·xWO3·0.5P2O5, x = 0, 0.1, 0.2, 0.3, 0.4, and 0.5 was studied by thermomechanical analysis. The structural relaxation was studied in the transformation region using the Tool–Narayanaswamy–Moynihan’s and Tool–Narayanaswamy–Mazurin’s models. The relaxation function of Kohlrausch Williams and Watts was used. The parameters of both models were calculated by nonlinear regression analysis of thermodilatometric curves measured by thermomechanical analyzer under the constant load. Both models very well describe the experimental data. It was found that the modulus is increasing with increasing amount of WO3 in all glasses. On the contrary, the width of the spectrum of relaxation times is decreasing with increasing amount of WO3 in all studied glasses. Both models possess the values of metastable melt thermal expansion coefficient equal to their experimental value.









Similar content being viewed by others
References
Brow RK. Review: the structure of simple phosphate glasses. J Non-Cryst Solids. 2000;263&264:1–28.
Šubčík J, Koudelka L, Mošner P, Montagne L, Tricot G, Delevoye L, Gregora I. Glass-forming ability and structure of ZnO–MoO3–P2O5 glasses. J Non-Cryst Solids. 2010;356:2509–16.
Mošner P, Vosejpková K, Koudelka L, Montagne L, Revel B. Structure and properties of glasses in ZnO–P2O5–TeO2 system. J Non-Cryst Solids. 2011;357:2648–52.
Šubčík J, Koudelka L, Mošner P, Montagne L, Revel B, Gregora I. Structure and properties of MoO3-containing zinc borophosphate glasses. J Non-Cryst Solids. 2009;355:970–5.
Sułowska J, Wacławska I, Szumera M. Effect of copper addition on glass transition of silicate–phosphate glasses. J Therm Anal Calorim. 2012;109:705–10.
Sinouh H, Bih L, Azrour M, El Bouari A, Benmokhtar S, Manoun B, Belhorma B, Baudin T, Berthet P, Solas D, Haumont R. Effect of TiO2 and SrO additions on some physical properties of 33Na2O−xSrO−xTiO2−(50 − 2x)B2O3−17P2O5 glasses. J Therm Anal Calorim. 2013;111:401–8.
Garcia-Valles M, Hafez HS, Cruz-Matias I, Verges E, Aly MH, Nogues J, Ayala D, Martinez S. Calculation of viscosity–temperature curves for glass obtained from four wastewater treatment plants in Egypt. J Therm Anal Calorim. 2013;111:107–14.
Varshneya AK. Fundamentals of inorganic glasses. Sheffield: Society of Glass technology; 2006.
Vogel W. Glass chemistry. 2nd ed. Berlin: Springer; 1994.
Gutzow I, Schmelzer J. The vitreous state. Berlin: Springer; 1995.
Rao KJ. Structural chemistry of glasses. London: Elsevier; 2002.
SciGlass: Glass Property Information System. http://www.sciglass.info; http://www.sciglassweb.com. Jan 2013.
Scherer GW. Relaxation in glass and composites. New York: Willey; 1986. p. 331.
Mazurin OV. Steklovanie Nauka. Leningrad; 1986. p. 158.
Uhlmann DR, Kreidl NJ. Viscosity and relaxation glass: science and technology, vol. 3. New York: Academic Press; 1986.
Mazurin OV, Startsev JK, Chodakovskaja RJ. Relaksacionnaja teorija otžiga stekla i rasčet na jejo osnove režimov otžiga. Moskva: MChTI; 1986.
Tool AQ. Relation between inelastic deformability and thermal expansion of glass in its annealing range. J Am Ceram Soc. 1946;29:240–53.
Liška M, Štubňa I, Antalík J, Perichta P. Structural relaxation with viscous flow followed by thermodilatometry. Ceramics–Silikáty. 1996;40:15–9.
Liška M, Klyuev VP, Antalík J, Štubňa I. Thermodilatometry and structural relaxation of Na2O·2SiO2–Na2O·2TiO2 glasses. Ceramics–Silikáty. 1996;40:85–91.
Narayanaswamy OS. A model structural relaxation in glass. J Am Ceram Soc. 1971;54:491–8.
Scherer GW. Volume relaxation far from equilibrium. J Amer Ceram Soc. 1986;69:374–81.
Bach H, Krause D. Analysis of the composition and structure of glass and glass ceramics. Berlin: Springer; 1999.
Williams G, Watts DC, Dev BS, North AM. Further considerations of non symmetrical dielectric relaxation behavior arising from a simple empirical decay function. Trans Faraday Soc. 1971;67:1323–35.
Koudelka L, Lissová M, Rösslerová I, Mošner P, Černošek Z, Liška M, Montagne L, Delevoye L, Delevoye L. Structure and properties of lead tungstate phosphate glasses. Phys Chem Glasses: Eur J Glass Sci Technol B. 2012;53:86–92.
Acknowledgments
This work was supported by the Slovak Grant Agency for Science under the grant VEGA 1/0006/12 and the bilateral project evaluation SK-CZ-0007-11. This publication was created in the frame of the project ZDESJE, ITMS code 26220220084, of the Operational Program Research and Development funded from the European Fund of Regional Development. This work was supported by the Slovak Research and Development Agency Project ID: APVV-0487-11. The Czech authors are grateful for the financial support from the Grant Agency of the Czech Republic (Grant No. 13-00355S).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chromčíková, M., Liška, M., Lissová, M. et al. Structural relaxation of PbO–WO3–P2O5 glasses. J Therm Anal Calorim 114, 947–954 (2013). https://doi.org/10.1007/s10973-013-3082-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-013-3082-7