Abstract
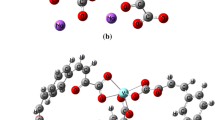
Solid-state LnL3·1.25H2O compounds, where L is oxamate and Ln is light trivalent lanthanides, have been synthesized. Simultaneous thermogravimetry and differential scanning calorimetry (TG–DSC), experimental and theoretical infrared spectroscopy, TG–DSC coupled to FTIR, elemental analysis, complexometry, and X-ray powder diffractometry were used to characterize and to study the thermal behavior of these compounds. The results led to information about the composition, dehydration, thermal stability, thermal decomposition, and gaseous products evolved during the thermal decomposition of these compounds in dynamic air atmosphere. The dehydration occurs in a single step and through a slow process. The thermal decomposition of the anhydrous compounds occur in a single (Ce), two (Pr), and three (La, Nd to Gd) steps with the formation of the respective oxides, CeO2, Pr6O11, and Ln2O3 (Ln = La, Nd to Gd). The theoretical and experimental spectroscopic study suggests that the carboxylate group and amide carbonyl group of oxamate are coordinate to the metals in a bidentate chelating mode.





Similar content being viewed by others
References
Wallace F, Wagner E. Infrared and far-infrared spectra of solid oxamic acid, deutero-oxamic acid and their salts. Spectrochim Acta A. 1978;34:589–606.
Perlepes SP, Zafiropoulos THF, Kouinis JK, Galinos AG. Complexes of zinc-group metals with oxamic acid. Inorg Nucl Chem Lett. 1980;16:475–80.
Shoeters G, Deleersnuder D, Desseyn HO. The complexes of oxamic acid with Ni(II). Spectrochim Acta A. 1983;39:71–6.
Allan JR, Dalrymple J. The thermal, spectral and magnetic studies of oxamic acid compounds of cobalt(II), nickel(II) and copper(II) ions. Thermochim Acta. 1993;221:199–204.
Vansant C, Desseyn HO, Perlepes SP. The synthesis, spectroscopic and thermal study of oxamic acid compounds of some metal(II) ions. Trans Met Chem. 1995;20:454–9.
Keuleers R, Janssens J, Desseyn HO. Thermal analysis of oxamates, thiooxamates and their complexes Part 1 The ligands. Thermochim Acta. 1998;311:149–54.
Keuleers R, Janssens J, Desseyn HO. Thermal analysis of oxamates, thiooxamates and their complexes Part 2. The Cu(II) complexes. Thermochim Acta. 1998;311:155–62.
Rodrigues-Martin Y, Ruiz-Pérez C, Gonzáles-Platas J, Sanchiz J, Lloret F, Julve M. A new eight-coordinate complex of manganese(II): synthesis, crystal structure, spectroscopy and magnetic properties of [Mn(Hoxam)2(H2O)4] (H2 oxam_oxamic acid). Inorg Chim Acta. 2001;315:120–5.
Caires FJ, Lima LS, Carvalho CT, Siqueira AB, Treu-Filho O, Ionashiro M. Thermal and spectroscopic data to investigate the oxamic acid, sodium oxamate and its compounds with some bivalent transition metal ions. J Therm Anal Calorim. 2011;107:335–344.
Nunes RS, Bannach G, Luiz JM, Caires FJ, Carvalho CT, Ionashiro M. Thermal studies on solid 1,4-bis(3-carboxy-3-oxo-prop-1-enyl) benzene of lighter trivalent lanthanides. J Therm Anal Calorim. 2011;1006:525–9.
Dametto PR, Caires FJ, Ambrozini B, Ionashiro M. Synthesis, characterization and thermal behaviour of light trivalent lanthanides folates on solid state. J Therm Anal Calorim. 2011;1005:831–6.
Perlepes SP, Zafiropoulos TF, Kouinis JK, Galinos AG. Lanthanide(III) complexes of oxamic acid. Z Naturforsch B. 1981;36:697–703.
Veltsistas PG, Christos PD, Karayannis MI. Synthesis and X-ray structure of an (oxamato)praseodymium polymer, [Pr(HNCOCO2)1.5(H2O)3]n·2.75H2O. Polyhedron. 1995;14:1251–3.
Lazaridou V, Perlepes SP, Tsangaris M. Synthesis, physical properties ans spectroscopic studies of oxamato (-1) lanthanide(III) omplexes. J Less-Common Met. 1990;158:1–14.
D’Assunção LM, Giolito I, Ionashiro M. Thermal decomposition of the hydrated basic carbonates of lanthanides and yttrium. Thermochim Acta. 1980;137:319–30.
Flaschka HA. EDTA titrations. 2nd ed. Oxford: Pergamon Press; 1964.
Ionashiro M, Graner CAF, Zuanon Netto J. Titulação complexométrica de lantanídeos e ítrio. Ecl Quim. 1983;8:29–32.
Becke AD. Density-functional thermochemistry. 3. The role of exact exchange. J Chem Phys. 1993;98:5648–52.
Lee C, Yang W, Parr RG. Development of the colle-salvetti correlation-energy formula into a functional of the electrondensity. Phys Rev B. 1988;37:785–9.
Treu-Filho O, Pinheiro JC, da Costa EB, Ferreira JEV, de Figueiredo AF, Kondo RT, de Lucca Neto VA, de Souza RA, Legendre AO, Mauro AE. Experimental and theoretical study of the compound [Pd(dmba)(NCO)(imz)]. J Mol Struct. 2007;829:195–201.
Treu Filho O, Pinheiro JC, Kondo RT, Marques RFC, Paiva-Santos CO, Davolos MR, Jafelicci M Jr. Gaussian basis sets to the theoretical study of the electronic structure of perovskite (LaMnO3). J Mol Struct (Theochem). 2003;631:93–9.
Treu Filho O, Pinheiro JC, Kondo RT. Designing Gaussian basis sets to the theoretical study of the piezoelectric effect of perovskite (BaTiO3). J Mol Struct. 2004;671:71–5.
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, et al. Gaussian 09, Revision A.02. Wallingford CT: Gaussian, Inc; 2009.
Goodson DZ, Sarpal SK, Wolfsberg M. Influence on isotope effect calculations of the method of obtaining force constants from vibrational data. J Phys Chem. 1982;86:659–63.
Schelegel HB. In: Bertran J, editor. New theoretical concepts for understanding organic reactions. Dordrecht: Kluwer Academic Publishers; 1989. p. 33–53.
Dennington R, Keith T, Millam J. GaussView, Version 5.0.8. Semichem Inc., Shawnee Mission KS; 2000–2008.
Silverstein RM, Webster FX, Kiemle DJ. Spectrometric identification of organic compounds. 7th ed. John Wiley & Sons: New York; 2005.
Lima LS, Caires FJ, Carvalho CT, Siqueira AB, Ionashiro M. Synthesis, characterization and thermal behaviour of solid-state compounds of light trivalent lanthanide succinates. Thermochim Acta. 2010;501:50–4.
Acknowledgements
The authors acknowledge Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for financial support. This research was supported by resources supplied by the Center for Scientific Computing (NCC/GridUNESP) of the Sao Paulo State University (UNESP), Instituto de Química de Araraquara, UNESP – Campus de Araraquara and CENAPAD-UNICAMP.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Caires, F.J., Lima, L.S., Gomes, D.J.C. et al. Thermal and spectroscopic studies of solid oxamate of light trivalent lanthanides. J Therm Anal Calorim 111, 349–355 (2013). https://doi.org/10.1007/s10973-012-2220-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-012-2220-y