Abstract
The curing behaviour of diglycidyl ether of bisphenol-A (DGEBA) was investigated by the dynamic differential scanning calorimetry using varying molar ratios of aromatic imide-amines and 4,4′-diaminodiphenylsulfone (DDS). The imide-amines were prepared by reacting 1 mole of naphthalene 1,4,5,8-tetracarboxylic dianhydride (N) and 4,4′-oxodiphthalic anhydride (O) with 2.5 moles of 4,4′-diaminodiphenyl ether (E) or 4,4′-diaminodiphenyl methane (M) or 4,4′-diaminodiphenylsulfone (S) and designated as NE/OE or NM/OM or NS/OS. The mixture of the imide-amines and DDS at ratio of 0:1, 0.25:0.75, 0.5:0.5, 0.75:0.25 and 1:0 were used to investigate the curing behaviour of DGEBA.
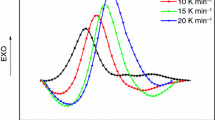
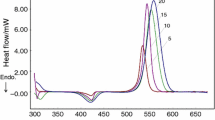
A single exotherm was observed on curing with mixture of imide-amines and DDS. This clearly shows that the two amines act as co-curing agents. Curing temperatures were higher with imide-amines having sulfone linkage irrespective of anhydride. Curing of DGEBA with mixture of imide-amines and or DDS resulted in a decrease in characteristic curing temperatures. The thermal stability of the isothermally cured resins was also evaluated using dynamic thermogravimetry in a nitrogen atmosphere. The char yield was higher in case of resins cured imide-amines based on N and E. The activation energy of decomposition and integral procedural decomposition temperature were also calculated from the TG data.
Similar content being viewed by others
References
H. Lee and K. Neville, Handbook of Epoxy Resins, McGraw Hill, New York 1972.
Van Nostrand Reinhold, Handbook of Composites, G. Lubin, Ed., New York 1982.
K. Iko, Y. Nakamura, M. Yamaguchi and N. Imamura, IEEE Elec. Insul. Mgg., 6 (1990) 25.
S. Hagiwara and S. Ichimura, Plastics, 39 (1990) 104.
P. Jain, V. Choudhary and I. K. Varma, J. Macromol. Sci. Polym. Rev., C42 (2002) 139.
W. K. Chin, M. D. Shau and J. C. Salamone, Polymeric Materials Encyclopedia (CRC Press), 3 (1996) 2210.
T. Ichino and Y. Haruda, J. Appl. Polym. Sci., 34 (1987) 1667.
S. Sasaki and Y. Hasuda, J. Polym. Sci. Part C Polym. Lett., 25 (1987) 377.
A. Serra, V. Cadiz and A. A. Mantecan, Makromol. Chem., 155 (1987) 93.
S.-H. Hwang and J.-C. Jung, J. Appl. Polym. Sci., 81 (2001) 279.
K. Adhinarayanan, S. Packirisamy, R. S. George, V. Lakshman Rao and R. Ramaswamy, J. Appl. Polym. Sci., 43 (1991) 783.
J. N. Hay, B. Wood Fine and M. Davies, High Performance Polym., 8 (1996) 35.
S. Ishikawa, M. Natakani, H. Fukuda and S. Yamamato, Japan Pat. 08253677 (1996).
K. Gaw, M. Kikei, M. Kakimoto and Y. Imai, React. Funct. Polym., 30 (1996) 85.
K. Gaw, M. Kikei, M. Kakimoto and Y. Imai, Polymer, 38 (1997) 4413.
W. R. Ashcroft, Chemistry and Technology of Epoxy Resin, B. Ellis, Ed., Chapmann and Hall, London 1993, Ch-2.
C. B. Bucknall, Toughened Plastics, John Wiley & Sons, New York 1977.
L. Becker, D. Lenoir, G. Matuschek and A. Kettrup, J. Anal. Appl. Pyrolysis, 60 (2001) 55.
P. Sharma, V. Choudhary and A. K. Narula, J. Appl. Polym. Sci., submitted.
D.W. Van Krevelen and P. J. Hoftyzer, Properties of Polymers, Elsevier, NewYork 1976, p. 529.
J. Y. Shieh and C. S. Wang, Polymer, 42 (2001) 7615.
C. D. Doyle, Anal. Chem., 33 (1961) 77.
E. S. Freeman and B. Caroll, J. Phys. Chem., 62 (1958) 394.
A. W. Coats and J. R. Redfern, Nature, 201 (1964) 68.
H. H. Horowitz and G. Metzger, Anal. Chem., 35 (1963) 1464.
L. Reich, J. Polym. Sci., B-3 (1965) 231.
S. R. Dharwadkar and M. D. Kharkhanawala, Thermal Analysis in Inorganic Materials and Physical Chemistry, Vol. 2, R. P. Schwenker and P. D. Garn, Eds, Academic Press, New York 1969.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sharma, P., Choudhary, V. & Narula, A.K. Curing and thermal behaviour of epoxy resin in the presence of a mixture of imide-amines. J Therm Anal Calorim 94, 805–815 (2008). https://doi.org/10.1007/s10973-007-8982-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-007-8982-y