Summary
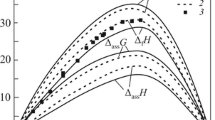
The melting temperatures and absolute values of melting enthalpies of lanthanide trichlorides decrease from LaCl3to TbCl3and then increase to LuCl3. The preceding decrease cannot be explained by the lattice energies of the trichlorides, since they increase continuously from the lanthanum to the lutetium compounds. However, it may be attributed to the structural features of the liquid state. The liquids near the melting points consist of clusters of complex units, which become larger with decreasing radii of the metal ions. To prove this assumption additional quantitative investigations are necessary.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Seifert, H. Melting points of lanthanide trichlorides. J Therm Anal Calorim 82, 575–580 (2005). https://doi.org/10.1007/s10973-005-0936-7
Issue Date:
DOI: https://doi.org/10.1007/s10973-005-0936-7