Summary
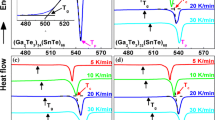
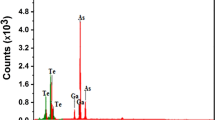
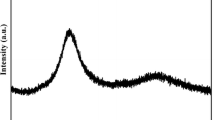
The structure identification of the quenched compositions GeSe<Subscript>2</Subscript>, GeSe<Subscript>4</Subscript>, GeSe<Subscript>2</Subscript>Tl and GeSe<Subscript>4</Subscript>Tl has been confirmed by energy dispersive X-ray analysis (EDX), X-ray diffraction (XRD) and differential thermal analysis (DTA). The differential thermal analysis curves of the compositions under investigation were recorded at the heating rates 5, 10, 20, 30 and 50°C min<Superscript>-1</Superscript>. Addition of thallium to GeSe<Subscript>2</Subscript> and GeSe<Subscript>4</Subscript> results in decreasing the glass transition temperature <Emphasis Type=”Italic”>T</Emphasis><Subscript>g</Subscript>, the crystallization temperature <Emphasis Type=”Italic”>T</Emphasis><Subscript>c</Subscript> and the melting temperature <Emphasis Type=”Italic”>T</Emphasis><Subscript>m</Subscript>. The effect of heating rates on the transition temperatures (<Emphasis Type=”Italic”>T</Emphasis><Subscript>g</Subscript>, <Emphasis Type=”Italic”>T</Emphasis><Subscript>c</Subscript> and <Emphasis Type=”Italic”>T</Emphasis><Subscript>m</Subscript>) was studied and the variation of the crystallization peak temperature <Emphasis Type=”Italic”>T</Emphasis><Subscript>p</Subscript> position was used to calculate the activation energy. The kinetic parameters of crystallization for the devitrification processes have been determined by three methods and discussed on the basis of Johnson-Mehl-Avrami model.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abdel-Aziz, M. Effect of thallium on the crystallization kinetics of the chalcogenide glasses GeSe<Subscript>2</Subscript> and GeSe<Subscript>4</Subscript>. J Therm Anal Calorim 79, 709–714 (2005). https://doi.org/10.1007/s10973-005-0600-2
Issue Date:
DOI: https://doi.org/10.1007/s10973-005-0600-2