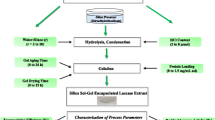
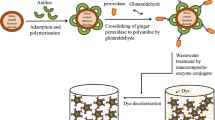
Abstract
Oxidoreductases are widely recognized for their capability to degrade phenolic pollutants and versatile. However, the lack of enzyme stability makes this technique unrealistic for industrial applications. In order to enhance their catalytic activity, stability and reusability, oxidoreductases namely laccases and peroxidases were entrapped in sol–gel silica and their catalytic activities were measured by an enzymatic assay using 2,6-dimethoxyphenol and guaiacol as substrates, respectively. The sol–gel silica matrices acted as a polymeric framework around the enzyme is a promising tool for improving enzyme stability. After entrapment, the catalytic activity and stability of sol–gel laccase and peroxidase toward pH, temperature and storage duration remarkably enhanced.

Highlights
-
The enzyme immobilization using novel sol-gel method is a promising method for improving enzyme catalytic activity and stability.
-
After entrapment, the catalytic activity and stability of the immobilized laccase and immobilized peroxidase towards pH, temperature and storage time are significantly improved.
-
Oxidoreductase entrapment in sol-gel silica-based matrix shows a bright future for the use of immobilized enzyme in vast industrial process particularly removal of dye from wastewater treatment.




Similar content being viewed by others
References
Georgieva S, Godjevargova T, Mita DG, Diano N, Menale C, Nicolucci C, Romano Carratelli C, Mita L, Golovinsky E (2010) Non-isothermal bioremediation of waters polluted by phenol and some of its derivatives by laccase covalently immobilized on polypropylene membranes. J Mol Catal B 66:210–218
Majeau J, Brar SK, Tyagi RD (2010) Laccases for removal of recalcitrant and emerging pollutants. Bioresour Technol 101:2331–2350
Qiu H, Lu L, Huang X, Zhang Z, Qu Y (2010) Immobilization of horseradish peroxidase on nanoporous copper and its potential applications. Bioresour Technol 101:415–9420
Li X, Li S, Liang X, Julian D, McClements, Liu X, Liu F (2020) Applications of oxidases in modification of food molecules and colloidal systems: laccase, peroxidase and tyrosinase. Trends Food Sci Technol 103:78–93
Debnath R, Saha T (2020) An insight into the production strategies and applications of the ligninolytic enzyme laccase from bacteria and fungi. Biocatal Agric Biotechnol 26:101645
Peralta-Zamora P, Pereira CM, Tiburtius ERL, Moraes SG, Rosa MA, Minussi RC (2003) Decolorization of reactive dyes by immobilized laccase. Appl Catal B 42:131–144
Mohidem NA, Mat HB (2012) Catalytic activity and stability of laccase entrapped in sol–gel silica with additives. J Sol–Gel Sci Technol 61:96–103
Mohidem (2018) Biodegradation of alkanolamines in batch and packed-bed reactors using free laccase and sol–gel laccase. Ph.D thesis. Universiti Teknologi Malaysia, Skudai, Malaysia
Wahab RA, Elias N, Abdullah F, Ghoshal SK (2020) On the taught new tricks of enzymes immobilization: an all-inclusive overview. React Funct Polym 152:104613
Fopase R, Paramasivam S, Kale P, Paramasivan B (2020) Strategies, challenges and opportunities of enzyme immobilization on porous silicon for biosensing applications. J Environ Chem Eng 8(5):104266
Salis A, Pisano M, Monduzzi M, Solinas V, Sanjust E (2009) Laccase from Pleurotus sajor-caju on functionalised SBA-15 mesoporous silica: Immobilization and use for the oxidation of phenolic compounds. J Mol Catal B Enzym 58:175–180
Rekuc A, Bryjak J, Szymansja K, Jarzebski AB (2010) Very stable silica–gel-bound laccase biocatalysts for the selective oxidation in continuous systems. Bioresour Technol 101:2076–2083
Tian F, Xu B, Zhu L, Zhu G (2001) Hydrogen peroxide biosensor with enzyme entrapped within electrodeposited polypyrrole based on mediated sol–gel derived composite carbon electrode. Anal Chim Acta 443(1):9–16
Yamanaka SA, Nishida F, Ellerby LM, Nishida CR, Dunn B, Valentine JS, Zink J (1992) Enzymatic activity of glucose oxidase encapsulated in transparent glass by the sol–gel method. Chem Mater 4(3):495–497
Matijosyte I, Arends IWCE, de Vries S, Sheldon RA (2010) Preparation and use of cross-linked enzyme aggregates (CLEAs) of laccase. J Mol Catal B 62:142–148
Michizoe J, Goto M, Furusaki S (2001) Catalytic activity of laccase hosted in reversed micelles. J Biosci Bioeng 92:67–71
Vasquez-Duhalt R, Tinoco R, D’Antonio P, Topoleski LDT, Payne GF (2001) Enzyme conjugation to the polysaccharide chitosan: smart biocatalysts and biocatalytic hydrogels. Bioconjugate Chem 12:301–306
Miao Y, Tan SN (2001) Amperometric hydrogen peroxide biosensor with silica sol–gel/chitosan film as immobilization matrix. Anal Chim Acta 437(1):87–93
Zawisza I, Rogalski J, Opallo M (2006) Electrocatalytic reduction of dioxygen by redox mediator and laccase immobilized in silicate thin film. J Electroanal Chem 588:244–252
Ellerby LM, Nishida CR, Nishida F, Yamanaka S, Dunn B, Valentine JS, Zink JI (1992) Encapsulation of proteins in transparent porous silicate glasses prepared by the sol–gel method. Science 255(5048):1113–1115
Gill I, Ballesteros A (1998) Encapsulation of biologicals within silicate, siloxane, and hybrid sol–gel polymers: an efficient and generic approach. J Am Chem Soc 120:8587–8598
Glad M, Norrlo¨w O, Sellergren B, Siegbahn N, Mosnach K (1985) Use of silane monomers for molecular imprinting and enzyme entrapment in polysiloxane-coated porous silica. J Chromatogr 347:11–23
Matsune H, Jogasaki H, Date M, Takenaka S, Kishida M (2006) One-pot synthesis and characterization of laccase-entrapped magnetic nanobeads. Chem Lett 35:1356–1357
Crecchio C, Ruggiero P, Pizzigallo MDR (1995) Polyphenoloxidases immobilized in organic gels: Properties and applications in the detoxification of aromatic compounds. Biotechnol Bioeng 48:585–591
Okazaki S, Goto M, Wariishi H, Tanaka H, Furusaki S (2000) Characterization and catalytic property of surfactant-laccase complex in organic media. Biotechnol Prog 16:583–588
Khani Z, Jolivalt C, Cretin M, Tingry S, Innocent C (2006) Alginate/carbon composite beads for laccase and glucose oxidase encapsulation: application in biofuel cell technology. Biotechnol Lett 28:1779–1786
Shin MY, Park JY, Park K, Song SH, Yoo YJ (2007) Novel sol–gel immobilization of horseradish peroxidase employing a detergentless micro-emulsion system. Biotechnol Bioprocess Eng 12:640–645
Shakeri M, Shoda M (2010) Efficient decolorization of an anthraquinone dye by recombinant dye-decolorizing peroxidase (rDyP) immobilized in silica-based mesocellular foam. J Mol Catal B 62:277–281
Mat HB, Mohidem NA (2012) The catalytic activity enhancement and biodegradation potential of free laccase and novel sol–gel laccase in non-conventional solvents. Bioresour Technol 114:472–477
Mohidem NA, Mat HB (2009) The catalytic activity of laccase immobilized in sol–gel silica. J Appl Sci 9:3141–3145
Vera-Avila LE, Morales-Zamudio E, Garcia-Camacho MK (2004) Activity and reusability of sol–gel encapsulated α-amylase and catalase. Performance in flow-through systems. Sol–Gel Sci Technol 30:197–204
Leoniwicz A, Sarkar JM, Bollag JM (1988) Improvement fungal laccase. J Chem Biotechnol 29:129–135
Luckarift HR, Ku BS, Dordick JS, Spain JS (2007) Silica-immobilized enzymes for multi-step synthesis in microfluidic devices. Biotechnol Bioeng 98:701–705
Guisan JM (2006) Immobilization of enzymes and cells. Humana Press, New Jersey
Moniruzzaman M, Nakashima K, Kamiya N, Goto M (2010) Recent advances of enzymatic reactions in ionic liquids. Biochem Eng J 48:295–314
Bilal M, Asgher M (2016) Enhanced catalytic potentiality of Ganoderma lucidum IBL-05 manganese peroxidase immobilized on sol–gel matrix. J Mol Catal B: Enzym 128:82–93
Nawawi NN, Hashim Z, Rahman RA, Murad AMA, Bakar FDA, Illias RM (2020) Entrapment of porous cross-linked enzyme aggregates of maltogenicamylase from Bacillus lehensisG1 into calcium alginate formaltooligosaccharides synthesis. Int J Biol Macromol 150:80–89
Das S, Berke-Schlessel D, Ji H-F, McDonough J, Wei Y (2011) Enzymatic hydrolysis of biomass with recyclable use of cellobiase enzymeimmobilized in sol–gel routed mesoporous silica. J Mol Catal B: Enzym 70:49–54
Braun S, Rappoport S, Zusman R, Avnir D, Ottolenghi M (2007) Biochemically active sol–gel glasses: The trapping of enzymes. Mater Lett 61:2843–2846
Yi Y, Neufeld R, Kermasha S (2007) Controlling sol–gel properties enhancing entrapped membrane protein activity through doping additives. Sol–Gel Sci Technol 43:161–170
Fernandez-Lafuente R (2009) Stabilization of multimeric enzymes: Strategies to prevent subunit dissociation. Enzyme Microb Technol 45:405–418
Bhatia RB, Brinker CJ (2000) Aqueous sol–gel process for protein encapsulation. Chem Mater 12:2434–2441
Makas YG, Kalkan NA, Aksoy S, Altinok H, Hasirci N (2010) Immobilization of laccase in κ-carrageenan based semi-interpenetrating polymer networks. J Biotechnol 148:216–220
Arica MY, Bayramoglu G, Bicak N (2004) Characterization of tyrosinase immobilized onto spacer-arm attached glycidyl methacrylate-based reactive microbeads. Process Biochem 39:2007–2017
Chen JP, Lin WS (2003) Sol–gel powders and supported sol–gel polymers for immobilization of lipase in ester synthesis. Enzyme Microb Technol 32:801–811
Creighton TE (1996) Proteins structure and molecular properties. WH Freeman and Company, New York, NY
Shuler ML, Kargi F (2005) Bioprocess engineering. Prentice Hall, Upper Saddle River, New Jersey
Mohidem NA, Mansor AF, Wan Mohd Zawawi WNI, Othman NS, Mat HB (2016) Effect of synthesis conditions on physical properties, catalytic activity and stability of sol–gel laccase. J Sol–Gel Sci Technol 80:587–597
Ahmad Shamsuri NA, Mohidem NA, Mansor AF, Mat HB (2012) Biodegradation of dye using free laccase and sol–gel laccase. J Teknol 59:39–42
Aegerter MA, Mennig M (2004) Sol–gel technologies for glass producers and users. Springer, Boston MA
Acknowledgements
The authors would like to acknowledge the financial support from the Ministry of Higher Education (MOHE), Malaysia under the Fundamental Research Grant Scheme (FRGS Project No. 78213).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohidem, N.A., Bin Mat, H., Mohamad, M. et al. Strategy to enhance catalytic activity and stability of sol–gel oxidoreductases. J Sol-Gel Sci Technol 98, 462–469 (2021). https://doi.org/10.1007/s10971-021-05522-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-021-05522-0