Abstract
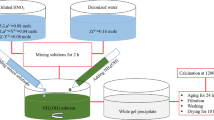
Aqueous sol–gel technique is reported for synthesizing nanosize, rod shaped lanthanum phosphate particles starting from lanthanum chloride, which is suitable for variety of applications such as machinable ceramics, thermal barrier coatings and luminescent materials. The phosphate particles are having rod like morphology having an overall size in the range 25–100 nm and with an average aspect ratio 4. The morphology is retained even after calcination at 800 °C. The average crystallite size for the as prepared particles was calculated to be 8 nm using Scherrer equation applied on X-ray diffraction (XRD) data. The particles obtained were characterized using photon correlation spectroscopy (PCS), FT-IR spectroscopy, XRD and Electron Microscopy techniques (TEM and SEM). The phase stability was found using thermo gravimetric analysis. The surface wettability monitored using tensiometer through dynamic contact angle method indicated that lanthanum phosphate surface possesses considerable hydrophobic character.








Similar content being viewed by others
References
Kashiwagi D, Takai A, Takubo T, Yamada H, Inoue T, Nagaoka K, Takita Y (2009) J Colloid Interface Sci 332:136–144
Onoda H, Nariai H, Moriwaki A, Maki H, Motooka I (2002) J Mater Chem 12:1754–1760
Cao XQ, Vassen R, Stoever D (2004) J Eur Ceram Soc 24:1–10
Davis JB, Marshall DB, Housley RM, Morgan PED (1998) J Am Ceram Soc 81:2169–2175
Schuetz P, Caruso F (2002) Chem Mater 14:4509–4516
Meiser F, Cortez C, Caruso F (2004) Angew Chem Int Ed 43:5954–5957
Zhang F, Wong SS (2010) ACS Nano 4:99–112
Hikichi Y, Nomura T (1987) J Am Ceram Soc 70:C252–C253
Narasimha B, Choudhary RNP, Rao KV (1988) J Mater Sci 23:1416–1418
Hikichi Y, Ota T, Hattori T (1997) Miner J 19:123–130
Sambasivan S (2000) US pat 6036372
Kitamura N, Amezawa K, Tomii Y, Yamamoto N (2003) Solid State Ionics 162:161–165
Takita Y, Sano K, Muraya T, Nishiguchi H, Kawata N, Ito M, Akbay T, Ishihara T (1998) Appl Catal A 170:23–31
Li L, Jiang WG, Pan HH, Xu XR, Tang YX, Ming JZ, Xu ZD, Tang RK (2007) J Phys Chem C 111:4111–4115
Chai ZL, Gao L, Wang C, Zhang HJ, Zheng RK, Webley PA, Wang HT (2009) New J Chem 33:1657–1662
Fang YP, Xu AW, Song RQ, Zhang HX, You LP, Yu JC, Liu HQ (2003) J Am Chem Soc 125:16025–16034
Wang RG, Pan W, Chen J, Fang MH, Cao ZZ, Luo YM (2003) Mater Chem Phys 79:30–36
Buissette V, Moreau M, Gacoin T, Boilot JP, Chane-Ching JY, Le Mercier T (2004) Chem Mater 16:3767–3773
Buehler G, Feldmann C (2006) Angew Chem Int Ed 45:4864–4867
Brown SS, Im HJ, Rondinone AJ, Dai S (2005) J Colloid Interface Sci 292:127–132
Rajesh K, Mukundan P, Pillai PK, Nair VR, Warrier KGK (2004) Chem Mater 16:2700–2705
Lucas S, Champion E, Bregiroux D, Bernache-Assollant D, Audubert F (2004) J Solid State Chem 177:1302–1311
Onoda H, Nariai H, Maki H, Motooka I (2002) Mater Chem Phys 73:19–23
Chen P, Mah TI (1997) J Mater Sci 32:3863–3867
Rajesh K, Shajesh P, Seidel O, Mukundan P, Warrier KGK (2007) Adv Func Mater 17:1682–1690
Schindler DW (1974) Science 4139:897–899
Kijowska R, Colewka E, Duszak B (2003) J Mater Sci 38:223–228
Ross SD, Hezel A (1966) Spectrochim Acta 22:1949–1961
Acknowledgments
Authors acknowledge Indian Rare Earths Limited, Mumbai for funding and acknowledgements are due to Dr. V. Mangalaraga, University of Concepcion, Chile for TEM studies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sankar, S., Warrier, K.G. Aqueous sol–gel synthesis of lanthanum phosphate nano rods starting from lanthanum chloride precursor. J Sol-Gel Sci Technol 58, 195–200 (2011). https://doi.org/10.1007/s10971-010-2377-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-010-2377-4