Abstract
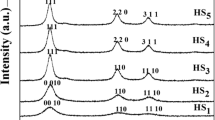
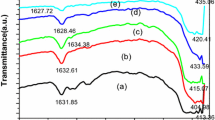
The simultaneous formation of nanometer sized zinc oxide (ZnO), and acetate zinc hydroxide double salt (Zn-HDS) is described. These phases, obtained using the sol-gel synthesis route based on zinc acetate salt in alcoholic media, were identified by direct characterization of the reaction products in solution using complementary techniques: nephelometry, in situ Small-Angle X-ray Scattering (SAXS), UV-Vis spectroscopy and Extended X-ray Absorption Fine Structures (EXAFS). In particular, the hydrolytic pathway of ethanolic zinc acetate precursor solutions promoted by addition of water with the molar ratio N=[H2O]/[Zn2+] = 0.05 was investigated in this paper. The aim was to understand the formation mechanism of ZnO colloidal suspension and to reveal the factors responsible for the formation of Zn-HDS in the final precipitates. The growth mechanism of ZnO nanoparticles is based on primary particle (radius ≈ 1.5 nm) rotation inside the primary aggregate (radius <3.5 nm) giving rise to an epitaxial attachment of particles and then subsequent coalescence. The growth of second ZnO aggregates is not associated with the Otswald ripening, and could be associated with changes in equilibrium between solute species induced by the superficial etching of Zn-HDS particles at the advanced stage of kinetic.
Similar content being viewed by others
References
Wang J, Cao J, Fang B, Lu P, Deng S, Wang H (2005) Mater. Lett. 59:1405
Cohen ML (2000) Annu. Rev. Mater. Sci. 30:1
Brus LE (1996) J Phys Chem 90:2555
Clarke DR (1999) J Am Ceram Soc 82:485
Paneva R, Gotchev D (1999) Sensor Actuator A: Phys. 72:79
Granqvist CG (2003) Adv. Mater. 15:1789
Ni J, Yan H, Wang A, Yang Y, Stern CL, Metz AW, Jin S, Wang L, Marks TJ, Ireland JR, Kannewurf CR (2005) J Am Ceram Soc 127:5613
Spanhel L, Anderson MA (1991) J Am Chem Soc 113:2826
Meulenkamp EA (1998) J Phys Chem B 102:5566;5566; ibdem 7764
Wong EM, Bonevich JE, Searson PC (1998) J Phys Chem B 102:7770
Hu Z, Oskam G, Searson PC (2003) J Colloid Interface Sc 263:454
Hu Z, Oskam G, Lee Penn R, Pesika N, Searson PC (2003) J Phys Chem B 107:3124
Tokumoto M, Briois V, Santilli CV, Pulcinelli SH (2003) J Sol-Gel Sc Tech 26:547
Livage J, Henry M, Sanchez C (1988) Prog Solid State Chem 18:259
Tokumoto MS, Pulcinelli SH, Santilli CV, Briois V (2003) J Phys Chem B 107:568
Liang C, Shimizu Y, Masuda M, Sasaki T, Koshizaki N (2004) Chem Mater 16:963
Meyn M, Beneke K, Lagaly G (1993) Inorg Chem 32:1209
Morioka H, Tagaya H, Karasu M, Kadokawa J, Chiba K (1999) Inorg Chem 38:4211
Poul L, Jouini N, Fievet F (2000) Chem Mater 12:3123
Villain F, Briois V, Castro I, Helary C, Verdaguer M (1993) Anal Chem 65:2545
Briois V, Belin S, Villain F, Bouamrane F, Lucas H, Lescouëzec R, Julve M, Verdarguer M, Tokumoto MS, Santilli CV, Pulcinelli SH, Carrier X, Krafft JM, Jubin C, Che M (2005) Physica Scripta T115:38
Briois V, Lützenkircken-Hecht D, Villain F, Fonda E, Belin S, Griesebock B, Frahm R (2005) J Phys Chem A 109:320
Brus LE (1992) Nanostructured Materials 1:71
Hale PS, Maddox LM, Shapter JG, Voelcker NH, Ford MJ, Waclawik ZR (2005) J Chem Educ 82:775
Nedelijkovic JM, Patel R, Kaufman P, Joyce-Pruden C, O’Leary N (1993) J Chem Educ 70:342
Nielsen JA (1993) Diffraction, refraction and absorption of X-ray and neutrons: A comparative exposition. (Springer Verlag & Les Editions de Physique, Paris, France, 1993), Chap. 1
Porod G, Kratky O, Glatter O (eds) (1982) Small angle X-ray scattering. Academic Press, London, pp 17–51
Guinier A (1964) Théorie et Technique de la Radiocristallographie. Dunod, Paris, France
Svergun DI, Semeenyuk AV (1993) In: Svergun DI, Semeenyuk AV, Feigin LA, (ed) Program package GNOM: Small angle scattering data processing by means of regularization technique, Version E 4.2., Acta Cryst. A 44, 244 (1988)
Baudelet F, Dartyge E, Fontaine A, Brouder C, Krill G, Kappler JP, Piecuch M (1991) Phys. Rev. B 43:5857
Michalowicz A (1991) EXAFS pour le Mac, Logiciels pour la Chimie. Société Française de Chimie, Paris, p 102
Lorentz C, Emmerling A, Fricke J, Schmidt T, Hilgendorff M, Spahnel L (1998) J Non Cryst Solids 238:1
Briois V, Giorgetti C, Baudelet F, Flank AM, Tokumoto MS, Pulcinelli SH, Santilli CV (2004) In: Faulques E, Perry D, Yeremenko A, (ed) Nato Sciences series; Nato ARW “Spectroscopy of Emerging Materials”, Kluwer Academic Publishers, pp 15–30
Wood A, Giersig M, Hilgendorff M, Vilas-Campos A, Liz-Marzan LM, Mulvaney P (2003) Aust J Chem 56:1051
Hilgendorff M, Spahnel L, Rothenhausler C, Müller G (1998) J Electrochem Soc 145:3632
Tokumoto MS, Pulcinelli SH, Santilli CV (1999) Adv Sci Technol 14:73
Haase M, Weller H, Henglein A (1988) J Phys Chem 92:482
Lifshitz LM, Slyozov VV (1961) J Phys Chem Solids 19: 35 C. Wagner (1961) Z. Elektrochem. 65:581
Hu Z, Herrera Santos JF, Oskam G, Searson PC (2005) J Colloid Interface Sci 288:313
Meakin P, Family F (1989) J Phys A Math Gen 22:L225
Penn RL, Banfield JF (1998) Science 281: 969 Banfield JF, Welch SA, Zhang H, Ebert TT, Penn RL, (2000), Science 289:751
Leite ER, Giraldi TR, Pontes FM, Longo E, Beltran A Andrés J (2003) Appl Phys Lett 83:1566
Moldovan D, Yamakov V, Wolf D, Phillpot SR (2002) Phys. Rev. Lett. 89:206101-1
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Briois, V., Giorgetti, C., Dartyge, E. et al. In situ and simultaneous nanostructural and spectroscopic studies of ZnO nanoparticle and Zn-HDS formations from hydrolysis of ethanolic zinc acetate solutions induced by water. J Sol-Gel Sci Technol 39, 25–36 (2006). https://doi.org/10.1007/s10971-006-6973-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-006-6973-2