Abstract
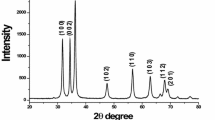
A systematic study is presented on the synthesis of ZnS nanoparticles by using simple chemical precipitation method without using any capping agent. The ZnS nanoparticles have been synthesized using precursor solution with different Zn2+:S2− ratio. The as synthesized nanoparticles have been characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), atomic force microscopy (AFM), UV–Vis absorption spectroscopy and impedance spectroscopy. The XRD patterns reveal the formation of ZnS nanoparticles with wurtzite hexagonal phase which changes to sphalarite phase for higher concentration of S2− in the precursor solution. XRD, SEM and AFM studies have shown enhancement in particle size on increasing sulphur content. SEM and AFM images depict formation of deformed spherical nanoparticles with particle size ranging from 10 to 50 nm. Optical absorption spectra of synthesized material show a red shift in the optical absorption on increase of S2− ion concentration. Variation in electrical conductivity obtained from impedance measurements at different temperatures has been suitably correlated to Davis–Mott model.






Similar content being viewed by others
References
X. wang, H. Huang, B. Liang, Z. Liu, D. Chen, G. Shen, ZnS nanostructures: synthesis, properties and applications. Crit. Rev. Solid State Mater. Sci. 38(1), 57–90 (2013)
K.S. Rathore, D. Patidar, Y. Janu, N.S. Saxena, K. Sharma, T.P. Sharma, Structural and optical characterization of chemically synthesized ZnS nanoparticles. Chalcognide Lett. 5, 105–110 (2008)
W. Park, J.S. King, C.W. Neff, C. Liddell, C.J. Summers, ZnS based photonic crystals. Phys. Status Solidi 229, 949–960 (2002)
B. Gillbert, B.H. Frazer, H. Zhang, F. Huang, J.F. Banfield, D. Haskel, J.C. Lang, X-ray absorption spectroscopy of the cubic and hexagonal poly type ZnS”. Phys. Rev. B 66, 245205 (2002)
S.B. Qadri, E.F. Sketton, “The effect of particle size on the structural transition in Zinc Sulphide”. J. Appl. Phy 89, 115–119 (2001)
S.D. Esgreniers, L. Beaulieu, I. Lepage, “Pressure-induced structural changes in ZnS”. Phys. Rev. B 61, 8726–8733 (2000)
J.F. Suyver, S.F. Weister, J.J. Kelly, A. Meijerink, Synthesis and photoluminescence of nanocrystallite ZnS. Nano Lett. 1, 429 (2001)
X. Fang, T. Zhai, U. K. Gautam, L. Li, L. Wu, Y. Bando, D. Golberg, ZnS nanostructures: from synthesis to applications. Prog. Mater. Sci. 56,175–287 (2011)
Y. Ding, X.D. Wang, L.Z. Wang, Phase controlled synthesis of ZnS nanobelts: zinc blende vs wurtzite. Chem. Phys. Lett. 398, 32–36 (2004)
A. Marusamy, S. Agilan, M. Kumarsamy, C.K. Senthil kumaran, M. Sarvanakumar, The influence of the Zn/S source concentration ratio on its structural, optical and magnetic properties of ZnS quantum dots. Chalcogenide Lett. 10, 179–187 (2003)
H.K. Sharma, P.K. Shukla, L.S. Agrawal. Structural and electrical studies on ZnS nanoparticles prepared without using capping agent. J. Optoelectron. Adv. Mater. 17, 608–613 (2015)
C.C. Yang, S. Li, Size, dimensionality and constituent stoichiometry dependence of bandgap energies in semiconductor quantum dots and wires. Phys. Chem. C 112, 2851 (2008)
P.C. Patel, N. Srivastav, C.P. Srivastav, Synthesis of wurtzite ZnS nanocrystallites at low temperature. J. Mater. Sci. 13, 1367 (2013)
F.A. La Porta, J. Andre´s, M.S. Li, J.R. Sambrano, J.A. Varelaa, E. Longoa, Zinc blende versus wurtzite ZnS nanoparticles: control of the phase and optical properties by tetrabutylammonium hydroxide. Phys. Chem. Chem. Phys. 16, 20127 (2014)
B.D. Culty, Elements of X-ray Diffraction, 2nd edn. (Addision Wesley company, USA, 1956), p. 102
G. Nabiyouni, R. Sahraei, M. Toghiany, M. Majles, K. Hedayati, Preparation and characterization of nanostructured ZnS thin film grown on glass and N-type Si substrate using a new chemical bath deposition. Adv. Mater. Sci. 27, 52–57 (2011)
M.A. Martinz, C. Guillen, J. Herrero, Cadmium sulphide growth investigations on different SnO2 substrates. Appl. Surf. Sci. 140, 182–189 (1999)
D.A. Skoog, F. Holler, C. James, R. Stanley, Principles of Instrumental Analysis, 6th edn. (Thomson Brooks, Cole, Belmont, CA, 2007), pp. 169–173
P. Nagels, Topics in Applied Physics, Amorphous Semiconductors, ed. by M.H. Brodsky (Springer, New York, 1979)
N.F. Mott, E.A. Davis, Electronic Processes in Non-Crystalline Materials, (Clareendon, Oxford University Press, New York, 1979)
M.A. Majeed Khan, S. Kumar, M. Alhoshan, A.S. Aidwayyan, Spray pyrolysed Cu2ZnSnS4 absorbing layer: a potential candidate for photovoltaic applications. Opt. Laser Technol. 49, 196–201 (2013)
Y. Tian, H. Lu, J. Tian, C. Li, C. Hui, X. Shi, Y. Huang, C. Shen, H. Gao, Influence of Si Co-doping on electrical transport properties of magnesium doped boron nanoswords. Appl. Phys. Lett. 100, 103112 (2012)
W.J. Lu, B.C. Zhao, R. Ang, W.H. Song, P.Y. Sun Studies of electrical and thermal transport properties of the electron-doped manganite Sr0.9Ce0.1MnO3. Phys. B 367, 243–248 (2005)
O.S. Panwar, I. Rawal, R.K. Tripathi, A.K. Srivastava, M. Kumar, Structural, nanomechanical and variable range hopping conduction behavior of nanocrystalline carbon thin films deposited by the ambient environment assisted filtered cathodic jet carbon arc technique. J. Alloys Compd. 628, 135–145 (2015)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, H.K., Shukla, P.K. & Agrawal, S.L. Effect of sulphur concentration on the structural and electronic properties of ZnS nanoparticles synthesized using chemical precipitation method. J Mater Sci: Mater Electron 28, 6226–6232 (2017). https://doi.org/10.1007/s10854-016-6302-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-016-6302-7