Abstract
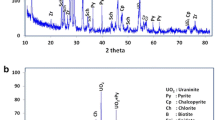
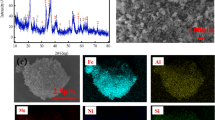
A well-characterized low-grade fluoritized uranium samples from new occurrence in Gabal El-Missikat prospect, Eastern Desert, Egypt was subjected to sulfuric acid leaching. The effects of leaching parameters on uranium dissolution mechanism were investigated. The shrinking core model was used to model leaching reactions. The kinetics equations indicates that the reactions appear to be controlled by layer diffusion process. The activation energy for uranium dissolution was evaluated. Low activation energy value (2.54 kJ mol−1) confirm the diffusion layer mechanism. The presence of fluoride ions in the solution increases the dissolution of uranium. The optimum process operating parameters were: sulfuric acid concentration: 1.5 M, solid–liquid ratio: 1:3, contact time 8 h; agitation speed rate 200 rpm; and ore particle size − 75 µm at temperature 60 °C, in the absence of an external oxidant. Under these experimental conditions, the extraction efficiency of uranium was about 91%.















Similar content being viewed by others
References
Budnitz RJ, Rogner HH, Shihab-Eldin A (2018) Expansion of nuclear power technology to new countries—SMRs, safety culture issues, and the need for an improved international safety regime. Energy Policy 119:535–544. https://doi.org/10.1016/j.enpol.2018.04.051
Ammar AA (1973) Application of aerial radiometry to the geology of Wadi El Gidami area, Eastern Desert Ph.D. Thesis, Geology department, Faculty of science, Cairo University, Cairo, Egypt
Abdallah SM (2004) Geological and mineralogical studies on some surface and subsurface sections from El-Missikat and El-Erediya uranium occurences, Central Eastern Desert, Egypt, Ph.D. Thesis, Geology department, Faculty of science, Ain Shams University, Geiza, Egypt
Abu-Dief A (1985) Geology of Uranium Mineralization in El-Missikat Area, Qena-Safaga road. Eastern Desert, Egypt, M.Sc. Thesis, Faculty of science, Al Azhar University, Cairo, Egypt
Abu-Dief A (1992) The relation between the uranium mineralization and tectonics in some Pan-African granites, West of Safaga, Eastern Desert, Egypt, Ph.D. Thesis, Faculty of science, Assiut University, Cairo, Egypt
Ahmed NA (1991) Comparative study of the accessory heavy minerals in some radioactive rocks of gabal El-missikat and gabal el erediya, eastern desert. Egypt and their alluvial deposits, M.Sc. Thesis, Geology department, Faculty of science, Cairo University, Cairo, Egypt
Ammar FA, Omar SAM, El Sawe ESHY (2016) Genetic affiliation of gold and uranium mineralization in el-missikat granite, Central Eastern Desert, Egypt. Nucl Sci Sci J 5:33–47
Attawiya MY (1983) Geochemistry and genesis of the uranium mineralization of Gebel El Misikat, Egypt. Ann Geol Surv Egypt 13:67–74
Bakhit FS, Kassas IAE (1989) Distribution and orientation of radioactive veins in the El Erediya-El Missikat area, Central Eastern Desert, Egypt. Int J Remote Sens 10(3):565–581. https://doi.org/10.1080/01431168908903893
El Tahir MA (1985) Radioactivity and mineralization of granitic rocks of El Erediya occurrence and comparison to El Missikat Rei El-Garra occurrence, Eastern Desert, Egypt, Ph.D. Thesis, Geology department, Faculty of science, Al-Azhar University, Cairo, Egypt
El-Sherif A (2013) Mineralogical characterization of the alteration facies at Gabal El-Missikat Area, Central Eastern Desert, Egypt. Nucl Sci Sci J 2:1–21
Ibrahim TMM (2002) Geologic and radioactive studies of the basement-sedimentary contact in the area West Gabal El Missikat, Eastern Desert, Egypt, Ph.D. Thesis, Geology department, Faculty of science, Mansoura University, Cairo, Egypt
Mohamed NA (1995) Distribution and extraction of uranium and some trace elements from the mineralized zones of El-Missikat—El-Erediya area, Eastern Desert, Egypt, Ph.D. Thesis, Geology department, Faculty of science, Cairo University, Cairo, Egypt
Amer TE-S, Ibrahim TM, Omar SA (2005) Micro-probe studies and some rare metal recovery from El-Missikat mineralized shear zone Eastern Desert, Egypt. Paper presented at the The fourth international conference on the geology of Africa, Assiut, Egypt, Nov 2005
Awad LSED (2017) Studies on the leaching and recovery of uranium and some associated economic elements from the mineralized shear zone of Jabal El Missikat, Eastern Desert, Egypt, M.Sc. Thesis, Chemistry department, Faculty of science, Benha University, Egypt
Haggag ESAESA (2014) Recovery of uranium and other economic elements from El-Mssikat pluton, Central Eastern Desert, Egypt, M.Sc. Thesis, Chemistry department, Faculty of science, Benha University, Egypt
Mohammed H, Sadeek S, Mahmoud AR, Zaky D (2016) Comparison of AAS, EDXRF, ICP-MS and INAA performance for determination of selected heavy metals in HFO ashes. Microchem J 128:1–6. https://doi.org/10.1016/j.microc.2016.04.002
Mohammed H, Sadeek S, Mahmoud AR (2016) Accurate determination of uranium and thorium in Egyptian oil ashes. Microchem J 124:699–702. https://doi.org/10.1016/j.microc.2015.10.034
Shapiro L (1975) Rapid analysis of silicate, carbonate, and phosphate rocks-revised edition. US Geological Survey
Xu W-D, Sun R, Zhu X-P, Liu P-H (2010) Determination of micro-amount of uranium in ore by microtitration with reduction of ammonium vanadate by titanous chloride. Rock Miner Anal 3:325–327
Bodkin J (1977) Determination of fluorine in silicates by use of an ion-selective electrode following fusion with lithium metaborate. Analyst 102(1215):409–413. https://doi.org/10.1039/AN9770200409
Levenspiel O (1992) Chemical reaction engineering. Wiley
Madakkaruppan V, Pius A, Sreenivas T, Shiv Kumar K (2015) Leaching kinetics of uranium from a quartz–chlorite–biotite rich low-grade Indian ore. J Radioanal Nucl Chem 303(3):1793–1801. https://doi.org/10.1007/s10967-014-3760-3
Gogoleva EM (2012) The leaching kinetics of brannerite ore in sulfate solutions with iron(III). J Radioanal Nucl Chem 293(1):185–191. https://doi.org/10.1007/s10967-012-1643-z
Merritt RC (1971) The extractive metallurgy of uranium. vol Colorado School of Mines Research Institute
Shvareva TY, Mazeina L, Gorman-Lewis D, Burns PC, Zymanowski JESS, Fein JB, Navrotsky A (2011) Thermodynamic characterization of boltwoodite and uranophane: enthalpy of formation and aqueous solubility. Geochim et Cosmochim Acta 75:5269–5282. https://doi.org/10.1016/j.gca.2011.06.041
Guillaumont R, Mompean FJ (2003) Update on the chemical thermodynamics of uranium, neptunium, plutonium, americium and technetium, vol 5. Elsevier, Amsterdam
Ram R, Charalambous F, McMaster S, Pownceby M, Tardio J, Bhargava S (2013) An investigation on the dissolution of natural uraninite ores. Miner Eng 50:83–92. https://doi.org/10.1016/j.mineng.2013.06.013
Ibrahim TMM, El-Hussaini OM (2007) Production of anhydrite–gypsum and recovery of rare earths as a by-product. Hydrometallurgy 87(1):11–17. https://doi.org/10.1016/j.hydromet.2006.11.017
Katz JJ, Rabinowitch E (1951) The chemistry of uranium, part I; the elements, its binary and related componds. McGraw-Hill, New York
Crundwell FK (2013) The dissolution and leaching of minerals: mechanisms, myths and misunderstandings. Hydrometallurgy 139:132–148. https://doi.org/10.1016/j.hydromet.2013.08.003
McMaster SA (2016) Investigations into the synthesis, characterisation and uranium extraction of the pyrochlore mineral betafite, Ph.D thesis, School of Applied Sciences, College of Science, Engineering and Health, RMIT University
McMaster SA, Ram R, Pownceby MI, Tardio J, Bhargava S (2015) Characterisation and leaching studies on the uranium mineral betafite [(U, Ca)2(Nb, Ti, Ta)2O7]. Miner Eng 81:58–70. https://doi.org/10.1016/j.mineng.2015.07.007
Ram R (2013) An investigation on the dissolution of synthetic and natural uraninite, Ph.D. thesis, School of Applied Sciences, College of Science, Engineering and Health, RMIT University
Acknowledgements
Authors are grateful to Professor Ehab Korany for assisting in mineralogical analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohammed, H.S., Abdel-Monem, Y.K., El-Feky, M.G. et al. Leaching of El-Missikat low-grade fluoritized uranium ore by sulfuric acid: mechanism and kinetic. J Radioanal Nucl Chem 319, 245–255 (2019). https://doi.org/10.1007/s10967-018-6289-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-018-6289-z