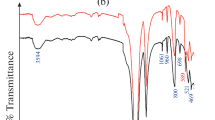
Abstract
The removal of 90Sr in liquid radioactive wastes is an important issue for waste disposal. In this paper, mesoporous silica SBA-15 (Santa Barbara amorphous 15) was employed to be a new sorbent for the sorption of Sr(II) from aqueous solution under different experimental conditions. Results showed that Sr(II) sorption with SBA-15 is fast with the equilibrium time within 100 min. The sorption is pH-dependent and good adsorption is at a relatively high pH and low ionic strength. Results suggest that SBA-15 is a potential sorbent for fast and efficient removal of Sr(II) from Sr-contaminated waste water.











Similar content being viewed by others
References
Yavari R, Huang YD, Mostofizadeh A (2010) Sorption of strontium ions from aqueous solutions by oxidized multiwall carbon nanotubes. J Radioanal Nucl Chem 285:703–710
Cao JG, Gu P, Zhao J, Zhang D, Deng Y (2010) Removal of strontium from an aqueous solution using co-precipitation followed by microfiltration (CPMF). J Radioanal Nucl Chem 285:539–546
Makrlík E (2004) Solvent extraction of strontium picrate from water into nitrobenzene in the presence of benzo-15-crown-5. J Radioanal Nucl Chem 262:513–515
Vorster C, Walt TN, Coetzee PP (2008) Ion exchange separation of strontium and rubidium on Dowex 50W-X8, using the complexation properties of EDTA and DCTA. Anal Bioanal Chem 392:287–296
Mulani K, Daniels S, Rajdeo K, Tambe S, Chavan N (2013) Adsorption of chromium(VI) from aqueous solutions by coffee polyphenol-formaldehyde/acetaldehyde resins. J Polym 2013:1–11
Chirkst DE, Litvinova TE, Cheremisina OV, Ivanov MV, Mironenkova NA (2003) Isotherm of strontium sorption on clay. Russ J Appl Chem 76:727–730
Rani R, Sasidhar P (2012) Geochemical and thermodynamic aspects of sorption of strontium on kaolinite dominated clay samples at Kalpakkam. Environ Earth Sci 65:1265–1274
Dyer A, James NU, Terrill NJ (1999) Uptake of cesium and strontium radioisotopes onto pillared clays. J Radioanal Nucl Chem 240:589–592
Kulyukhin S, Krasavina E, Mizina L, Rumer I, Tanashchuk N, Konovalova N (2005) Sorption of iodine, cesium, and strontium radionuclides on alkaline-earth hydroxyphosphates from aqueous solutions. Radiochemistry 47:84–88
Ivanets AI, Shashkova IL, Kitikova NV, Drozdova NV, Saprunova NV, Radkevich AV, Kulbitskaya LV (2014) Sorption of strontium ions from solutions onto calcium and magnesium phosphates. Radiochemistry 56:32–37
Rosskopfová O, Galamboš M, Rajec P (2011) Study of sorption processes of strontium on the synthetic hydroxyapatite. J Radioanal Nucl Chem 287:715–722
Wang X, Ye J (2008) Variation of crystal structure of hydroxyapatite in calcium phosphate cement by the substitution of strontium ions. J Mater Sci Mater Med 19:1183–1186
Lazić S, Vuković Ž (1991) Ion-exchange of strontium on synthetic hydroxyapatite. J Radioanal Nucl Chem 149:161–168
Kasap S, Tel H, Piskin S (2011) Preparation of TiO2 nanoparticles by sonochemical method, isotherm, thermodynamic and kinetic studies on the sorption of strontium. J Radioanal Nucl Chem 289:489–495
Merceille A, Weinzaepfel E, Barré Y, Grandjean A (2011) Effect of the synthesis temperature of sodium nonatitanate on batch kinetics of strontium-ion adsorption from aqueous solution. Adsorption 17:967–975
Galamboš M, Kufčáková J, Rosskopfová O, Rajec P (2010) Adsorption of cesium and strontium on natrified bentonites. J Radioanal Nucl Chem 283:803–813
Kresge CT, Leonowicz ME, Roth WJ, Vartuli JC, Beck JS (1992) Ordered mesoporous molecular sieves synthesized by a liquid–crystal template mechanism. Nature 359:710–712
Amit K, Santosh Y, Panagiotis G, Smirniotis N, Pinto G (2006) Synthesis of ordered large pore SBA-15 spherical particles for adsorption of biomolecules. J Chromatogr 1122:13–20
Liu AM, Hidajat K, Kawi S, Zhao DY (2000) A new class of hybrid mesoporous materials with functionalized organic monolayers for selective adsorption of heavy metal ions. Chem Commun 13:1145–1146
Luan Z, Maes E, vander Heide P, Zhao D, Czernuszewicz R, Kevan L (1999) Incorporation of titanium into mesoporous silica molecular sieve SBA-15. Chem Mater 11:3680–3686
Luan Z, Hartmann M, Zhao D, Zhou W, Kevan L (1999) Alumination and ion exchange of mesoporous SBA-15 molecular sieves. Chem Mater 11:1621–1627
Basaldella EI, Tara JC, Armenta GA, Iglesias ME (2007) Pt-promoted Cu/SBA-15 catalysts with excellent performance for chemoselective hydrogenation of dimethyl oxalate to ethylene glycol. J Porous Mater 14:273–278
Zheng XL, Lin HQ, Zheng JW, Ariga H, Asakura K, Yuan YZ (2014) Top Catal 57:1015–1025
Tao Z, Xie Y, Goodisman J, Asefa T (2010) Isomer-dependent adsorption and release of cis- and trans-platin anticancer drugs by mesoporous silica nanoparticles. Langmuir 26:8914–8924
Jeong EY, Ansari MB, Mo YH, Park SE (2011) Removal of Cu(II) from water by tetrakis(4-carboxyphenyl) porphyrin-functionalized mesoporous silica. J Hazard Mater 185:1311–1317
Liu Y, Liu Z, Gao J, Dai J, Han J, Wang Y, Xie J, Yan Y (2011) Selective adsorption behavior of Pb(II) by mesoporous silica SBA-15-supported Pb(II)-imprinted polymer based on surface molecularly imprinting technique. J Hazard Mater 186:197–205
Liu Y, Yuan L, Yuan Y, Lan J, Li Z, Feng Y, Zhao Y, Chai Z, Shi W (2012) A high efficient sorption of U(VI) from aqueous solution using amino-functionalzied SBA-15. J Radioanal Nucl Chem 292:803–810
Wang X, Yuan L, Wang Y, Li Z, Lan J, Liu Y, Feng Y, Zhao Y, Chai Z (2012) Mesoporous silica SBA-15 functionalized with phosphonate and amino groups for uranium uptake. Sci China Chem 55:1705–1711
Wang J, Liu Q (2005) Structural change and characterization in nitrogen-incorporated SBA15 oxynitride mesoporous materials via different thermal history. Microporous Mesoporous Mater 83:225–232
Wu BP, Tong ZW, Yuan XD (2012) Synthesis, characterization and catalytic application of mesoporous molecular sieves SBA-15 functionalized with phosphoric acid. J Porous Mater 19:641–647
Zhao DY, Yang PD, Margolese DI, Chmelkab BF, Stucky GD (1998) Synthesis of continuous mesoporous silica thin films with three-dimensional accessible pore structures. Chem Commun 22:2499–2500
Bois L, Bonhomme A, Ribes A, Pais B, Raffin G, Tessier F (2003) Functionalized silica for heavy metal ions adsorption. Colloids Surf A 221:221–230
Gallo J, Bisio C, Gatti G, Marchese L, Pastore H (2010) Physicochemical characterization and surface acid properties of mesoporous [Al]-SBA-15 obtained by direct synthesis. Langmuir 26:5791–5800
Cole T, Bidoglio G, Soupioni M, Gorman M, Gibson N (2000) Diffusion mechanisms of multiple strontium species in clay. Geochim Cosmochim Acta 64:385–396
Yang ST, Zhao DL, Sheng GD, Guo ZQ, Sun YB (2011) Investigation of solution chemistry effects on sorption behavior of radionuclide 64Cu(II) on illite. J Radioanal Nucl Chem 289:467–477
Wu ZJ, Liu HN, Zhang HF (2010) Research progress on mechanisms about the effect of ionic strength on adsorption. Environ Chem 29:997–1003
Oladoja NA, Aboluwoye CO, Oladimeji YB (2008) Kinetics and isotherm studies on methylene blue adsorption onto ground palm kernel coat. Turk J Eng Environ Sci 32:303–312
Lin SH, Juang RS (2002) Heavy metal removal from water by sorption using surfactant-modified montmorillonite. J Hazard Mater B92:315–326
Langmuir I (1916) The constitution and fundamental properties of solids and liquids. J Am Chem Soc 38:2221–2295
Freundlich HMF (1906) Uber die adsorption in losungen. J Phys Chem 57:385–470
Acknowledgments
The authors gratefully acknowledge the financial support from the National Science Foundation (21367001), Jiangxi Educational Committee Foundation (GJJ13471) and Fundamental Science on Radioactive Geology and Exploration Technology Laboratory of East China Institute of Technology (2011RGET015). State Key Laboratory Breeding Base of Nuclear Resources and Environment, East China Institute of Technology,provided technical assistance (NRE1318).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, N., Liu, S., Jiang, L. et al. Adsorption of strontium from aqueous solution by silica mesoporous SBA-15. J Radioanal Nucl Chem 303, 1671–1677 (2015). https://doi.org/10.1007/s10967-014-3681-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-014-3681-1