Abstract
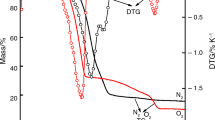
The effect of the structure of a mixture of industrially produced iron and iron oxide on the decomposition of trichloroethylene (TCE) was investigated by gas chromatography, scanning electron microscopy, Fourier transform infrared spectroscopy, energy dispersive X-ray analysis, X-ray diffractometry, and 57Fe-Mössbauer spectroscopy. The concentration of 10 mg L−1 TCE aqueous solution decreased to 0.41, 0.52, 0.26, and 0.09 mg L−1 when stirred for 7 days with iron–iron oxide mixtures having mass ratios of 2:8, 3:7, 4:6, and 5:5, respectively. The Mössbauer spectra of the mixtures after leaching were composed of two sextets with respective isomer shifts (δ) and internal magnetic fields (H) of 0.29±0.01 mm s−1 and 48.8±0.1 T, and 0.64±0.01 mm s−1 and 45.5±0.1 T, attributed to the Fe3+ species in tetrahedral (T d) and the Fe2+ and Fe3+ mixed species (Fe2.5+) in octahedral (O h) sites, respectively. Mössbauer spectra of a 3:7 mass ratio iron–iron oxide mixture showed a gradual decrease in the absorption area (A) of zero valent iron (Fe0) from 40.6. to 12.6, 13.2, 3.8 2.8, and 1.0±0.5 % and an increase in A of Fe3O4 from 31.8 to 59.4, 71.4, 93.2, 95.6, and 98.0±0.5 % after leaching with 10 mg L−1 TCE aqueous solution for 1, 2, 3, 7, and 10 days, respectively. Consistent values of the first-order rate constant were calculated as 0.32 day−1 for Fe0 oxidation, 0.34 day−1 for Fe3O4 production, and 0.30 day−1 for TCE decomposition, which indicates that the oxidation of Fe0 was the rate-controlling factor for Fe3O4 production and TCE decomposition. It is concluded from the experimental results that an iron–iron oxide mixture is very effective for the decomposition of TCE.










Similar content being viewed by others
References
Section of Waterworks, Bureau of Health, Ministry of Health and Welfare (2011). http://www.mhlw.go.jp/topics/bukyoku/kenkou/suido/kijun/suishitsu23.html (in Japanese, Accessed 4 July 2012)
Serpieri N, Moneti G, Pieraccini G, Donati R, Mariottini E, Dolara P (2000) Urban Water 2:13
Leahy JG, Carrington TE, Eley MH (2004) J Environ Qual 33:1556
Leahy JG, Tracy KD, Eley MH (2003) Biotechnol Lett 25:479
Rabbu MF, Clarck B, Gale RJ, Ozsu-Acar E, Pardue J, Jackson A (2000) Waste Manag 20:279
Cheng S-F, Wu S-Ch (2001) Chemosphere 43:1023
Prommer H, Aziz LH, Bolaño N, Taubald H, Schüth C (2008) J Contam Hydrol 97:13
Greenwood NN, Gibb TC (1971) Mössbauer spectroscopy. Chapman and Hall Ltd., London
Cranshaw TE, Longworth G (1984). In: Long GJ (ed) Mössbauer spectroscopy applied to inorganic chemistry, Chap 7, vol 1. Plenum Press, New York
Sun Y-k, Ma M, Zhang Y, Gu N (2004) Colloids surf A Physicochem Eng Aspects 245:15
Shibano K, Kubuki S, Akiyama K, Homonnay Z, Kuzmann E, Nishida T (2012) In: Proceedings of the specialist research meeting on “science and engineering of unstable nuclei and their uses on condensed matter physics” (KURRI-KR-168, ISSN 1342-0852), p 74
Oliveira DQL, Oliveira LCA, Murad E, Fabris JD, Silva AC, de Menezes LM (2010) Hyperfine Interact 195:27
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kubuki, S., Shibano, K., Akiyama, K. et al. Effect of the structural change of an iron–iron oxide mixture on the decomposition of trichloroethylene. J Radioanal Nucl Chem 295, 23–30 (2013). https://doi.org/10.1007/s10967-012-1933-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-012-1933-5