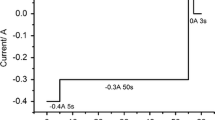
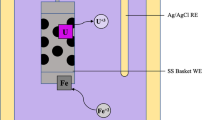
Abstract
In order to enhance the efficiency of pyrochemical technology, especially electrorefining process, physicochemical data of trivalent uranium in LiCl–KCl eutectic at 773 K were measured, including molar absorptivity, formal potential and diffusion coefficient of U3+ ions. The molar absorptivities of U3+ were determined to be 765 ± 48 and 686 ± 39 M−1 cm−1 at 465 and 550 nm, respectively. The formal potential of U3+/U4+ redox couple and diffusion coefficient of U3+ ions were measured to be −0.308 V vs. Ag/Ag+ and 8.7 × 10−6 M−1 cm−1, respectively. To elucidate the chemical behavior of U3+ ions under the existence of oxide ions, U3+ ions were reacted with oxides ions in situ produced at the LiCl–KCl media. Surprisingly, it was revealed from XRD patterns that UO2 was formed from the reaction between U3+ ions and O2− ions with the molar ratio of 1:1.





Similar content being viewed by others
References
Jeong SM, Park SB, Hong SS, Seo CS, Park SW (2006) Electrolytic production of metallic uranium from U3O8 in a 20-kg batch scale reactor. J Radioanal Nucl Chem 268:349–356
Park BH, Park SB, Jeong SM, Seo CS, Park SW (2006) Electrolytic reduction of spent oxide fuel in a molten LiCl–Li2O system. J Radioanal Nucl Chem 270:575–583
Fanxing G, Changshui W, Lisheng L, Jianhua G, Shangwen C, Li C, Ruixue L, Yinggen O (2009) Electrode processes of uranium ions and electrodeposition of uranium in molten LiCl–KCl. J Radioanal Nucl Chem 280:207–218
Cassayre L, Caravaca C, Jardin R, Malmbeck R, Masset P, Mendes E, Serp J, Soucek P, Glatz JP (2008) On the formation of U-Al alloys in the molten LiCl–KCl eutectic. J Nucl Mater 378:79–85
Fujii T, Moriyama H, Yamana H (2003) Electronic absorption spectra of lanthanides in a molten chloride I. Molar absorptivity measurement of neodymium(III) in molten eutectic mixture of LiCl–KCl. J Alloy Comp 351:L6–L9
Gruen DM, Mcbeth RL (1959) Oxidation states and complex ions of uranium in fused chlorides and nitrates. J Inorg Nucl Chem 9:290–301
Nagai T, Uehara A, Fujii T, Shirai O, Sato N, Yamana H (2005) Redox equilibrium of U4+/U3+ in molten NaCl-2CsCl by UV–Vis spectrophotometry and cyclic voltammetry. J Nucl Sci Tech 42:1025–1031
Nagai T, Fujii T, Shirai O, Yamana H (2004) Study on redox equilibrium of UO2 2+/UO2 + in molten NaCl-2CsCl by UV–Vis spectrophotometry. J Nucl Sci Tech 41:690–695
Kim TJ, Jeong YK, Kang JG, Jung Y, Ahn DH, Lee HS (2010) In situ analysis for spontaneous reduction of Eu3+ in LiCl pyroprocessing media at 923 K. J Radioanal Nucl Chem doi: 10.1007/s10967-010-0651-0
Bard AJ, Faulkner LR (2001) Electrochemical methods. John Wiley , New York
Prabhakara Reddy B, Vandarkuzhali S, Subramanian T, Venkatesh P (2004) Electrochemical studies on the redox mechanism of uranium chloride in molten LiCl–KCl eutectic. Electrochim Acta 49:2471–2478
Shirai O, Iwai T, Suzuki Y, Sakamura Y, Tanaka H (1998) Electrochemical behavior of actinide ions in LiCl–KCl eutectic melts. J Alloy Comp 271–273:685–688
De Cordoba G, Caravaca C (2006) Potentiometric study of Sm-O compounds formation in the molten LiCl–KCl eutectic at 450 °C. Determination of a E-pO2− stability diagram. J Phys Chem Solids 67:1862–1868
Suzuki S, Sato A, Tanaka K (1971) Absorption spectra of trivalent uranium in fused alkali chloride. J Jpn Inst Metals 35:589–593
Acknowledgments
This paper was supported by the New Professor Research Program of KUT and the Nuclear R & D program of the Korea Ministry of Education, Science and Technology (MEST).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kim, TJ., Jung, Y., Shim, JB. et al. Study on physicochemical properties of U3+ in LiCl–KCl eutectic media at 773 K. J Radioanal Nucl Chem 287, 347–350 (2011). https://doi.org/10.1007/s10967-010-0689-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-010-0689-z